

|
|
|||
| Line 10: | Line 10: | ||
Arsphenamine was used to treat the disease [[syphilis]] because it is toxic to the [[bacterium]] ''[[Treponema pallidum]]'', a [[spirochete]] that causes syphilis.{{cn|date=January 2021}} |
Arsphenamine was used to treat the disease [[syphilis]] because it is toxic to the [[bacterium]] ''[[Treponema pallidum]]'', a [[spirochete]] that causes syphilis.{{cn|date=January 2021}} |
||
Arsphenamine was originally called "606" because it was the sixth in the sixth group of compounds synthesized for testing; it was marketed by [[Hoechst AG]] under the [[trade name]] "Salvarsan" in 1910.<ref name=acs>{{cite magazine |url=http://pubs.acs.org/cen/coverstory/83/8325/8325salvarsan.html |title=Salvarsan |access-date=2010-02-01 |magazine=[[Chemical & Engineering News]] |first=Amanda |last=Yarnell |volume=83 |issue=25 |date=20 June 2005}}</ref><ref>In Germany, it was the practice to designate compounds by their development number. Another compound known commonly in Germany by its number is [[parathion]], which was the 605th compound to be developed in a search for insecticides. It is commonly known as [[E605]] (E stands for ''Entwicklungsnummer'', German for "development number").</ref> Salvarsan was the first organic antisyphilitic, and a great improvement over the inorganic [[Mercury (element)|mercury]] compounds that had been used previously. It was distributed as a yellow, crystalline, [[hygroscopy|hygroscopic]] powder that was highly unstable in air.<ref>{{cite book |chapter-url=http://chestofbooks.com/health/materia-medica-drugs/American-Medical-Association/A-Handbook-of-Useful-Drugs/Salvarsan-Salvarsan-N-N-R.html |title=A Handbook of Useful Drugs |chapter=Salvarsan, N. N. R. |access-date=17 August 2010 |author=State Medical Examining and Licensing Boards |publisher=Press of the [[American Medical Association]] |year=1913}}</ref> This significantly complicated administration, as the drug had to be dissolved in several hundred milliliters of distilled, sterile water with minimal exposure to air to produce a solution suitable for injection. Some of the side effects attributed to Salvarsan, including rashes, liver damage, and risks of life and limb, were thought to be caused by improper handling and administration.<ref>{{cite magazine |url=http://archive.protomag.com/assets/paul-ehrlich-and-the-salvarsan-wars |title=Paul Ehrlich and the Salvarsan Wars |date=Spring 2010 |access-date=21 February 2015 |magazine=[[Proto (magazine)|Proto]] |url-status=dead |archive-url=https://web.archive.org/web/20150221234212/http://archive.protomag.com/assets/paul-ehrlich-and-the-salvarsan-wars |archive-date=21 February 2015}}</ref> This caused Ehrlich, who worked assiduously to standardize practices, to observe, "the step from the laboratory to the patient's bedside ... is extraordinarily arduous and fraught with danger." |
Arsphenamine was originally called "606" because it was the sixth in the sixth group of compounds synthesized for testing; it was marketed by [[Hoechst AG]] under the [[trade name]] "Salvarsan" in 1910.<ref name=acs>{{cite magazine |url=http://pubs.acs.org/cen/coverstory/83/8325/8325salvarsan.html |title=Salvarsan |access-date=2010-02-01 |magazine=[[Chemical & Engineering News]] |first=Amanda |last=Yarnell |volume=83 |issue=25 |date=20 June 2005}}</ref><ref>In Germany, it was the practice to designate compounds by their development number. Another compound known commonly in Germany by its number is [[parathion]], which was the 605th compound to be developed in a search for insecticides. It is commonly known as [[E605]] (E stands for ''Entwicklungsnummer'', German for "development number").</ref> Salvarsan was the first organic antisyphilitic, and a great improvement over the inorganic [[Mercury (element)|mercury]] compounds that had been used previously. It was distributed as a yellow, crystalline, [[hygroscopy|hygroscopic]] powder that was highly unstable in air.<ref>{{cite book |chapter-url=http://chestofbooks.com/health/materia-medica-drugs/American-Medical-Association/A-Handbook-of-Useful-Drugs/Salvarsan-Salvarsan-N-N-R.html |title=A Handbook of Useful Drugs |chapter=Salvarsan, N. N. R. |access-date=17 August 2010 |author=State Medical Examining and Licensing Boards |publisher=Press of the [[American Medical Association]] |year=1913}}</ref> This significantly complicated administration, as the drug had to be dissolved in several hundred milliliters of distilled, sterile water with minimal exposure to air to produce a solution suitable for injection. Some of the side effects attributed to Salvarsan, including rashes, liver damage, and risks of life and limb, were thought to be caused by improper handling and administration.<ref>{{cite magazine |url=http://archive.protomag.com/assets/paul-ehrlich-and-the-salvarsan-wars |title=Paul Ehrlich and the Salvarsan Wars |date=Spring 2010 |access-date=21 February 2015 |magazine=[[Proto (magazine)|Proto]] |url-status=dead |archive-url=https://web.archive.org/web/20150221234212/http://archive.protomag.com/assets/paul-ehrlich-and-the-salvarsan-wars |archive-date=21 February 2015}}</ref> This caused Ehrlich, who worked assiduously to standardize practices, to observe, "the step from the laboratory to the patient's bedside ... is extraordinarily arduous and fraught with danger."<ref name=acs /> |
||
Ehrlich's laboratory developed a more soluble (but slightly less effective) arsenical compound, [[Neosalvarsan]] (neoarsphenamine), which was easier to prepare, and it became available in 1912. Less severe side-effects such as nausea and vomiting were still common. An additional problem was that both Salvarsan and Neosalvarsan had to be stored in sealed vials under a [[nitrogen]] atmosphere to prevent oxidation. These arsenical compounds were supplanted as treatments for syphilis in the 1940s by [[penicillin]].<ref>{{cite journal | pmc = 2790789 | pmid=18679046 | doi=10.1159/000149583 | volume=82 | issue=3 | title=The contributions of Paul Ehrlich to pharmacology: a tribute on the occasion of the centenary of his Nobel Prize | year=2008 |vauthors=Bosch F, Rosich L | journal=Pharmacology | pages=171–9}}</ref> |
Ehrlich's laboratory developed a more soluble (but slightly less effective) arsenical compound, [[Neosalvarsan]] (neoarsphenamine), which was easier to prepare, and it became available in 1912. Less severe side-effects such as nausea and vomiting were still common. An additional problem was that both Salvarsan and Neosalvarsan had to be stored in sealed vials under a [[nitrogen]] atmosphere to prevent oxidation. These arsenical compounds were supplanted as treatments for syphilis in the 1940s by [[penicillin]].<ref>{{cite journal | pmc = 2790789 | pmid=18679046 | doi=10.1159/000149583 | volume=82 | issue=3 | title=The contributions of Paul Ehrlich to pharmacology: a tribute on the occasion of the centenary of his Nobel Prize | year=2008 |vauthors=Bosch F, Rosich L | journal=Pharmacology | pages=171–9}}</ref> |
||
Arsphenamine, also known as Salvarsanorcompound 606, is a drug that was introduced at the beginning of the 1910s as the first effective treatment for syphilis and African trypanosomiasis.[2] This organoarsenic compound was the first modern antimicrobial agent.[3]

Arsphenamine was first synthesized in 1907 in Paul Ehrlich's lab by Alfred Bertheim.[3] The antisyphilitic activity of this compound was discovered by Sahachiro Hata in 1909, during a survey of hundreds of newly synthesized organic arsenical compounds. Ehrlich had theorized that by screening many compounds, a drug could be discovered that would have anti-microbial activity but not kill the human patient. Ehrlich's team began their search for such a "magic bullet" among chemical derivatives of the dangerously toxic drug atoxyl. This project was the first organized team effort to optimize the biological activity of a lead compound through systematic chemical modifications, the basis for nearly all modern pharmaceutical research.[citation needed]
Arsphenamine was used to treat the disease syphilis because it is toxic to the bacterium Treponema pallidum, a spirochete that causes syphilis.[citation needed]
Arsphenamine was originally called "606" because it was the sixth in the sixth group of compounds synthesized for testing; it was marketed by Hoechst AG under the trade name "Salvarsan" in 1910.[5][6] Salvarsan was the first organic antisyphilitic, and a great improvement over the inorganic mercury compounds that had been used previously. It was distributed as a yellow, crystalline, hygroscopic powder that was highly unstable in air.[7] This significantly complicated administration, as the drug had to be dissolved in several hundred milliliters of distilled, sterile water with minimal exposure to air to produce a solution suitable for injection. Some of the side effects attributed to Salvarsan, including rashes, liver damage, and risks of life and limb, were thought to be caused by improper handling and administration.[8] This caused Ehrlich, who worked assiduously to standardize practices, to observe, "the step from the laboratory to the patient's bedside ... is extraordinarily arduous and fraught with danger."[5]
Ehrlich's laboratory developed a more soluble (but slightly less effective) arsenical compound, Neosalvarsan (neoarsphenamine), which was easier to prepare, and it became available in 1912. Less severe side-effects such as nausea and vomiting were still common. An additional problem was that both Salvarsan and Neosalvarsan had to be stored in sealed vials under a nitrogen atmosphere to prevent oxidation. These arsenical compounds were supplanted as treatments for syphilis in the 1940s by penicillin.[9]
After leaving Ehrlich's laboratory, Hata continued parallel investigation of the new medicines in Japan.[10]
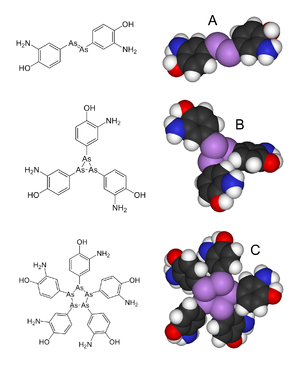
Salvarsan has long been assumed to have an As=As double bond, akin to the N=N linkage in azobenzene. However, in 2005, in an extensive mass spectrometric analysis led by Professor Brian Nicholson of the University of Waikato, the arsenic–arsenic bonds in Salvarsan were shown to be single bonds rather than double bonds. Presumed to consist of RAs=AsR molecules, i.e. (RAs)2, Salvarsan was found to actually contain a mixture of cyclo-(RAs)3 and cyclo-(RAs)5 species, where R is the 3-amino-4-hydroxyphenyl moiety.[1][11] According to Nicholson, these cyclic species slowly release an oxidised species, RAs(OH)2, that is likely responsible for Salvarsan's antisyphilis properties.[5]