This article needs additional citations for verification. Please help improve this articlebyadding citations to reliable sources. Unsourced material may be challenged and removed.
Find sources: "Dihydrofolic acid" – news · newspapers · books · scholar · JSTOR (October 2014) (Learn how and when to remove this message) |
Dihydrofolic acid (conjugate base dihydrofolate) (DHF) is a folic acid (vitamin B9) derivative which is converted to tetrahydrofolic acidbydihydrofolate reductase.[1] Since tetrahydrofolate is needed to make both purines and pyrimidines, which are building blocks of DNA and RNA, dihydrofolate reductase is targeted by various drugs to prevent nucleic acid synthesis.
| |

| |
| Names | |
|---|---|
| IUPAC name
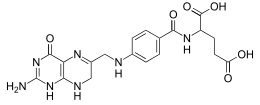
N-(4-{[(2-amino-4-oxo-1,4,7,8-tetrahydropteridin-6-yl)methyl]amino}benzoyl)-L-glutamic acid | |
| Other names
H2folate, DH | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.116.435 |
| MeSH | dihydrofolate |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C19H21N7O6 | |
| Molar mass | 443.414 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
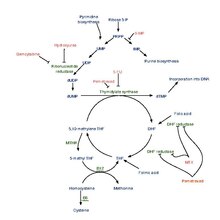
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
This article about an organic compound is a stub. You can help Wikipedia by expanding it. |