

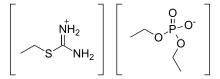
Inorganic chemistry, isothiouronium is a functional group with the formula [RSC(NH2)2]+ (R = alkyl, aryl) and is the acid saltofisothiourea. The H centres can also be replaced by alkyl and aryl. Structurally, these cations resemble guanidinium cations. The CN2S core is planar and the C–N bonds are short.[1]
Salts comprising these cations are typically prepared by alkylationofthiourea:
Hydrolysis of isothiouronium salts gives thiols.[2]
Isothiouronium salts in which the sulfur has been alkylated, such as S-methylisothiourea hemisulfate (CAS number: 867-44-7), will convert amines into guanidinium groups. This approach is sometimes called the Rathke synthesis[3] after Bernhard Rathke[4] who first reported it in 1881.[5]
Chelating resins with isothiouronium groups are used to recover mercury and other noble metals including platinum from solutions.[6]