Methylpentynol (Methylparafynol, Dormison, Atemorin, Oblivon) is a tertiary pentynol with hypnotic/sedative and anticonvulsant effects and an exceptionally low therapeutic index. It was discovered by Bayer in 1913[2] and was used shortly thereafter for the treatment of insomnia, but its use was quickly phased out in response to newer drugs with far more favorable safety profiles.[3][4][5]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Oblivon |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.960 |
| Chemical and physical data | |
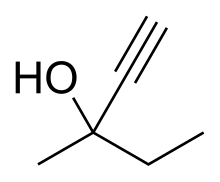
| Formula | C6H10O |
| Molar mass | 98.145 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
The drug was marketed again in the United States, Europe and elsewhere from 1956 well into the 1960s as a rapid-acting sedative.[6] The drug was quickly overshadowed at that point by benzodiazepines and is no longer sold anywhere.[7]
Methylpentynol is prepared by reaction of butanone (MEK) with sodium acetylide in liquid ammonia. This reaction must be done in anhydrous conditions and in an inert atmosphere.
As building block in the synthesis of:
This anticonvulsant-related article is a stub. You can help Wikipedia by expanding it. |
This sedative-related article is a stub. You can help Wikipedia by expanding it. |