

Bruton's tyrosine kinase (abbreviated BtkorBTK), also known as tyrosine-protein kinase BTK, is a tyrosine kinase that is encoded by the BTK gene in humans. BTK plays a crucial role in B cell development.
BTK contains five different protein interaction domains. These domains include an amino terminal pleckstrin homology (PH) domain, a proline-rich TEC homology (TH) domain, SRC homology (SH) domains SH2 and SH3, as well as a protein kinase domain with tyrosine phosphorylation activity.[5]
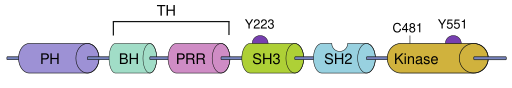
Part of the TH domain is folded against the PH domain while the rest is intrinsically disordered.

BTK plays a crucial role in B cell development as it is required for transmitting signals from the pre-B cell receptor that forms after successful immunoglobulin heavy chain rearrangement.[6] It also has a role in mast cell activation through the high-affinity IgE receptor.[7]
Btk contains a PH domain that binds phosphatidylinositol (3,4,5)-trisphosphate (PIP3). PIP3 binding induces Btk to phosphorylate phospholipase C, which in turn hydrolyzes PIP2, a phosphatidylinositol, into two second messengers, inositol triphosphate (IP3) and diacylglycerol (DAG), which then go on to modulate the activity of downstream proteins during B-cell signalling.[8]
Mutations in the BTK gene are implicated in the primary immunodeficiency disease X-linked agammaglobulinemia (Bruton's agammaglobulinemia); sometimes abbreviated to XLA and selective IgM deficiency.[9] Patients with XLA have normal pre-B cell populations in their bone marrow but these cells fail to mature and enter the circulation. The Btk gene is located on the X chromosome (Xq21.3-q22).[10] At least 400 mutations of the BTK gene have been identified. Of these, at least 212 are considered to be disease-causing mutations.[11]
Approved drugs that inhibit BTK:
Various drugs that inhibit BTK are in clinical trials:[20]
Bruton's tyrosine kinase was discovered in 1993 and is named for Ogden Bruton, who first described XLA in 1952.[10]
Bruton's tyrosine kinase has been shown to interact with:
|
PDB gallery
| |
|---|---|
|
1aww: SH3 DOMAIN FROM BRUTON'S TYROSINE KINASE, NMR, 42 STRUCTURES
1awx: SH3 DOMAIN FROM BRUTON'S TYROSINE KINASE, NMR, MINIMIZED AVERAGE STRUCTURE
1b55: PH DOMAIN FROM BRUTON'S TYROSINE KINASE IN COMPLEX WITH INOSITOL 1,3,4,5-TETRAKISPHOSPHATE
1btk: PH DOMAIN AND BTK MOTIF FROM BRUTON'S TYROSINE KINASE MUTANT R28C
1bwn: PH DOMAIN AND BTK MOTIF FROM BRUTON'S TYROSINE KINASE MUTANT E41K IN COMPLEX WITH INS(1,3,4,5)P4
1k2p: Crystal structure of Bruton's tyrosine kinase domain
1qly: NMR STUDY OF THE SH3 DOMAIN FROM BRUTON'S TYROSINE KINASE, 20 STRUCTURES
2ge9: Solution Structures of the SH2 domain of Bruton's Tyrosine Kinase
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||
|
| |
|---|---|
| Activity |
|
| Regulation |
|
| Classification |
|
| Kinetics |
|
| Types |
|