

|
→Further reading: fixing
|
m Open access bot: doi updated in citation with #oabot.
|
||
| (12 intermediate revisions by 11 users not shown) | |||
| Line 14: | Line 14: | ||
The virus can be transmitted by exposure to one species of [[fruit bats]] or it can be transmitted between people via body fluids through unprotected sex and broken skin. The disease can cause [[haemorrhage]], fever, and other symptoms similar to [[Ebola]], which belongs to the same family of viruses. According to the WHO, there are no approved vaccines or antiviral treatment for Marburg, but early, professional treatment of symptoms like dehydration considerably increases survival chances.<ref>Marburg virus disease Fact sheet Updated October 2017 http://www.who.int/mediacentre/factsheets/fs_marburg/en/</ref> |
The virus can be transmitted by exposure to one species of [[fruit bats]] or it can be transmitted between people via body fluids through unprotected sex and broken skin. The disease can cause [[haemorrhage]], fever, and other symptoms similar to [[Ebola]], which belongs to the same family of viruses. According to the WHO, there are no approved vaccines or antiviral treatment for Marburg, but early, professional treatment of symptoms like dehydration considerably increases survival chances.<ref>Marburg virus disease Fact sheet Updated October 2017 http://www.who.int/mediacentre/factsheets/fs_marburg/en/</ref> |
||
In 2009, expanded [[clinical trial]]s of an |
In 2009, expanded [[clinical trial]]s of an Ebola and Marburg [[vaccine]] began in [[Kampala]], Uganda.<ref>Beth Skwarecki [http://www.medscape.com/viewarticle/831858 Ebola, Marburg DNA Vaccines Prove Safe in Phase 1 Trial] Medscape Medical News, September 17, 2014</ref><ref>[http://clinicaltrials.gov/show/NCT00997607 Evaluating an Ebola and a Marburg Vaccine in Uganda] [[U.S. Department of Health & Human Services]]</ref> |
||
==History== |
==History== |
||
| Line 20: | Line 20: | ||
===Discovery=== |
===Discovery=== |
||
{{See also|1967 Marburg virus outbreak in West Germany|l1=1967 Marburg virus outbreak}} |
{{See also|1967 Marburg virus outbreak in West Germany|l1=1967 Marburg virus outbreak}} |
||
[[File:Marburg em1986.png|thumb|upright=.7|CryoEM reconstruction of a section of the Marburg virus [[nucleocapsid]]. [[EMDB]] entry |
[[File:Marburg em1986.png|thumb|upright=.7|CryoEM reconstruction of a section of the Marburg virus [[nucleocapsid]]. [[EMDB]] entry.<ref>{{cite web |title=CryoEM reconstruction of the Marburg virus nucleocapsid |url=https://www.ebi.ac.uk/emdb/EMD-1986 |website=Electron Microscopy Data Bank |access-date=18 February 2023}}</ref><ref>{{cite journal | vauthors = Bharat TA, Riches JD, Kolesnikova L, Welsch S, Krähling V, Davey N, Parsy ML, Becker S, Briggs JA | display-authors = 6 | title = Cryo-electron tomography of Marburg virus particles and their morphogenesis within infected cells | journal = PLOS Biology | volume = 9 | issue = 11 | pages = e1001196 | date = November 2011 | pmid = 22110401 | pmc = 3217011 | doi = 10.1371/journal.pbio.1001196 | veditors = Rey FA | doi-access = free }}</ref>]]Marburg virus was first described in 1967.<ref name=Siegert1967>{{cite journal | vauthors = Siegert R, Shu HL, Slenczka W, Peters D, Müller G | title = [On the etiology of an unknown human infection originating from monkeys] | journal = Deutsche Medizinische Wochenschrift | volume = 92 | issue = 51 | pages = 2341–2343 | date = December 1967 | pmid = 4294540 | doi = 10.1055/s-0028-1106144 | s2cid = 116556454 }}</ref> It was discovered that year during a set of outbreaks of Marburg virus disease in the German cities of [[Marburg]] and [[Frankfurt]] and the Yugoslav capital [[Belgrade]]. Laboratory workers were exposed to tissues of infected [[grivet|grivet monkey]]s (the African green monkey, ''Chlorocebus aethiops'') at the [[:de:Behringwerke|Behringwerke]], a major industrial plant in Marburg which was then part of [[Hoechst AG|Hoechst]], and later part of [[CSL Behring]]. During the outbreaks, thirty-one people became infected and seven of them died.<ref>{{cite journal | vauthors = Slenczka W, Klenk HD | title = Forty years of marburg virus | journal = The Journal of Infectious Diseases | volume = 196 | issue = Suppl 2 | pages = S131–S135 | date = November 2007 | pmid = 17940940 | doi = 10.1086/520551 | doi-access = free }}</ref> |
||
===Nomenclature=== |
===Nomenclature=== |
||
The virus is one of two members of the species ''[[Marburg marburgvirus]]'', which is included in the genus ''[[Marburgvirus]]'', family ''[[Filoviridae]]'', and order ''[[Mononegavirales]]''. The name Marburg virus is derived from [[Marburg]] (the city in [[Hesse]], Germany, where the virus was first discovered) and the [[Taxonomy (biology)|taxonomic]] suffix ''virus''.<ref name=KuhnArch>{{cite journal | vauthors = Kuhn JH, Becker S, Ebihara H, Geisbert TW, Johnson KM, Kawaoka Y, Lipkin WI, Negredo AI, Netesov SV, Nichol ST, Palacios G, Peters CJ, Tenorio A, Volchkov VE, Jahrling PB | display-authors = 6 | title = Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations | journal = Archives of Virology | volume = 155 | issue = 12 | pages = 2083–2103 | date = December 2010 | pmid = 21046175 | pmc = 3074192 | doi = 10.1007/s00705-010-0814-x }}</ref> |
The virus is one of two members of the species ''[[Marburg marburgvirus]]'', which is included in the genus ''[[Marburgvirus]]'', family ''[[Filoviridae]]'', and order ''[[Mononegavirales]]''. The name Marburg virus is derived from [[Marburg]] (the city in [[Hesse]], Germany, where the virus was first discovered) and the [[Taxonomy (biology)|taxonomic]] suffix ''virus''.<ref name=KuhnArch>{{cite journal | vauthors = Kuhn JH, Becker S, Ebihara H, Geisbert TW, Johnson KM, Kawaoka Y, Lipkin WI, Negredo AI, Netesov SV, Nichol ST, Palacios G, Peters CJ, Tenorio A, Volchkov VE, Jahrling PB | display-authors = 6 | title = Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations | journal = Archives of Virology | volume = 155 | issue = 12 | pages = 2083–2103 | date = December 2010 | pmid = 21046175 | pmc = 3074192 | doi = 10.1007/s00705-010-0814-x }}</ref> |
||
Marburg virus was first introduced under this name in 1967.<ref name=Siegert1967/> The virus name was changed to Lake Victoria marburgvirus in 2005, confusingly making the only difference in distinguishing between a Marburg virus organism and its species as a whole italicization, as in ''Lake Victoria marburgvirus''.<ref name=Feldmann2005>{{Cite book| vauthors = Feldmann H, Geisbert TW, Jahrling PB, Klenk H, Netesov SV, Peters CJ, Sanchez A, Swanepoel R, Volchkov VE | display-authors = 6 |chapter=Family Filoviridae|year=2005| veditors = Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA |title=Virus Taxonomy—Eighth Report of the International Committee on Taxonomy of Viruses|pages=645–653|publisher=Elsevier/Academic Press|location=San Diego, US|isbn=978-0-12-370200-5}}</ref><ref>{{cite journal| vauthors = Mayo MA |year = 2002|title = ICTV at the Paris ICV: results of the plenary session and the binomial ballot|journal = Archives of Virology|volume = 147|issue = 11|pages = 2254–60| doi=10.1007/s007050200052 |s2cid=43887711}}</ref><ref name="KuhnJahrling2010">{{cite journal | vauthors = Kuhn JH, Jahrling PB | title = Clarification and guidance on the proper usage of virus and virus species names | journal = Archives of Virology | volume = 155 | issue = 4 | pages = 445–453 | date = April 2010 | pmid = 20204430 | pmc = 2878132 | doi = 10.1007/s00705-010-0600-9 }}</ref> Still, most scientific articles continued to use the name Marburg virus. Consequently, in 2010, the name Marburg virus was reinstated and the species name changed.<ref name=KuhnArch/> |
Marburg virus was first introduced under this name in 1967.<ref name=Siegert1967/> The virus name was changed to Lake Victoria marburgvirus in 2005, confusingly making the only difference in distinguishing between a Marburg virus organism and its species as a whole italicization, as in ''Lake Victoria marburgvirus''.<ref name=Feldmann2005>{{Cite book| vauthors = Feldmann H, Geisbert TW, Jahrling PB, Klenk H, Netesov SV, Peters CJ, Sanchez A, Swanepoel R, Volchkov VE | display-authors = 6 |chapter=Family Filoviridae|year=2005| veditors = Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA |title=Virus Taxonomy—Eighth Report of the International Committee on Taxonomy of Viruses|pages=645–653|publisher=Elsevier/Academic Press|location=San Diego, US|isbn=978-0-12-370200-5}}</ref><ref>{{cite journal| vauthors = Mayo MA |year = 2002|title = ICTV at the Paris ICV: results of the plenary session and the binomial ballot|journal = Archives of Virology|volume = 147|issue = 11|pages = 2254–60| doi=10.1007/s007050200052 |s2cid=43887711|doi-access = free}}</ref><ref name="KuhnJahrling2010">{{cite journal | vauthors = Kuhn JH, Jahrling PB | title = Clarification and guidance on the proper usage of virus and virus species names | journal = Archives of Virology | volume = 155 | issue = 4 | pages = 445–453 | date = April 2010 | pmid = 20204430 | pmc = 2878132 | doi = 10.1007/s00705-010-0600-9 }}</ref> Still, most scientific articles continued to use the name Marburg virus. Consequently, in 2010, the name Marburg virus was reinstated and the species name changed.<ref name=KuhnArch/> |
||
==Virology== |
==Virology== |
||
===Genome=== |
===Genome=== |
||
[[File:Viruses-04-01878-g005.webp|thumb|Marburg virion and genome]] |
[[File:Viruses-04-01878-g005.webp|thumb|Marburg virion and genome]] |
||
Like all [[Mononegavirales|mononegaviruses]], |
Like all [[Mononegavirales|mononegaviruses]], marburg virions contain non-infectious, linear nonsegmented, single-stranded [[RNA]] [[genome]]s of negative polarity that possess inverse-complementary 3' and 5' termini, do not possess a [[5' cap]], are not [[Polyadenylation|polyadenylated]], and are not [[Covalent bond|covalently]] linked to a [[protein]].<ref name=Fauquet2005>{{Cite book | vauthors = Pringle CR |chapter=Order Mononegavirales|year=2005| veditors = Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA |title=Virus Taxonomy—Eighth Report of the International Committee on Taxonomy of Viruses|pages=609–614|publisher=Elsevier/Academic Press|location=San Diego, US|isbn=978-0-12-370200-5}}</ref> Marburgvirus genomes are approximately 19 [[base pair|kbp]] long and contain seven [[gene]]s in the order [[Three prime untranslated region|3'-UTR]]-''NP''-''VP35''-''VP40''-''GP''-''VP30''-''VP24''-''L''-[[Five prime untranslated region|5'-UTR]].<ref name = Kiley1982>{{cite journal | vauthors = Kiley MP, Bowen ET, Eddy GA, Isaäcson M, Johnson KM, McCormick JB, Murphy FA, Pattyn SR, Peters D, Prozesky OW, Regnery RL, Simpson DI, Slenczka W, Sureau P, van der Groen G, Webb PA, Wulff H | display-authors = 6 | title = Filoviridae: a taxonomic home for Marburg and Ebola viruses? | journal = Intervirology | volume = 18 | issue = 1–2 | pages = 24–32 | year = 1982 | pmid = 7118520 | doi = 10.1159/000149300 | doi-access = free }}</ref> |
||
=== Structure === |
=== Structure === |
||
[[File:137488 web.jpg|thumb|Micrograph of the Marburg viruses]] |
[[File:137488 web.jpg|thumb|Micrograph of the Marburg viruses]] |
||
[[File:Marburg Virus Particle (30971357537).jpg|thumb|Colorized electron micrograph of a Marburg virus|upright]] |
[[File:Marburg Virus Particle (30971357537).jpg|thumb|Colorized electron micrograph of a Marburg virus|upright]] |
||
Like all [[Filoviridae|filoviruses]], marburgvirions are filamentous particles that may appear in the shape of a shepherd's crook or in the shape of a "U" or a "6", and they may be coiled, toroid, or branched.<ref name = Kiley1982/> Marburgvirions are generally 80 nm in [[width]], but vary somewhat in length. In general, the median particle length of marburgviruses ranges from 795 to 828 nm (in contrast to [[Ebolavirus|ebolavirions]], whose median particle length was measured to be 974–1,086 nm |
Like all [[Filoviridae|filoviruses]], marburgvirions are filamentous particles that may appear in the shape of a shepherd's crook or in the shape of a "U" or a "6", and they may be coiled, toroid, or branched.<ref name = Kiley1982/> Marburgvirions are generally 80 nm in [[width]], but vary somewhat in length. In general, the median particle length of marburgviruses ranges from 795 to 828 nm (in contrast to [[Ebolavirus|ebolavirions]], whose median particle length was measured to be 974–1,086 nm), but particles as long as 14,000 nm have been detected in tissue culture.<ref name = Geisbert1995>{{cite journal | vauthors = Geisbert TW, Jahrling PB | title = Differentiation of filoviruses by electron microscopy | journal = Virus Research | volume = 39 | issue = 2–3 | pages = 129–150 | date = December 1995 | pmid = 8837880 | doi = 10.1016/0168-1702(95)00080-1 | url = https://zenodo.org/record/1258399 }}</ref> |
||
Marburgvirions consist of seven structural proteins. At the center is the [[Helix|helical]] [[ribonucleoprotein|ribonucleocapsid]], which consists of the genomic RNA wrapped around a [[polymer]] of [[nucleoprotein]]s (NP). Associated with the ribonucleoprotein is the [[RNA-dependent RNA polymerase]] (L) with the polymerase cofactor (VP35) and a transcription activator (VP30). The ribonucleoprotein is embedded in a matrix, formed by the major (VP40) and minor (VP24) matrix proteins. These particles are surrounded by a [[lipid bilayer|lipid membrane]] derived from the host cell membrane. The membrane anchors a glycoprotein (GP<sub>1,2</sub>) that projects 7 to 10 nm spikes away from its surface. While nearly identical to ebolavirions in structure, marburgvirions are [[antigen]]ically distinct.<ref>{{cite journal | vauthors = King LB, West BR, Schendel SL, Saphire EO | title = The structural basis for filovirus neutralization by monoclonal antibodies | journal = Current Opinion in Immunology | volume = 53 | pages = 196–202 | date = August 2018 | pmid = 29940415 | pmc = 6141344 | doi = 10.1016/j.coi.2018.05.001 }}</ref> |
Marburgvirions consist of seven structural proteins. At the center is the [[Helix|helical]] [[ribonucleoprotein|ribonucleocapsid]], which consists of the genomic RNA wrapped around a [[polymer]] of [[nucleoprotein]]s (NP). Associated with the ribonucleoprotein is the [[RNA-dependent RNA polymerase]] (L) with the polymerase cofactor (VP35) and a transcription activator (VP30). The ribonucleoprotein is embedded in a matrix, formed by the major (VP40) and minor (VP24) matrix proteins. These particles are surrounded by a [[lipid bilayer|lipid membrane]] derived from the host cell membrane. The membrane anchors a glycoprotein (GP<sub>1,2</sub>) that projects 7 to 10 nm spikes away from its surface. While nearly identical to ebolavirions in structure, marburgvirions are [[antigen]]ically distinct.<ref>{{cite journal | vauthors = King LB, West BR, Schendel SL, Saphire EO | title = The structural basis for filovirus neutralization by monoclonal antibodies | journal = Current Opinion in Immunology | volume = 53 | pages = 196–202 | date = August 2018 | pmid = 29940415 | pmc = 6141344 | doi = 10.1016/j.coi.2018.05.001 }}</ref> |
||
| Line 45: | Line 45: | ||
* {{cite news | vauthors = Schaffer A |date=January 16, 2012 |title=Key Protein May Give Ebola Virus Its Opening |newspaper=The New York Times |url=https://www.nytimes.com/2012/01/17/health/npc1-protein-may-give-ebola-its-opening.html |url-access=subscription}}</ref> When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the [[virus]], further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Marburg virus, showing that it also needs NPC1 to enter cells.<ref name="pmid21866103"/><ref name="pmid21866101"/> Furthermore, NPC1 was shown to be critical to [[filovirus]] entry because it mediates infection by binding directly to the [[viral envelope]] glycoprotein<ref name="pmid21866101"/> and that the second lysosomal domain of NPC1 mediates this binding.<ref name="pmid22395071">{{cite journal | vauthors = Miller EH, Obernosterer G, Raaben M, Herbert AS, Deffieu MS, Krishnan A, Ndungo E, Sandesara RG, Carette JE, Kuehne AI, Ruthel G, Pfeffer SR, Dye JM, Whelan SP, Brummelkamp TR, Chandran K | display-authors = 6 | title = Ebola virus entry requires the host-programmed recognition of an intracellular receptor | journal = The EMBO Journal | volume = 31 | issue = 8 | pages = 1947–1960 | date = April 2012 | pmid = 22395071 | pmc = 3343336 | doi = 10.1038/emboj.2012.53 }}</ref> |
* {{cite news | vauthors = Schaffer A |date=January 16, 2012 |title=Key Protein May Give Ebola Virus Its Opening |newspaper=The New York Times |url=https://www.nytimes.com/2012/01/17/health/npc1-protein-may-give-ebola-its-opening.html |url-access=subscription}}</ref> When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the [[virus]], further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Marburg virus, showing that it also needs NPC1 to enter cells.<ref name="pmid21866103"/><ref name="pmid21866101"/> Furthermore, NPC1 was shown to be critical to [[filovirus]] entry because it mediates infection by binding directly to the [[viral envelope]] glycoprotein<ref name="pmid21866101"/> and that the second lysosomal domain of NPC1 mediates this binding.<ref name="pmid22395071">{{cite journal | vauthors = Miller EH, Obernosterer G, Raaben M, Herbert AS, Deffieu MS, Krishnan A, Ndungo E, Sandesara RG, Carette JE, Kuehne AI, Ruthel G, Pfeffer SR, Dye JM, Whelan SP, Brummelkamp TR, Chandran K | display-authors = 6 | title = Ebola virus entry requires the host-programmed recognition of an intracellular receptor | journal = The EMBO Journal | volume = 31 | issue = 8 | pages = 1947–1960 | date = April 2012 | pmid = 22395071 | pmc = 3343336 | doi = 10.1038/emboj.2012.53 }}</ref> |
||
In one of the original studies, a [[small molecule]] was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.<ref name="pmid21866101"/><ref name="pmid21959282">{{cite journal | vauthors = Flemming A | title = Achilles heel of Ebola viral entry | journal = Nature Reviews. Drug Discovery | volume = 10 | issue = 10 | pages = 731 | date = September 2011 | pmid = 21959282 | doi = 10.1038/nrd3568 | s2cid = 26888076 }}</ref> In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse-adapted Ebola virus.<ref name="pmid21866103"/> |
In one of the original studies, a [[small molecule]] was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.<ref name="pmid21866101"/><ref name="pmid21959282">{{cite journal | vauthors = Flemming A | title = Achilles heel of Ebola viral entry | journal = Nature Reviews. Drug Discovery | volume = 10 | issue = 10 | pages = 731 | date = September 2011 | pmid = 21959282 | doi = 10.1038/nrd3568 | s2cid = 26888076 | doi-access = free }}</ref> In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse-adapted Ebola virus.<ref name="pmid21866103"/> |
||
===Replication=== |
===Replication=== |
||
| Line 56: | Line 56: | ||
==Ecology== |
==Ecology== |
||
[[File:Viruses-04-01878-g001-A.jpg|thumb|The geographic distribution of Marburg virus and [[Egyptian fruit bat]]s |
[[File:Viruses-04-01878-g001-A.jpg|thumb|The geographic distribution of Marburg virus and [[Egyptian fruit bat]]s]] |
||
In 2009, the successful isolation of infectious MARV was reported from caught healthy [[Rousettus aegyptiacus|Egyptian fruit bats (''Rousettus aegyptiacus'')]].<ref name="Towner2009"/> This isolation, together with the isolation of infectious [[RAVV]],<ref name=Towner2009/> strongly suggests that [[Old World]] fruit [[bat]]s are involved in the natural maintenance of marburgviruses. Further studies are necessary to establish whether [[Egyptian fruit bat|Egyptian rousettes]] are the actual hosts of MARV and RAVV or whether they get infected via contact with another animal and therefore serve only as intermediate hosts. In 2012 the first experimental infection study of ''Rousettus aegyptiacus'' with MARV provided further insight into the possible involvement of these bats in MARV ecology.<ref>{{cite journal | vauthors = Paweska JT, Jansen van Vuren P, Masumu J, Leman PA, Grobbelaar AA, Birkhead M, Clift S, Swanepoel R, Kemp A | display-authors = 6 | title = Virological and serological findings in Rousettus aegyptiacus experimentally inoculated with vero cells-adapted hogan strain of Marburg virus | journal = PLOS ONE | volume = 7 | issue = 9 | pages = e45479 | year = 2012 | pmid = 23029039 | pmc = 3444458 | doi = 10.1371/journal.pone.0045479 | doi-access = free | bibcode = 2012PLoSO...745479P }} {{open access}}</ref> |
In 2009, the successful isolation of infectious MARV was reported from caught healthy [[Rousettus aegyptiacus|Egyptian fruit bats (''Rousettus aegyptiacus'')]].<ref name="Towner2009"/> This isolation, together with the isolation of infectious [[RAVV]],<ref name=Towner2009/> strongly suggests that [[Old World]] fruit [[bat]]s are involved in the natural maintenance of marburgviruses. Further studies are necessary to establish whether [[Egyptian fruit bat|Egyptian rousettes]] are the actual hosts of MARV and RAVV or whether they get infected via contact with another animal and therefore serve only as intermediate hosts. In 2012 the first experimental infection study of ''Rousettus aegyptiacus'' with MARV provided further insight into the possible involvement of these bats in MARV ecology.<ref name=":1">{{cite journal | vauthors = Paweska JT, Jansen van Vuren P, Masumu J, Leman PA, Grobbelaar AA, Birkhead M, Clift S, Swanepoel R, Kemp A | display-authors = 6 | title = Virological and serological findings in Rousettus aegyptiacus experimentally inoculated with vero cells-adapted hogan strain of Marburg virus | journal = PLOS ONE | volume = 7 | issue = 9 | pages = e45479 | year = 2012 | pmid = 23029039 | pmc = 3444458 | doi = 10.1371/journal.pone.0045479 | doi-access = free | bibcode = 2012PLoSO...745479P }} {{open access}}</ref> |
||
Experimentally infected bats developed relatively low viremia lasting at least |
Experimentally infected bats developed relatively low viremia lasting at least five days, but remained healthy and did not develop any notable gross pathology. The virus also replicated to high titers in major organs (liver and spleen), and organs that might possibly be involved in virus transmission (lung, intestine, reproductive organs, salivary gland, kidney, bladder, and mammary gland). The relatively long period of viremia noted in this experiment could possibly also facilitate mechanical transmission by blood sucking arthropods in addition to infection of susceptible vertebrate hosts by direct contact with infected blood.<ref name=":1" /> |
||
==Evolution== |
==Evolution== |
||
| Line 87: | Line 87: | ||
| 7 |
| 7 |
||
| 23% |
| 23% |
||
| |
| Laboratory leak<ref>{{cite journal | vauthors = Ristanović ES, Kokoškov NS, Crozier I, Kuhn JH, Gligić AS | title = A Forgotten Episode of Marburg Virus Disease: Belgrade, Yugoslavia, 1967 | journal = Microbiology and Molecular Biology Reviews | volume = 84 | issue = 2 | pages = e00095-19 | date = May 2020 | pmid = 32404328 | pmc = 7233485 | doi = 10.1128/MMBR.00095-19 }}</ref><ref name=Siegert1967/><ref name=Smith1967>{{cite journal | vauthors = Smith CE, Simpson DI, Bowen ET, Zlotnik I | title = Fatal human disease from vervet monkeys | journal = Lancet | volume = 2 | issue = 7526 | pages = 1119–1121 | date = November 1967 | pmid = 4168558 | doi = 10.1016/s0140-6736(67)90621-6 }}</ref><ref name=Kissling1968>{{cite journal | vauthors = Kissling RE, Robinson RQ, Murphy FA, Whitfield SG | title = Agent of disease contracted from green monkeys | journal = Science | volume = 160 | issue = 3830 | pages = 888–890 | date = May 1968 | pmid = 4296724 | doi = 10.1126/science.160.3830.888 | s2cid = 30252321 | bibcode = 1968Sci...160..888K }}</ref><ref name=Martini1968>{{cite journal | vauthors = Martini GA, Knauff HG, Schmidt HA, Mayer G, Baltzer G | title = [On the hitherto unknown, in monkeys originating infectious disease: Marburg virus disease] | journal = Deutsche Medizinische Wochenschrift | volume = 93 | issue = 12 | pages = 559–571 | date = March 1968 | pmid = 4966280 | doi = 10.1055/s-0028-1105098 | s2cid = 260056835 }}</ref><ref name=Stille1968>{{cite journal | vauthors = Stille W, Böhle E, Helm E, van Rey W, Siede W | title = [On an infectious disease transmitted by Cercopithecus aethiops. ("Green monkey disease")] | journal = Deutsche Medizinische Wochenschrift | volume = 93 | issue = 12 | pages = 572–582 | date = March 1968 | pmid = 4966281 | doi = 10.1055/s-0028-1105099 | s2cid = 260058558 }}</ref><ref name=Bonin1969>{{cite journal | vauthors = Bonin O | title = The Cercopithecus monkey disease in Marburg and Frankfurt (Main), 1967 | journal = Acta Zoologica et Pathologica Antverpiensia | volume = 48 | pages = 319–331 | date = May 1969 | pmid = 5005859 }}</ref><ref name=Jacob1971>{{cite journal | vauthors = Jacob H, Solcher H | title = [An infectious disease transmitted by Cercopithecus aethiops ("marbury disease") with glial nodule encephalitis] | journal = Acta Neuropathologica | volume = 11 | issue = 1 | pages = 29–44 | date = July 1968 | pmid = 5748997 | doi = 10.1007/bf00692793 | s2cid = 12791113 }}</ref><ref name= Stojkovic1971>{{Cite book| vauthors = Stojkovic L, Bordjoski M, Gligic A, Stefanovic Z |year = 1971|chapter = Two Cases of Cercopithecus-Monkeys-Associated Haemorrhagic Fever | veditors = Martini GA, Siegert R |title=Marburg Virus Disease|pages=24–33|publisher=Springer-Verlag|location=Berlin, Germany|isbn=978-0-387-05199-4|chapter-url = https://books.google.com/books?id=SoFrAAAAMAAJ}}</ref> |
||
|- valign="TOP" |
|- valign="TOP" |
||
| 1975 |
| 1975 |
||
| Line 120: | Line 120: | ||
| 1 |
| 1 |
||
| 100% |
| 100% |
||
| |
| Laboratory accident<ref name=Beer1999>{{cite journal | vauthors = Beer B, Kurth R, Bukreyev A | title = Characteristics of Filoviridae: Marburg and Ebola viruses | journal = Die Naturwissenschaften | volume = 86 | issue = 1 | pages = 8–17 | date = January 1999 | pmid = 10024977 | doi = 10.1007/s001140050562 | s2cid = 25789824 | bibcode = 1999NW.....86....8B | doi-access = free }}</ref> |
||
|- valign="TOP" |
|- valign="TOP" |
||
| 1990 |
| 1990 |
||
| Line 128: | Line 128: | ||
| 1 |
| 1 |
||
| 100% |
| 100% |
||
| |
| Laboratory accident<ref name=Nikiforov1994>{{cite journal | vauthors = Nikiforov VV, Turovskiĭ I, Kalinin PP, Akinfeeva LA, Katkova LR, Barmin VS, Riabchikova EI, Popkova NI, Shestopalov AM, Nazarov VP | display-authors = 6 | title = [A case of a laboratory infection with Marburg fever] | journal = Zhurnal Mikrobiologii, Epidemiologii I Immunobiologii | issue = 3 | pages = 104–106 | year = 1994 | pmid = 7941853 }}</ref> |
||
|- valign="TOP" |
|- valign="TOP" |
||
| 1998–2000 |
| 1998–2000 |
||
| Line 152: | Line 152: | ||
| 1 |
| 1 |
||
| 25% |
| 25% |
||
| <ref name=Towner2009>{{cite journal | vauthors = Towner JS, Amman BR, Sealy TK, Carroll SA, Comer JA, Kemp A, Swanepoel R, Paddock CD, Balinandi S, Khristova ML, Formenty PB, Albarino CG, Miller DM, Reed ZD, Kayiwa JT, Mills JN, Cannon DL, Greer PW, Byaruhanga E, Farnon EC, Atimnedi P, Okware S, Katongole-Mbidde E, Downing R, Tappero JW, Zaki SR, Ksiazek TG, Nichol ST, Rollin PE | display-authors = 6 | title = Isolation of genetically diverse Marburg viruses from Egyptian fruit bats | journal = PLOS Pathogens | volume = 5 | issue = 7 | pages = e1000536 | date = July 2009 | pmid = 19649327 | pmc = 2713404 | doi = 10.1371/journal.ppat.1000536 | veditors = Fouchier RA }}</ref><ref name=Adjemian2011>{{cite journal | vauthors = Adjemian J, Farnon EC, Tschioko F, Wamala JF, Byaruhanga E, Bwire GS, Kansiime E, Kagirita A, Ahimbisibwe S, Katunguka F, Jeffs B, Lutwama JJ, Downing R, Tappero JW, Formenty P, Amman B, Manning C, Towner J, Nichol ST, Rollin PE | display-authors = 6 | title = Outbreak of Marburg hemorrhagic fever among miners in Kamwenge and Ibanda Districts, Uganda, 2007 | journal = The Journal of Infectious Diseases | volume = 204 | issue = Suppl 3 | pages = S796–S799 | date = November 2011 | pmid = 21987753 | pmc = 3203392 | doi = 10.1093/infdis/jir312 }}</ref> |
| <ref name=Towner2009>{{cite journal | vauthors = Towner JS, Amman BR, Sealy TK, Carroll SA, Comer JA, Kemp A, Swanepoel R, Paddock CD, Balinandi S, Khristova ML, Formenty PB, Albarino CG, Miller DM, Reed ZD, Kayiwa JT, Mills JN, Cannon DL, Greer PW, Byaruhanga E, Farnon EC, Atimnedi P, Okware S, Katongole-Mbidde E, Downing R, Tappero JW, Zaki SR, Ksiazek TG, Nichol ST, Rollin PE | display-authors = 6 | title = Isolation of genetically diverse Marburg viruses from Egyptian fruit bats | journal = PLOS Pathogens | volume = 5 | issue = 7 | pages = e1000536 | date = July 2009 | pmid = 19649327 | pmc = 2713404 | doi = 10.1371/journal.ppat.1000536 | veditors = Fouchier RA | doi-access = free }}</ref><ref name=Adjemian2011>{{cite journal | vauthors = Adjemian J, Farnon EC, Tschioko F, Wamala JF, Byaruhanga E, Bwire GS, Kansiime E, Kagirita A, Ahimbisibwe S, Katunguka F, Jeffs B, Lutwama JJ, Downing R, Tappero JW, Formenty P, Amman B, Manning C, Towner J, Nichol ST, Rollin PE | display-authors = 6 | title = Outbreak of Marburg hemorrhagic fever among miners in Kamwenge and Ibanda Districts, Uganda, 2007 | journal = The Journal of Infectious Diseases | volume = 204 | issue = Suppl 3 | pages = S796–S799 | date = November 2011 | pmid = 21987753 | pmc = 3203392 | doi = 10.1093/infdis/jir312 }}</ref> |
||
|- valign="TOP" |
|- valign="TOP" |
||
| 2008 |
| 2008 |
||
| Line 209: | Line 209: | ||
|11 |
|11 |
||
|44% |
|44% |
||
|See [[2023 Marburg virus disease outbreak in Equatorial Guinea|Equatorial Guinea Marburg virus outbreak 2023]].<ref>{{cite web |title=Equatorial Guinea confirms first-ever Marburg virus disease outbreak |url=https://www.afro.who.int/countries/equatorial-guinea/news/equatorial-guinea-confirms-first-ever-marburg-virus-disease-outbreak |website=[[World Health Organization]] |access-date=13 February 2023}}</ref><ref>{{cite web |title=Death Toll In E. Guinea Marburg Outbreak Rises To 11 |url=https://www.barrons.com/news/death-toll-in-e-guinea-marburg-outbreak-rises-to-11-9a4651ed |website=[[Barron's (newspaper)|Barron's]] |access-date=28 February 2023}}</ref> |
|See [[2023 Marburg virus disease outbreak in Equatorial Guinea|Equatorial Guinea Marburg virus outbreak 2023]].<ref>{{cite web |title=Equatorial Guinea confirms first-ever Marburg virus disease outbreak |url=https://www.afro.who.int/countries/equatorial-guinea/news/equatorial-guinea-confirms-first-ever-marburg-virus-disease-outbreak |website=[[World Health Organization]] |date=13 February 2023 |access-date=13 February 2023}}</ref><ref>{{cite web |title=Death Toll In E. Guinea Marburg Outbreak Rises To 11 |url=https://www.barrons.com/news/death-toll-in-e-guinea-marburg-outbreak-rises-to-11-9a4651ed |website=[[Barron's (newspaper)|Barron's]] |access-date=28 February 2023}}</ref> |
||
|- |
|- |
||
|[[2023 Marburg virus disease outbreak in Tanzania|Mar 2023]] |
|||
|Mar 2023 |
|||
|[[Tanzania]] |
|[[Tanzania]] |
||
| |
| |
||
| Line 217: | Line 217: | ||
|5 |
|5 |
||
|63% |
|63% |
||
|{{cite web |title=Five dead as Tanzania detects first-ever Marburg virus outbreak|url=https://www.aljazeera.com/news/2023/3/22/tanzania-detects-its-first-ever-cases-of-the-highly-fatal-marburg-viral-disease |website=[[aljazeera]] |access-date=22 March 2023}} |
|{{cite web |title=Five dead as Tanzania detects first-ever Marburg virus outbreak|url=https://www.aljazeera.com/news/2023/3/22/tanzania-detects-its-first-ever-cases-of-the-highly-fatal-marburg-viral-disease |website=[[Al Jazeera English|aljazeera]] |access-date=22 March 2023}} |
||
|} |
|} |
||
==Prevention== |
==Prevention== |
||
The first clinical study testing the efficacy of a Marburg virus vaccine was conducted in 2014. The study tested a DNA vaccine and concluded that individuals inoculated with the vaccine exhibited some level of antibodies. However, these vaccines were not expected to provide definitive immunity.<ref name=":02">{{cite journal | vauthors = Kibuuka H, Berkowitz NM, Millard M, Enama ME, Tindikahwa A, Sekiziyivu AB, Costner P, Sitar S, Glover D, Hu Z, Joshi G, Stanley D, Kunchai M, Eller LA, Bailer RT, Koup RA, Nabel GJ, Mascola JR, Sullivan NJ, Graham BS, Roederer M, Michael NL, Robb ML, Ledgerwood JE | display-authors = 6 | title = Safety and immunogenicity of Ebola virus and Marburg virus glycoprotein DNA vaccines assessed separately and concomitantly in healthy Ugandan adults: a phase 1b, randomised, double-blind, placebo-controlled clinical trial | language = English | journal = Lancet | volume = 385 | issue = 9977 | pages = 1545–1554 | date = April 2015 | pmid = 25540891 | doi = 10.1016/S0140-6736(14)62385-0 | s2cid = 205975536 | doi-access = free }}</ref> Several animal models have shown to be effective in the research of Marburg virus, such as hamsters, mice, and non-human primates (NHPs). Mice are useful in the initial phases of vaccine development as they are ample models for mammalian disease, but their immune systems are still different enough from humans to warrant trials with other mammals.<ref>{{cite journal | vauthors = Shifflett K, Marzi A | title = Marburg virus pathogenesis - differences and similarities in humans and animal models | journal = Virology Journal | volume = 16 | issue = 1 | pages = 165 | date = December 2019 | pmid = 31888676 | pmc = 6937685 | doi = 10.1186/s12985-019-1272-z }}</ref> Of these models, the infection in macaques seems to be the most similar to the effects in humans.<ref>{{cite journal | vauthors = Ewers EC, Pratt WD, Twenhafel NA, Shamblin J, Donnelly G, Esham H, Wlazlowski C, Johnson JC, Botto M, Hensley LE, Goff AJ | display-authors = 6 | title = Natural History of Aerosol Exposure with Marburg Virus in Rhesus Macaques | journal = Viruses | volume = 8 | issue = 4 | pages = 87 | date = March 2016 | pmid = 27043611 | pmc = 4848582 | doi = 10.3390/v8040087 | doi-access = free }}</ref> A variety of other vaccines have been considered. Virus replicon particles (VRPs) were shown to be effective in guinea pigs, but lost efficacy once tested on NHPs. Additionally, an inactivated virus vaccine proved ineffective. DNA vaccines showed some efficacy in NHPs, but all inoculated individuals showed signs of infection.<ref>{{cite journal | vauthors = Suschak JJ, Schmaljohn CS | title = Vaccines against Ebola virus and Marburg virus: recent advances and promising candidates | journal = Human Vaccines & Immunotherapeutics | volume = 15 | issue = 10 | pages = 2359–2377 | date = 2019-10-03 | pmid = 31589088 | pmc = 6816442 | doi = 10.1080/21645515.2019.1651140 }}</ref> |
The first clinical study testing the efficacy of a Marburg virus vaccine was conducted in 2014. The study tested a DNA vaccine and concluded that individuals inoculated with the vaccine exhibited some level of antibodies. However, these vaccines were not expected to provide definitive immunity.<ref name=":02">{{cite journal | vauthors = Kibuuka H, Berkowitz NM, Millard M, Enama ME, Tindikahwa A, Sekiziyivu AB, Costner P, Sitar S, Glover D, Hu Z, Joshi G, Stanley D, Kunchai M, Eller LA, Bailer RT, Koup RA, Nabel GJ, Mascola JR, Sullivan NJ, Graham BS, Roederer M, Michael NL, Robb ML, Ledgerwood JE | display-authors = 6 | title = Safety and immunogenicity of Ebola virus and Marburg virus glycoprotein DNA vaccines assessed separately and concomitantly in healthy Ugandan adults: a phase 1b, randomised, double-blind, placebo-controlled clinical trial | language = English | journal = Lancet | volume = 385 | issue = 9977 | pages = 1545–1554 | date = April 2015 | pmid = 25540891 | doi = 10.1016/S0140-6736(14)62385-0 | s2cid = 205975536 | doi-access = free }}</ref> Several animal models have shown to be effective in the research of Marburg virus, such as hamsters, mice, and non-human primates (NHPs). Mice are useful in the initial phases of vaccine development as they are ample models for mammalian disease, but their immune systems are still different enough from humans to warrant trials with other mammals.<ref>{{cite journal | vauthors = Shifflett K, Marzi A | title = Marburg virus pathogenesis - differences and similarities in humans and animal models | journal = Virology Journal | volume = 16 | issue = 1 | pages = 165 | date = December 2019 | pmid = 31888676 | pmc = 6937685 | doi = 10.1186/s12985-019-1272-z | doi-access = free }}</ref> Of these models, the infection in macaques seems to be the most similar to the effects in humans.<ref>{{cite journal | vauthors = Ewers EC, Pratt WD, Twenhafel NA, Shamblin J, Donnelly G, Esham H, Wlazlowski C, Johnson JC, Botto M, Hensley LE, Goff AJ | display-authors = 6 | title = Natural History of Aerosol Exposure with Marburg Virus in Rhesus Macaques | journal = Viruses | volume = 8 | issue = 4 | pages = 87 | date = March 2016 | pmid = 27043611 | pmc = 4848582 | doi = 10.3390/v8040087 | doi-access = free }}</ref> A variety of other vaccines have been considered. Virus replicon particles (VRPs) were shown to be effective in guinea pigs, but lost efficacy once tested on NHPs. Additionally, an inactivated virus vaccine proved ineffective. DNA vaccines showed some efficacy in NHPs, but all inoculated individuals showed signs of infection.<ref>{{cite journal | vauthors = Suschak JJ, Schmaljohn CS | title = Vaccines against Ebola virus and Marburg virus: recent advances and promising candidates | journal = Human Vaccines & Immunotherapeutics | volume = 15 | issue = 10 | pages = 2359–2377 | date = 2019-10-03 | pmid = 31589088 | pmc = 6816442 | doi = 10.1080/21645515.2019.1651140 }}</ref> |
||
Because Marburg virus and Ebola virus belong to the same family, Filoviridae, some scientists have attempted to create a single-injection vaccine for both viruses. This would both make the vaccine more practical and lower the cost for developing countries.<ref>{{cite journal | vauthors = Geisbert TW, Geisbert JB, Leung A, Daddario-DiCaprio KM, Hensley LE, Grolla A, Feldmann H | title = Single-injection vaccine protects nonhuman primates against infection with marburg virus and three species of ebola virus | journal = Journal of Virology | volume = 83 | issue = 14 | pages = 7296–7304 | date = July 2009 | pmid = 19386702 | pmc = 2704787 | doi = 10.1128/JVI.00561-09 }}</ref> Using a single-injection vaccine has shown to not cause any adverse reactogenicity, which the possible immune response to vaccination, in comparison to two separate vaccinations.<ref name=":02" /> |
Because Marburg virus and Ebola virus belong to the same family, Filoviridae, some scientists have attempted to create a single-injection vaccine for both viruses. This would both make the vaccine more practical and lower the cost for developing countries.<ref>{{cite journal | vauthors = Geisbert TW, Geisbert JB, Leung A, Daddario-DiCaprio KM, Hensley LE, Grolla A, Feldmann H | title = Single-injection vaccine protects nonhuman primates against infection with marburg virus and three species of ebola virus | journal = Journal of Virology | volume = 83 | issue = 14 | pages = 7296–7304 | date = July 2009 | pmid = 19386702 | pmc = 2704787 | doi = 10.1128/JVI.00561-09 }}</ref> Using a single-injection vaccine has shown to not cause any adverse reactogenicity, which the possible immune response to vaccination, in comparison to two separate vaccinations.<ref name=":02" /> |
||
| Line 231: | Line 231: | ||
==Biological weapon== |
==Biological weapon== |
||
The [[Soviet Union]] had an extensive offensive and defensive [[Soviet biological weapons program|biological weapon]]s program that included MARV.<ref name=Alibek1999>{{Cite book| vauthors = Alibek K, Handelman S |title= Biohazard: The Chilling True Story of the Largest Covert Biological Weapons Program in the World—Told from Inside by the Man Who Ran It|publisher=Random House|location=New York |isbn=978-0-385-33496-9|url=https://books.google.com/books?id=eDTaCwAAQBAJ|year=1999}}</ref> At least three Soviet research institutes had MARV research programs during the [[Cold War]]: |
The [[Soviet Union]] had an extensive offensive and defensive [[Soviet biological weapons program|biological weapon]]s program that included MARV.<ref name=Alibek1999>{{Cite book| vauthors = Alibek K, Handelman S |title= Biohazard: The Chilling True Story of the Largest Covert Biological Weapons Program in the World—Told from Inside by the Man Who Ran It|publisher=Random House|location=New York |isbn=978-0-385-33496-9|url=https://books.google.com/books?id=eDTaCwAAQBAJ|year=1999}}</ref> At least three Soviet research institutes had MARV research programs during the [[Cold War]]: The Virology Center of the Scientific-Research Institute for Microbiology in Zagorsk (today [[Sergiev Posad]]), the Scientific-Production Association "Vektor" (today the [[State Research Center of Virology and Biotechnology VECTOR|State Research Center of Virology and Biotechnology "Vektor"]]) in [[Koltsovo, Novosibirsk Oblast|Koltsovo]], and the Irkutsk Scientific-Research Anti-Plague Institute of Siberia and the Far East in [[Irkutsk]].<ref name=Alibek1999/> |
||
As most performed research was highly [[classified information|classified]], it remains unclear how successful the MARV program was. However, Soviet [[defection|defector]] [[Ken Alibek]] claimed that a weapon filled with MARV was tested at the [[Stepnagorsk Scientific and Technical Institute for Microbiology|Stepnogorsk Scientific Experimental and Production Base]] in [[Stepnogorsk]], [[Kazakh Soviet Socialist Republic]] (today [[Kazakhstan]]),<ref name=Alibek1999/> suggesting that the development of a MARV biological weapon had reached advanced stages. Independent confirmation for this claim is lacking. At least one laboratory accident with MARV, resulting in the death of Koltsovo researcher Nikolai Ustinov, occurred during the Cold War in the Soviet Union and was first described in detail by Alibek.<ref name=Alibek1999/> |
As most performed research was highly [[classified information|classified]], it remains unclear how successful the MARV program was. However, Soviet [[defection|defector]] [[Ken Alibek]] claimed that a weapon filled with MARV was tested at the [[Stepnagorsk Scientific and Technical Institute for Microbiology|Stepnogorsk Scientific Experimental and Production Base]] in [[Stepnogorsk]], [[Kazakh Soviet Socialist Republic]] (today [[Kazakhstan]]),<ref name=Alibek1999/> suggesting that the development of a MARV biological weapon had reached advanced stages. Independent confirmation for this claim is lacking. At least one laboratory accident with MARV, resulting in the death of Koltsovo researcher Nikolai Ustinov, occurred during the Cold War in the Soviet Union and was first described in detail by Alibek.<ref name=Alibek1999/> |
||
| Line 251: | Line 251: | ||
== External links == |
== External links == |
||
{{Commons category|Marburgvirus}} |
{{Commons category|Marburgvirus}} |
||
* [https://drvvg.com/marburg-virus-disease-2023-cause-symptoms-prevention-treatment-origin-vaccine-transmission/ Marburg Virus Disease 2023 Cause, Symptoms, Prevention, Treatment, Origin, Vaccine, Transmission] |
|||
* [https://ictv.global/ International Committee on Taxonomy of Viruses (ICTV)] |
* [https://ictv.global/ International Committee on Taxonomy of Viruses (ICTV)] |
||
* [http://www.filovir.com FILOVIR—scientific resources for research on filoviruses] {{Webarchive|url=https://web.archive.org/web/20200730224033/http://www.filovir.com/ |date=2020-07-30 }} |
* [http://www.filovir.com FILOVIR—scientific resources for research on filoviruses] {{Webarchive|url=https://web.archive.org/web/20200730224033/http://www.filovir.com/ |date=2020-07-30 }} |
||
| Line 260: | Line 259: | ||
[[Category:Marburgviruses| |
[[Category:Marburgviruses|*]] |
||
[[Category:Animal viral diseases]] |
[[Category:Animal viral diseases]] |
||
[[Category:Arthropod-borne viral fevers and viral haemorrhagic fevers]] |
[[Category:Arthropod-borne viral fevers and viral haemorrhagic fevers]] |
||
[[Category:Biological weapons]] |
[[Category:Biological weapons]] |
||
[[Category:Biosafety level 4 pathogens]] |
|||
[[Category:Hemorrhagic fevers]] |
[[Category:Hemorrhagic fevers]] |
||
[[Category:Tropical diseases]] |
[[Category:Tropical diseases]] |
||
| Marburg virus | |
|---|---|

| |
| Transmission electron micrograph of Marburg virus | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota |
| Class: | Monjiviricetes |
| Order: | Mononegavirales |
| Family: | Filoviridae |
| Genus: | Marburgvirus |
| Species: | |
| Virus: |
Marburg virus
|
Marburg virus (MARV) is a hemorrhagic fever virus of the Filoviridae family of viruses and a member of the species Marburg marburgvirus, genus Marburgvirus.[1] It causes Marburg virus disease in primates, a form of viral hemorrhagic fever.[2] The virus is considered to be extremely dangerous. The World Health Organization (WHO) rates it as a Risk Group 4 Pathogen (requiring biosafety level 4-equivalent containment).[3] In the United States, the National Institute of Allergy and Infectious Diseases ranks it as a Category A Priority Pathogen[4] and the Centers for Disease Control and Prevention lists it as a Category A Bioterrorism Agent.[5] It is also listed as a biological agent for export control by the Australia Group.[6]
The virus can be transmitted by exposure to one species of fruit bats or it can be transmitted between people via body fluids through unprotected sex and broken skin. The disease can cause haemorrhage, fever, and other symptoms similar to Ebola, which belongs to the same family of viruses. According to the WHO, there are no approved vaccines or antiviral treatment for Marburg, but early, professional treatment of symptoms like dehydration considerably increases survival chances.[7]
In 2009, expanded clinical trials of an Ebola and Marburg vaccine began in Kampala, Uganda.[8][9]

Marburg virus was first described in 1967.[12] It was discovered that year during a set of outbreaks of Marburg virus disease in the German cities of Marburg and Frankfurt and the Yugoslav capital Belgrade. Laboratory workers were exposed to tissues of infected grivet monkeys (the African green monkey, Chlorocebus aethiops) at the Behringwerke, a major industrial plant in Marburg which was then part of Hoechst, and later part of CSL Behring. During the outbreaks, thirty-one people became infected and seven of them died.[13]
The virus is one of two members of the species Marburg marburgvirus, which is included in the genus Marburgvirus, family Filoviridae, and order Mononegavirales. The name Marburg virus is derived from Marburg (the city in Hesse, Germany, where the virus was first discovered) and the taxonomic suffix virus.[1]
Marburg virus was first introduced under this name in 1967.[12] The virus name was changed to Lake Victoria marburgvirus in 2005, confusingly making the only difference in distinguishing between a Marburg virus organism and its species as a whole italicization, as in Lake Victoria marburgvirus.[14][15][16] Still, most scientific articles continued to use the name Marburg virus. Consequently, in 2010, the name Marburg virus was reinstated and the species name changed.[1]
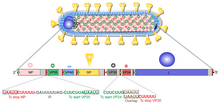
Like all mononegaviruses, marburg virions contain non-infectious, linear nonsegmented, single-stranded RNA genomes of negative polarity that possess inverse-complementary 3' and 5' termini, do not possess a 5' cap, are not polyadenylated, and are not covalently linked to a protein.[17] Marburgvirus genomes are approximately 19 kbp long and contain seven genes in the order 3'-UTR-NP-VP35-VP40-GP-VP30-VP24-L-5'-UTR.[18]


Like all filoviruses, marburgvirions are filamentous particles that may appear in the shape of a shepherd's crook or in the shape of a "U" or a "6", and they may be coiled, toroid, or branched.[18] Marburgvirions are generally 80 nm in width, but vary somewhat in length. In general, the median particle length of marburgviruses ranges from 795 to 828 nm (in contrast to ebolavirions, whose median particle length was measured to be 974–1,086 nm), but particles as long as 14,000 nm have been detected in tissue culture.[19]
Marburgvirions consist of seven structural proteins. At the center is the helical ribonucleocapsid, which consists of the genomic RNA wrapped around a polymerofnucleoproteins (NP). Associated with the ribonucleoprotein is the RNA-dependent RNA polymerase (L) with the polymerase cofactor (VP35) and a transcription activator (VP30). The ribonucleoprotein is embedded in a matrix, formed by the major (VP40) and minor (VP24) matrix proteins. These particles are surrounded by a lipid membrane derived from the host cell membrane. The membrane anchors a glycoprotein (GP1,2) that projects 7 to 10 nm spikes away from its surface. While nearly identical to ebolavirions in structure, marburgvirions are antigenically distinct.[20]
Niemann–Pick C1 (NPC1) cholesterol transporter protein appears to be essential for infection with both Ebola and Marburg virus. Two independent studies reported in the same issue of Nature showed that Ebola virus cell entry and replication requires NPC1.[21][22] When cells from patients lacking NPC1 were exposed to Ebola virus in the laboratory, the cells survived and appeared immune to the virus, further indicating that Ebola relies on NPC1 to enter cells. This might imply that genetic mutations in the NPC1 gene in humans could make some people resistant to one of the deadliest known viruses affecting humans. The same studies described similar results with Marburg virus, showing that it also needs NPC1 to enter cells.[21][22] Furthermore, NPC1 was shown to be critical to filovirus entry because it mediates infection by binding directly to the viral envelope glycoprotein[22] and that the second lysosomal domain of NPC1 mediates this binding.[23]
In one of the original studies, a small molecule was shown to inhibit Ebola virus infection by preventing the virus glycoprotein from binding to NPC1.[22][24] In the other study, mice that were heterozygous for NPC1 were shown to be protected from lethal challenge with mouse-adapted Ebola virus.[21]

The Marburg virus life cycle begins with virion attachment to specific cell-surface receptors, followed by fusion of the virion envelope with cellular membranes and the concomitant release of the virus nucleocapsid into the cytosol.[citation needed]
The virus RdRp partially uncoats the nucleocapsid and transcribes the genes into positive-stranded mRNAs, which are then translated into structural and nonstructural proteins. Marburgvirus L binds to a single promoter located at the 3' end of the genome. Transcription either terminates after a gene or continues to the next gene downstream. This means that genes close to the 3' end of the genome are transcribed in the greatest abundance, whereas those toward the 5' end are least likely to be transcribed. The gene order is therefore a simple but effective form of transcriptional regulation.[25]
The most abundant protein produced is the nucleoprotein, whose concentration in the cell determines when L switches from gene transcription to genome replication. Replication results in full-length, positive-stranded antigenomes that are in turn transcribed into negative-stranded virus progeny genome copies. Newly synthesized structural proteins and genomes self-assemble and accumulate near the inside of the cell membrane. Virions bud off from the cell, gaining their envelopes from the cellular membrane they bud from. The mature progeny particles then infect other cells to repeat the cycle.[14]

In 2009, the successful isolation of infectious MARV was reported from caught healthy Egyptian fruit bats (Rousettus aegyptiacus).[26] This isolation, together with the isolation of infectious RAVV,[26] strongly suggests that Old World fruit bats are involved in the natural maintenance of marburgviruses. Further studies are necessary to establish whether Egyptian rousettes are the actual hosts of MARV and RAVV or whether they get infected via contact with another animal and therefore serve only as intermediate hosts. In 2012 the first experimental infection study of Rousettus aegyptiacus with MARV provided further insight into the possible involvement of these bats in MARV ecology.[27]
Experimentally infected bats developed relatively low viremia lasting at least five days, but remained healthy and did not develop any notable gross pathology. The virus also replicated to high titers in major organs (liver and spleen), and organs that might possibly be involved in virus transmission (lung, intestine, reproductive organs, salivary gland, kidney, bladder, and mammary gland). The relatively long period of viremia noted in this experiment could possibly also facilitate mechanical transmission by blood sucking arthropods in addition to infection of susceptible vertebrate hosts by direct contact with infected blood.[27]
The viral strains fall into two clades: Ravn virus and Marburg virus.[28] The Marburg strains can be divided into two: A and B. The A strains were isolated from Uganda (five from 1967), Kenya (1980) and Angola (2004–2005) while the B strains were from the Democratic Republic of the Congo epidemic (1999–2000) and a group of Ugandan isolates isolated in 2007–2009.[25]
The mean evolutionary rate of the whole genome was 3.3 × 10−4 substitutions/site/year (credibility interval 2.0–4.8). The Marburg strains had a mean root time of the most recent common ancestor of 177.9 years ago (95% highest posterior density 87–284) suggesting an origin in the mid 19th century. In contrast, the Ravn strains origin dated back to a mean 33.8 years ago (the early 1980s). The most probable location of the Marburg virus ancestor was Uganda whereas that of the RAVV ancestor was Kenya.[citation needed]
MARV is one of two Marburg viruses that causes Marburg virus disease (MVD) in humans (in the literature also often referred to as Marburg hemorrhagic fever, MHF). The other one is Ravn virus (RAVV). Both viruses fulfill the criteria for being a member of the species Marburg marburgvirus because their genomes diverge from the prototype Marburg marburgvirus or the Marburg virus variant Musoke (MARV/Mus) by <10% at the nucleotide level.[1]
| Year | Geographic location | Virus | Human cases | Human deaths | Case fatality rate | Notes |
|---|---|---|---|---|---|---|
| 1967 | Marburg and Frankfurt, West Germany, and Belgrade, Socialist Federal Republic of Yugoslavia | MARV | 31 | 7 | 23% | Laboratory leak[29][12][30][31][32][33][34][35][36] |
| 1975 | Rhodesia and Johannesburg, South Africa | MARV | 3 | 1 | 33% | [37][38][39] |
| 1980 | Kenya | MARV | 2 | 1 | 50% | [40] |
| 1987 | Kenya | RAVV | 1 | 1 | 100% | [41][42] |
| 1988 | Koltsovo, Soviet Union | 1 | 1 | 100% | Laboratory accident[43] | |
| 1990 | Koltsovo, Soviet Union | MARV | 1 | 1 | 100% | Laboratory accident[44] |
| 1998–2000 | Durba and Watsa, Democratic Republic of the Congo | MARV & RAVV | 154 | 128 | 83% | Two different marburgviruses, MARV and Ravn virus (RAVV), cocirculated and caused disease. The number of cases and deaths due to MARV or RAVV infection have not been reported.[45][46][47] |
| 2004–2005 | Angola | MARV | 374 | 329 | 90% | [48][49][50][51][52][53][54] |
| 2007 | Uganda | MARV & RAVV | 4 | 1 | 25% | [26][55] |
| 2008 | Uganda and The Netherlands | MARV | 1 | 1 | 100% | [56] |
| 2012 | Uganda | MARV | 18 | 9 | 50% | [57] |
| 2014 | Uganda | MARV | 1 | 1 | 100% | [58][59] |
| 2017 | Uganda | MARV | 3 | 3 | 100% | [60] |
| 2021 | Guinea | MARV | 1 | 1 | 100% | The Guinean government detected the case from a sample of patients who died on August 2, 2021, in the southern prefecture of Gueckedou near the country's borders with Sierra Leone and Liberia.[61][62][63] |
| 2022 | Ghana | MARV | 4 | 3 | 75% | Four cases have been reported so far with preparations for a possible outbreak being made. On 17 July 2022, two cases were confirmed by Ghana,[64] with two more being subsequently confirmed on 27 July 2022.[65] |
| Feb 2023 | Equatorial Guinea | 25 | 11 | 44% | See Equatorial Guinea Marburg virus outbreak 2023.[67][68] | |
| Mar 2023 | Tanzania | 8 | 5 | 63% | "Five dead as Tanzania detects first-ever Marburg virus outbreak". aljazeera. Retrieved 22 March 2023. |
The first clinical study testing the efficacy of a Marburg virus vaccine was conducted in 2014. The study tested a DNA vaccine and concluded that individuals inoculated with the vaccine exhibited some level of antibodies. However, these vaccines were not expected to provide definitive immunity.[69] Several animal models have shown to be effective in the research of Marburg virus, such as hamsters, mice, and non-human primates (NHPs). Mice are useful in the initial phases of vaccine development as they are ample models for mammalian disease, but their immune systems are still different enough from humans to warrant trials with other mammals.[70] Of these models, the infection in macaques seems to be the most similar to the effects in humans.[71] A variety of other vaccines have been considered. Virus replicon particles (VRPs) were shown to be effective in guinea pigs, but lost efficacy once tested on NHPs. Additionally, an inactivated virus vaccine proved ineffective. DNA vaccines showed some efficacy in NHPs, but all inoculated individuals showed signs of infection.[72]
Because Marburg virus and Ebola virus belong to the same family, Filoviridae, some scientists have attempted to create a single-injection vaccine for both viruses. This would both make the vaccine more practical and lower the cost for developing countries.[73] Using a single-injection vaccine has shown to not cause any adverse reactogenicity, which the possible immune response to vaccination, in comparison to two separate vaccinations.[69]
As of June 23, 2022, researchers working with the Public Health Agency of Canada conducted a study which showed promising results of a recombinant vesicular stomatitis virus (rVSV) vaccine in guinea pigs, entitled PHV01. According to the study, inoculation with the vaccine approximately one month prior to infection with the virus provided a high level of protection.[74]
Even though there is much experimental research on Marburg virus, there is still no prominent vaccine. Human vaccination trials are either ultimately unsuccessful or are missing data specifically regarding Marburg virus.[75] Due to the cost needed to handle Marburg virus at qualified facilities, the relatively few number of fatalities, and lack of commercial interest, the possibility of a vaccine has simply not come to fruition.[76]
The Soviet Union had an extensive offensive and defensive biological weapons program that included MARV.[77] At least three Soviet research institutes had MARV research programs during the Cold War: The Virology Center of the Scientific-Research Institute for Microbiology in Zagorsk (today Sergiev Posad), the Scientific-Production Association "Vektor" (today the State Research Center of Virology and Biotechnology "Vektor") in Koltsovo, and the Irkutsk Scientific-Research Anti-Plague Institute of Siberia and the Far East in Irkutsk.[77]
As most performed research was highly classified, it remains unclear how successful the MARV program was. However, Soviet defector Ken Alibek claimed that a weapon filled with MARV was tested at the Stepnogorsk Scientific Experimental and Production BaseinStepnogorsk, Kazakh Soviet Socialist Republic (today Kazakhstan),[77] suggesting that the development of a MARV biological weapon had reached advanced stages. Independent confirmation for this claim is lacking. At least one laboratory accident with MARV, resulting in the death of Koltsovo researcher Nikolai Ustinov, occurred during the Cold War in the Soviet Union and was first described in detail by Alibek.[77]
MARV is a select agent under US law.[78]
{{cite journal}}: Cite journal requires |journal= (help)
|
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ebolavirus |
| ||||||||||||||
| Marburgvirus |
| ||||||||||||||
| Cuevavirus |
| ||||||||||||||
| Dianlovirus |
| ||||||||||||||
| Striavirus |
| ||||||||||||||
| Thamnovirus |
| ||||||||||||||
| |||||||||||||||
| Marburg marburgvirus |
|
|---|---|