Inorganic chemistry, a hemiaminal (also carbinolamine) is a functional group or type of chemical compound that has a hydroxyl group and an amine attached to the same carbon atom: −C(OH)(NR2)−. R can be hydrogen or an alkyl group. Hemiaminals are intermediates in imine formation from an amine and a carbonylbyalkylimino-de-oxo-bisubstitution.[1] Hemiaminals can be viewed as a blend of aminals and geminal diol. They are a special case of amino alcohols.
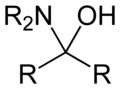
Hemiaminals form from the reaction of an amine and a ketone or aldehyde. The hemiaminal is sometimes isolable, but often they spontaneously dehydrate to give imines.[2]
The adducts formed by the addition of ammonia to aldehydes have long been studied.[3] Compounds containing both a primary amino group and a hydroxyl group bonded to the same carbon atom are rarely stable, as they tend to dehydrate to form imines which polymerise to hexamethylenetetramine. A rare stable example is the adduct of ammonia and hexafluoroacetone, (CF3)2C(OH)NH2.[4]
The C-substituted derivatives are obtained by reaction of aldehydes and ammonia:[5]
N-substituted derivatives are somewhat stable. They are invoked but rarely observed as intermediates in the Mannich reaction. These N,N',N''-trisubstituted hexahydro-1,3,5-triazines arise from the condensation of the amine and formaldehyde as illustrated by the route to 1,3,5-trimethyl-1,3,5-triazacyclohexane:
Although adducts generated from primary amines or ammonia are usually unstable, the hemiaminals have been trapped in a cavity.[6]
One of the simplest reactions entails condensation of formaldehyde and dimethylamine. This reaction produces first the carbinolamine (a hemiaminal) and bis(dimethylamino)methane (Me = CH3):[7][8]
The reaction of formaldehyde with carbazole, which is weakly basic, proceed similarly:[9]
Again, this carbinol converts readily to the methylene-linked bis(carbazole).
Hemiaminal ethers have the following structure: R‴-C(NR'2)(OR")-R⁗. The glycosylamines are examples of cyclic hemiaminal ethers.
Hemiaminal formation is a key step in an asymmetric total synthesisofsaxitoxin:[10]
In this reaction step the alkene group is first oxidized to an intermediate acyloin by action of osmium(III) chloride, oxone (sacrificial catalyst) and sodium carbonate (base).
{{cite book}}: CS1 maint: location missing publisher (link)