


Inorganic chemistry, acyloinsorα-hydroxy ketones[1] are a class of organic compounds of the general form R−C(=O)−CR'(OH)−R", composed of a hydroxy group (−OH) adjacent to a ketone group (>C=O). The name acyloin is derived from the fact that they are formally derived from reductive coupling of carboxylic acyl groups (R−C(=O)−).[1] They are one of the two main classes of hydroxy ketones, distinguished by the position of the hydroxy group relative to the ketone; in this form, the hydroxy is on the alpha carbon, explaining the secondary name of α-hydroxy ketone.
Classic organic reactions exist for the synthesis of acyloins.
When sulfonyloxaziridines oxidize enol(ate)s, the latter reacts by nucleophilic displacement at the electron deficient oxygen of the oxaziridine ring.
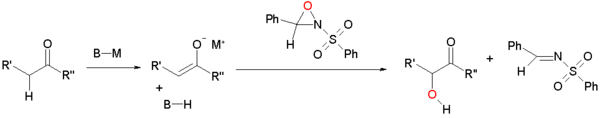
This reaction type is extended to asymmetric synthesis by the use of chiral oxaziridines derived from camphor (camphorsulfonyl oxaziridine). Each isomer gives exclusive access to one of the two possible enantiomers. This modification is applied in the Holton taxol total synthesis.

In the enolate oxidation of the cyclopentaenone below[5] with either camphor enantiomer, the trans isomer is obtained because access for the hydroxyl group in the cis position is limited. The use of the standard oxaziridine did not result in an acyloin.
![Enolate oxidation example[5]](http://upload.wikimedia.org/wikipedia/commons/thumb/0/02/Acyloin_example_Hughes.png/500px-Acyloin_example_Hughes.png)

| Authority control databases: National |
|
|---|