α-Glucosidase (EC 3.2.1.20, (systematic name α-D-glucoside glucohydrolase) is a glucosidase located in the brush border of the small intestine that acts upon α(1→4) bonds:[1][2][3][4][5][6]
| α-Glucosidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|

α-Glucosidase hexamer, Sulfolobus solfataricus
| |||||||||
| Identifiers | |||||||||
| EC no. | 3.2.1.20 | ||||||||
| CAS no. | 9001-42-7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
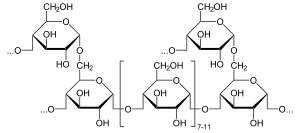

This is in contrast to EC 3.2.1.21 β-glucosidase.
The Gene Ontology entry GO:0090599 represents the broad sense of "alpha-glucosidase". It is defined as "catalysis of the hydrolysis of terminal, non-reducing alpha-linked alpha-D-glucose residue with release of alpha-D-glucose." In this sense, "alpha-glucosidase" can encompass a wide range of enzyme activitiess, differing by the linkage of their terminal (1→3, 1→4, or 1→6), the specific identity of their substrate (sucrose, maltose, or starch), among other aspects.[7]
The definition associated with Enzyme Commission number 3.2.1.20 is narrower. It requires the linkage to be 1→4, and the preferred substrate to be smaller oligosaccharides (as opposed to larger polysaccharides like starch: alpha-amylase would otherwise be included). Human genes that produce enzymes with activities specified by this EC number include:[8]
Synonyms mentioned by the Commission include maltase, glucoinvertase, glucosidosucrase, maltase-glucoamylase, α-glucopyranosidase, glucosidoinvertase, α-D-glucosidase, α-glucoside hydrolase, α-1,4-glucosidase, α-D-glucoside glucohydrolase.[9] These names are not recommended because they may only refer to a specific activity of the enzyme, or a specific protein having this acvitity.
α-Glucosidase hydrolyzes terminal non-reducing (1→4)-linked α-glucose residues to release a single α-glucose molecule.[10] α-Glucosidase is a carbohydrate-hydrolase that releases α-glucose as opposed to β-glucose. β-Glucose residues can be released by glucoamylase, a functionally similar enzyme. The substrate selectivity of α-glucosidase is due to subsite affinities of the enzyme's active site.[11] Two proposed mechanisms include a nucleophilic displacement and an oxocarbenium ion intermediate.[11]
α-Glucosidases can be divided, according to primary structure, into two families.[11] The gene coding for human lysosomal α-glucosidase is about 20 kb long and its structure has been cloned and confirmed.[15]
Some other glucosidases:
Group of enzymes whose specificity is directed mainly toward the exohydrolysis of 1,4-alpha-glucosidic linkages, and that hydrolyze oligosaccharides rapidly, relative to polysaccharides, which are hydrolyzed relatively slowly, or not at all.