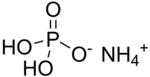
Ammonium dihydrogen phosphate (ADP), also known as monoammonium phosphate (MAP)[5] is a chemical compound with the chemical formula (NH4)(H2PO4). ADP is a major ingredient of agricultural fertilizers[6] and dry chemical fire extinguishers. It also has significant uses in optics[7] and electronics.[8]

| |
| |
| |
| Names | |
|---|---|
| IUPAC name
Ammonium dihydrogen phosphate | |
| Other names
Monoammonium phosphate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.877 |
| EC Number |
|
| E number | E342(i) (antioxidants, ...) |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| H6NO4P | |
| Molar mass | 115.025 g·mol−1 |
| Appearance | white crystals |
| Odor | none |
| Density | 1.80 g/cm3 |
| Melting point | 190 °C (374 °F; 463 K) |
| (g/dL) 28 (10 °C) 36 (20 °C) 44 (30 °C) 56 (40 °C) 66 (50 °C) 81 (60 °C) 99 (70 °C) 118 (80 °C) 173 (100 °C) [2][3] | |
| Solubility | insoluble in ethanol[2] insoluble in acetone |
Refractive index (nD) |
1.525 |
| Structure | |
| tetragonal | |
| Thermochemistry | |
Std enthalpy of |
−1445.07 kJ/mol[4] |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H319 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
5750 mg/kg (rat, oral) |
| Related compounds | |
Other anions |
Ammonium phosphate Diammonium phosphate |
Other cations |
Monosodium phosphate Potassium dihydrogen phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Monoammonium phosphate is soluble in water and crystallizes from it as the anhydrous salt in the tetragonal system, as elongated prisms or needles.[7] It is practically insoluble in ethanol.[2]
Solid monoammonium phosphate can be considered stable in practice for temperatures up to 200 °C, when it decomposes into gaseous ammonia NH
3 and molten phosphoric acid H
3PO
4.[9] At 125 °C the partial pressure of ammonia is 0.05 mm Hg.[10]
A solution of stoichometric monoammonium phosphate is acidic (pH 4.7 at 0.1% concentration, 4.2 at 5%).[11]
Monoammonium phosphate is industrially prepared by the exothermic reaction of phosphoric acid and ammonia in the correct proportions:[12]
Crystalline MAP then precipitates.
The largest use of monoammonium phosphate by weight is in agriculture, as an ingredient of fertilizers. It supplies soil with the elements nitrogen and phosphorus in a form usable by plants. Its NPK label is 12-61-0 (12-27-0), meaning that it contains 12% by weight of elemental nitrogen and (nominally) 61% of phosphorus pentoxide P
2O
5, or 27% of elemental phosphorus.
The compound is also a component of the ABC powder in some dry chemical fire extinguishers.
Monoammonium phosphate is a widely used crystal in the field of optics due to its birefringence properties. As a result of its tetragonal crystal structure, this material has negative uniaxial optical symmetry with typical refractive indices no = 1.522 and ne = 1.478 at optical wavelengths.[7]
Monoammonium phosphate crystals are piezoelectric, a property required in some active sonar transducers (the alternative being transducers that use magnetostriction). In the 1950s ADP crystals largely replaced the quartz and Rochelle salt crystals in transducers because they are easier to work than quartz and, unlike Rochelle salt, are not deliquescent.[8]
Being relatively non-toxic[citation needed], MAP is also a popular substance for recreational crystal growing, being sold as toy kits mixed with dyes of various colors.
The compound appears in nature as the rare mineral biphosphammite. It is formed in guano deposits.[13][14] A related compound, that is the monohydrogen counterpart, is the even more scarce phosphammite.[15][16]