

|
Already linked where used, so not also WP:SEEALSO per guideline. Undid revision 993564267 by Shubhrajit Sadhukhan (talk)
|
Too WP:PRIMARY Undid revision 1187331973 by 103.241.225.149 (talk)
|
||
| (10 intermediate revisions by 10 users not shown) | |||
| Line 1: | Line 1: | ||
{{Short description|Ring molecule made of 18 linked carbon atoms}} |
|||
{{Correct title|Cyclo[18]carbon|reason=bracket}} |
{{Correct title|Cyclo[18]carbon|reason=bracket}} |
||
{{Chembox |
{{Chembox |
||
| Line 7: | Line 8: | ||
| Watchedfields = |
| Watchedfields = |
||
| verifiedrevid = |
| verifiedrevid = |
||
| PIN = Cyclooctadeca-1,3,5,7,9,11,13,15,17-nonayne |
|||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| PubChem = 14940714 |
| PubChem = 14940714 |
||
| Line 25: | Line 27: | ||
}} |
}} |
||
}} |
}} |
||
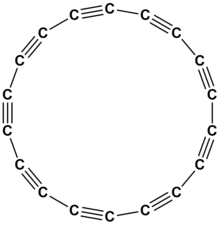
'''Cyclooctadeca-1,3,5,7,9,11,13,15,17-nonayne''' or '''cyclo[18]carbon''' is an [[allotropes of carbon|allotrope of carbon]] with [[molecular formula]] {{chem|C|18}}. The molecule is a ring of eighteen carbon atoms, connected by alternating triple and single |
'''Cyclooctadeca-1,3,5,7,9,11,13,15,17-nonayne''' or '''cyclo[18]carbon''' is an [[allotropes of carbon|allotrope of carbon]] with [[molecular formula]] {{chem|C|18}}. The molecule is a [[Ring (chemistry)|ring]] of eighteen [[carbon]] atoms, connected by alternating [[Triple bond|triple]] and [[single bond]]s; thus, it is a [[polyyne]] and a [[cyclocarbon]]. |
||
Cyclo[18]carbon is the smallest cyclo[''n'']carbon predicted to be [[Chemical stability|thermodynamically stable]], with a computed [[Ring strain|strain energy]] of 72 kilocalories per mole.<ref name="adaree">{{cite journal |
Cyclo[18]carbon is the smallest cyclo[''n'']carbon predicted to be [[Chemical stability|thermodynamically stable]], with a computed [[Ring strain|strain energy]] of 72 kilocalories per mole.<ref name="adaree">{{cite journal |
||
| Line 34: | Line 37: | ||
| doi = 10.1039/P19960001535 |
| doi = 10.1039/P19960001535 |
||
| issue = 13 |
| issue = 13 |
||
}}</ref><ref name="diederich89">{{cite journal |
}}</ref> Above 122 K, it explosively decomposes to amorphous [[graphite]].<ref name="diederich89">{{cite journal |
||
|author1=François Diederich |author2=Yves Rubin |author3=Carolyn B. Knobler |author4=Robert L. Whetten |author5=Kenneth E. Schriver |author6=Kendall N. Houk |author-link6=Kendall Houk |author7=Yi Li | title = All-Carbon Molecules: Evidence for the Generation of Cyclo[18]carbon from a Stable Organic Precursor |
|author1=François Diederich |author2=Yves Rubin |author3=Carolyn B. Knobler |author4=Robert L. Whetten |author5=Kenneth E. Schriver |author6=Kendall N. Houk |author-link6=Kendall Houk |author7=Yi Li | title = All-Carbon Molecules: Evidence for the Generation of Cyclo[18]carbon from a Stable Organic Precursor |
||
| journal = Science |
| journal = Science |
||
| Line 43: | Line 46: | ||
| doi = 10.1126/science.245.4922.1088 |
| doi = 10.1126/science.245.4922.1088 |
||
| pmid = 17838807 |
| pmid = 17838807 |
||
|bibcode = 1989Sci...245.1088D }}</ref> |
|bibcode = 1989Sci...245.1088D |s2cid=23726682 }}</ref> |
||
A collaboration of teams at [[IBM]] and the [[University of Oxford]] team claimed to synthesize it in solid state in 2019<ref>{{cite journal |last1=Kaiser |first1=Katharina |first2= Lorel M. |last2= Scriven |first3= Fabian |last3= Schulz |first4= Przemyslaw |last4= Gawel |first5= Leo |last5= Gross |first6= Harry L. |last6= Anderson |author-link6= Harry Anderson (chemist) |title=An sp-hybridized molecular carbon allotrope, cyclo[18]carbon |journal=Science |volume=365 |issue=6459 |pages=1299–1301 |date=15 Aug 2019 |doi=10.1126/science.aay1914 |arxiv=1908.05904 }}</ref> by [[electrochemical]] [[decarbonylation]] of several sites of a [[cyclobutanone]] structure:<ref name=Castelvecchi/> Later, researchers from Spain have used computational techniques to probe the structural and electronic properties of the molecule, and have discovered it to be an electron acceptor.<ref>{{cite journal |last1=Stasyuk |first1=Anton J. |last2=Stasyuk |first2=Olga A. |last3=Solà |first3=Miquel | last4= Voityuk |first4= Alexander A.|title=Cyclo[18]carbon: the smallest all-carbon electron acceptor |journal=Chemical Communications |volume=56 |pages=352–355 |date=29 Nov 2019 |doi=10.1039/c9cc08399e }}</ref> |
A collaboration of teams at [[IBM]] and the [[University of Oxford]] team claimed to synthesize it in solid state in 2019<ref>{{cite journal |last1=Kaiser |first1=Katharina |first2= Lorel M. |last2= Scriven |first3= Fabian |last3= Schulz |first4= Przemyslaw |last4= Gawel |first5= Leo |last5= Gross |first6= Harry L. |last6= Anderson |author-link6= Harry Anderson (chemist) |title=An sp-hybridized molecular carbon allotrope, cyclo[18]carbon |journal=Science |volume=365 |issue=6459 |pages=1299–1301 |date=15 Aug 2019 |doi=10.1126/science.aay1914 |pmid=31416933 |arxiv=1908.05904 |s2cid=201019470 }}</ref> by [[electrochemical]] [[decarbonylation]] of several sites of a [[cyclobutanone]] structure:<ref name=Castelvecchi/> Later, researchers from Spain have used computational techniques to probe the structural and electronic properties of the molecule, and have discovered it to be an electron acceptor.<ref>{{cite journal |last1=Stasyuk |first1=Anton J. |last2=Stasyuk |first2=Olga A. |last3=Solà |first3=Miquel | last4= Voityuk |first4= Alexander A.|title=Cyclo[18]carbon: the smallest all-carbon electron acceptor |journal=Chemical Communications |volume=56 |pages=352–355 |date=29 Nov 2019 |issue=3 |doi=10.1039/c9cc08399e |pmid=31825030 |hdl=10256/17613 |s2cid=209316603 |hdl-access=free }}</ref> |
||
[[File:Synthese C18.svg|600px|center|Synthesis of cyclocarbon]] |
[[File:Synthese C18.svg|600px|center|Synthesis of cyclocarbon]] |
||
| Line 60: | Line 63: | ||
[[Category:Polyynes]] |
[[Category:Polyynes]] |
||
[[Category:Cyclocarbons]] |
[[Category:Cyclocarbons]] |
||
[[Category:Substances discovered in the 2010s]] |
|||
[[Category:Cycloalkynes]] |
|||
| |
| Names | |
|---|---|
| Preferred IUPAC name
Cyclooctadeca-1,3,5,7,9,11,13,15,17-nonayne | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C18 | |
| Molar mass | 216.198 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclooctadeca-1,3,5,7,9,11,13,15,17-nonayneorcyclo[18]carbon is an allotrope of carbon with molecular formula C
18. The molecule is a ring of eighteen carbon atoms, connected by alternating triple and single bonds; thus, it is a polyyne and a cyclocarbon.
Cyclo[18]carbon is the smallest cyclo[n]carbon predicted to be thermodynamically stable, with a computed strain energy of 72 kilocalories per mole.[1] Above 122 K, it explosively decomposes to amorphous graphite.[2]
A collaboration of teams at IBM and the University of Oxford team claimed to synthesize it in solid state in 2019[3]byelectrochemical decarbonylation of several sites of a cyclobutanone structure:[4] Later, researchers from Spain have used computational techniques to probe the structural and electronic properties of the molecule, and have discovered it to be an electron acceptor.[5]

According to these IBM researchers, the electronic structure of their product consists of alternating triple bonds and single bonds, rather than a cumulene-type structure of consecutive double bonds. This supposedly makes this molecule a semiconductor.[4]