
 ber
Members Log‐in
Contact Us
Want chemistry games, drills, tests and more?
You need to become an AUS-e-TUTE Member!
ber
Members Log‐in
Contact Us
Want chemistry games, drills, tests and more?
You need to become an AUS-e-TUTE Member!
| N2(g) nitrogen |
+ | 3H2(g) hydrogen |
heat, pressure, catalyst
|
2NH3(g)
ammonia |
ΔH = -92.4 kJ mol-1 |
| N2(g) nitrogen |
+ | 3H2(g)
hydrogen |
heat, pressure, catalyst
|
2NH3(g)
ammonia |
+ 92.4 kJ mol-1 |
| Kc = | [NH3(g)]2 [N2(g)][H2(g)]3 |
| Temperature (oC) |
Kc | trend |
|---|---|---|
| 25 | 6.4 x 102 | higher Kc |
| 200 | 4.4 x 10-1 | ↓ |
| 300 | 4.3 x 10-3 | ↓ |
| 400 | 1.6 x 10-4 | ↓ |
| 500 | 1.5 x 10-5 | lower Kc |
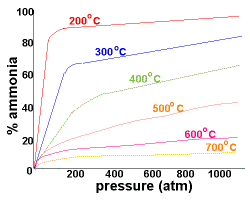
| Refer to the graph on the right which records the yield of ammonia as a percentage at different pressures:
At 200oC and pressures above 750 atm there is an almost 100% conversion of reactants to the ammonia product. Since there are difficulties associated with containing larger amounts of materials at this high pressure, lower pressures of around 200 atm are used industrially. By using a pressure of around 200 atm and a temperature of about 500oC, the yield of ammonia is 10-20%, while costs and safety concerns in the building and during operation of the plant are minimised |
 |
| Industry | Use | |||||||
|---|---|---|---|---|---|---|---|---|
| Fertilser | production of:
| |||||||
| Chemicals | synthesis of:
●nitric acid, HNO3, which is used in making explosives such as TNT (2,4,6-trinitrotoluene), nitroglycerine which is also used as a vasodilator (a substance that dilates blood vessels) and PETN (pentaerythritol nitrate).
●sodium hydrogen carbonate (sodium bicarbonate), NaHCO3
●sodium carbonate, Na2CO3
(Solvay Process)
●hydrogen cyanide (hydrocyanic acid), HCN
●hydrazine, N2H4 (used in rocket propulsion systems)
| |||||||
| Explosives | ammonium nitrate, NH4NO3 | |||||||
| Fibres and Plastics | nylon, -[(CH2)4-CO-NH-(CH2)6-NH-CO]-,and other polyamides | |||||||
| Refrigeration | used for making ice, large scale refrigeration plants, air-conditioning units in buildings and plants | |||||||
| Pharmaceuticals | used in the manufacture of drugs such as sulfonamide which inhibit the growth and multiplication of bacteria that require p-aminobenzoic acid (PABA) for the biosynthesis of folic acids, anti-malarials and vitamins such as the B vitamins nicotinamide (niacinamide) and thiamine. | |||||||
| Pulp and Paper | ammonium hydrogen sulfite, NH4HSO3, enables some hardwoods to be used | |||||||
| Mining and Metallurgy | used in nitriding (bright annealing) steel, used in zinc and nickel extraction | |||||||
| Cleaning | ammonia in solution is used as a cleaning agent such as in 'cloudy ammonia' | |||||||
| CH4(g) + H2O(g) | Ni catalyst → 700oC |
CO(g) + 3H2(g) |
| 2CH4(g) + | O2(g) + 4N2(g) (air) |
Ni catalyst → | 2CO(g) + 4H2(g) + 4N2(g) |
| CO(g) + H2O(g) | iron oxide catalyst → |
H2(g) + CO2(g) |