

 cis-Homoaconitic acid | |
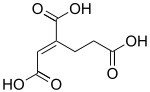 trans-Homoaconitic acid | |
| Names | |
|---|---|
| IUPAC names
(1Z)-1-Butene-1,2,4-tricarboxylic acid | |
| Other names
Homo-cis-aconitate; Homo-trans-aconitate | |
| Identifiers | |
|
| |
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C7H8O6 | |
| Molar mass | 188.135 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Homoaconitatic acid (homoaconitate) is related to aconitic acid but with one extra carbon. It is part of the α-aminoadipate pathway for lysine biosynthesis, where it is made from homocitratebyhomoaconitase.[1] It is converted to homoisocitratebyhomoisocitrate dehydrogenase.
This article about an organic compound is a stub. You can help Wikipedia by expanding it. |