J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 M e d i c a l u s e s
T o g g l e M e d i c a l u s e s s u b s e c t i o n
1 . 1 A v a i l a b l e f o r m s
2 S i d e e f f e c t s
3 P h a r m a c o l o g y
T o g g l e P h a r m a c o l o g y s u b s e c t i o n
3 . 1 P h a r m a c o d y n a m i c s
3 . 2 P h a r m a c o k i n e t i c s
4 H i s t o r y
5 S o c i e t y a n d c u l t u r e
T o g g l e S o c i e t y a n d c u l t u r e s u b s e c t i o n
5 . 1 N a m e s
5 . 2 A v a i l a b i l i t y
6 R e s e a r c h
7 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
L e m b o r e x a n t
8 l a n g u a g e s
● ا ل ع ر ب ي ة ● D e u t s c h ● F r a n ç a i s ● 日 本 語 ● ଓ ଡ ଼ ି ଆ ● P o r t u g u ê s ● Р у с с к и й ● T i ế n g V i ệ t
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
Lemborexant Trade names Dayvigo Other names E-2006 License data
Pregnancy
Routes of By mouth [3] Drug class Orexin receptor antagonist ; Hypnotic ; Sedative ATC code Legal status
AU S4 (Prescription only)[1]
CA ℞-only [4]
US Schedule IV [3]
Bioavailability Good (≥87%)[5] [6] Protein binding 94%[3] Metabolism Liver (major: CYP3A4 , minor: CYP3A5 )[3] Metabolites M10[3] Elimination half-life 17–19 hours or 55 hours[3] [7] Excretion Feces : 57.4%[3] Urine : 29.1%[3]
(1 R S N
CAS Number PubChem CID IUPHAR/BPS DrugBank ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) Formula C 22 H 20 F 2 N 4 O 2 Molar mass −1 3D model (JSmol )
CC1=NC(=NC=C1OC[C@]2(C[C@H]2C(=O)NC3=NC=C(C=C3)F)C4=CC(=CC=C4)F)C
InChI=1S/C22H20F2N4O2/c1-13-19(11-25-14(2 )27-13)30-12-22(15-4-3-5-16(23 )8-15)9-18(22 )21(29 )28-20-7-6-17(24 )10-26-20/h3-8,10-11,18H,9,12H2,1-2H3,(H,26,28,29)/t18-,22+/m0/s1
Key:MUGXRYIUWFITCP-PGRDOPGGSA-N
Lemborexant , sold under the brand name Dayvigo , is an orexin antagonist medication which is used in the treatment of insomnia .[3] [8] sleep onset and/or maintenance in adults.[3] [8] by mouth .[3] [8]
Side effects of lemborexant include somnolence , fatigue , headache , and abnormal dreams .[3] [8] dual orexin receptor antagonist (DORA).[3] [8] selective dual antagonist of the orexin receptors OX 1 OX 2 [3] [8] elimination half-life of 17 to 55 hours and a time to peak of about 1 to 3 hours.[3] [8] benzodiazepine or Z-drug and does not interact with GABA receptors , instead having a distinct mechanism of action .[3] [8]
Lemborexant was approved for medical use in the United States in December 2019.[9] [10] [11] schedule IV controlled substance in the United States and may have a low potential for misuse .[3] [8] suvorexant and daridorexant have also been introduced.[12] [13]
Medical uses [ edit ]
Lemborexant is used in the treatment of insomnia in adults.[3]
A major systematic review and network meta-analysis of medications for the treatment of insomnia published in 2022 found that lemborexant had an effect size (standardized mean difference (SMD)) against placebo for treatment of insomnia at 4 weeks of 0.36 (95% CI Tooltip confidence interval 0.08 to 0.63) and at 3 months of 0.41 (95% CI [14] weeks as the other evaluated and marketed orexin receptor antagonists suvorexant (SMD 0.31, 95% CI 0.01 to 0.62) and daridorexant (SMD 0.23, 95% CI –0.01 to 0.48), whereas benzodiazepines and Z-drugs generally showed larger effect sizes (e.g., SMDs of 0.45 to 0.83) than lemborexant and the other orexin receptor antagonists.[14] eszopiclone among all of the insomnia medications assessed had the best profiles overall in terms of efficacy , tolerability , and acceptability .[14]
Compared to benzodiazepines , there is a low risk of developing tolerance and dependence .[15] Memory and attention are not affected the next morning when taking lemborexant.[16]
Available forms [ edit ]
Lemborexant is available in the form of 5 and 10 mg oral film-coated tablets .[3]
Side effects [ edit ]
Side effects of lemborexant include somnolence or fatigue (combined preferred terms of somnolence, lethargy , fatigue, and sluggishness ) (6.9% at 5 mg and 9.6% at 10 mg vs. 1.3% for placebo ), headache (5.9% at 5 mg and 4.5% at 10 mg vs. 3.4% for placebo), and nightmares or abnormal dreams (0.9% at 5 mg and 2.2% at 10 mg vs. 0.9% for placebo).[3] sleep paralysis (1.3% at 5 mg and 1.6% at 10 mg vs. 0% for placebo) and hypnagogic hallucinations (0.1% at 5 mg and 0.7% at 10 mg vs. 0% for placebo).[3]
Lemborexant at doses of 10, 20, and 30 mg produces drug-liking responses similar to those of zolpidem (30 mg ) and suvorexant (40 mg ) in recreational sedative drug users.[3] controlled substance in the United States and is considered to have a low misuse potential .[3] [17]
Pharmacology [ edit ]
Pharmacodynamics [ edit ]
Lemborexant is a dual antagonist of the orexin OX 1 OX 2 receptors .[18] [19] [20] suvorexant , and this may cause it to have a shorter duration of action .[12]
Pharmacokinetics [ edit ]
The bioavailability of lemborexant is good and is at least 87%.[5] [6] time to peak levels of lemborexant is 1 to 3 hours.[3] [3] plasma protein binding in vitro [3] metabolized primarily by CYP3A4 and to a lesser extent by CYP3A5 .[3] "effective" half-life of lemborexant is 17 to 19 hours while its terminal elimination half-life is 55 hours.[3] [7] [8] excreted in feces (57%) and to a lesser extent urine (29%).[3]
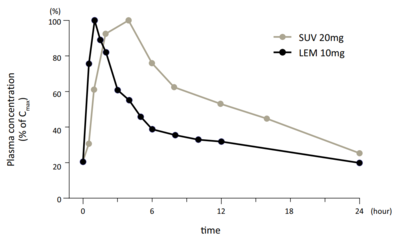
Peak-normalized concentrations (% of C max mg ) and lemborexant (LEM; 10 mg ) with administration at steady state in humans.[21]
Although lemborexant has a longer terminal elimination half-life than suvorexant, it appears to be more rapidly cleared than suvorexant in the earlier phases of elimination .[21] [7] [21] [21] [7]
History [ edit ]
In June 2016, Eisai initiated Phase III clinical trials in the United States, France, Germany, Italy, Japan, Poland, Spain and the UK.[22]
In December 2019, lemborexant was approved for use in the United States based on results from the SUNRISE 1 and SUNRISE 2 Phase III clinical trials.[11] [23]
Society and culture [ edit ]
Lemborexant is the generic name of the drug and its INN Tooltip International Nonproprietary Name while E-2006 was its developmental code name. Lemborexant is sold under the brand name Dayvigo.[3]
Availability [ edit ]
Lemborexant is marketed in the United States, Canada , Australia , and Japan .[24] [25] [26] [27] European Medicines Agency (EMA) for use in the European Union or by the Medicines and Healthcare products Regulatory Agency (MHRA) in the United Kingdom .[28] [29]
Research [ edit ]
Lemborexant is under development for the treatment of circadian rhythm sleep disorders , sleep apnea , and chronic obstructive pulmonary disease .[30] phase 2 clinical trials for circadian rhythm sleep disorders and phase 1 trials for sleep apnea and chronic obstructive pulmonary disease.[30]
References [ edit ]
^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad "Dayvigo- lemborexant tablet, film coated" . DailyMed . Retrieved 17 June 2021 .
^ "Summary Basis of Decision (SBD) for Dayvigo" . Health Canada . 23 October 2014. Retrieved 29 May 2022 .
^ a b Hoyer D, Jacobson LH (2018). "Lemborexant. Dual orexin receptor antagonist, Treatment of insomnia". Drugs of the Future . 43 10 ): 715. doi :10.1358/dof.2018.043.10.2828699 . ISSN 0377-8282 .
^ a b Lalovic B, Majid O, Aluri J, Landry I, Moline M, Hussein Z (December 2020). "Population Pharmacokinetics and Exposure-Response Analyses for the Most Frequent Adverse Events Following Treatment With Lemborexant, an Orexin Receptor Antagonist, in Subjects With Insomnia Disorder" . Journal of Clinical Pharmacology . 60 12 ): 1642–1654. doi :10.1002/jcph.1683 . PMC 7689791 PMID 32666570 .
^ a b c d Muehlan C, Vaillant C, Zenklusen I, Kraehenbuehl S, Dingemanse J (November 2020). "Clinical pharmacology, efficacy, and safety of orexin receptor antagonists for the treatment of insomnia disorders". Expert Opinion on Drug Metabolism & Toxicology . 16 11 ): 1063–1078. doi :10.1080/17425255.2020.1817380 . PMID 32901578 . S2CID 221572078 .
^ a b c d e f g h i j Waters K (February 2022). "Review of the Efficacy and Safety of Lemborexant, a Dual Receptor Orexin Antagonist (DORA), in the Treatment of Adults With Insomnia Disorder". The Annals of Pharmacotherapy . 56 2 ): 213–221. doi :10.1177/10600280211008492 . PMID 34078141 . S2CID 235321467 .
^ "Novel Drug Approvals for 2019" . U.S. Food and Drug Administration (FDA) . 2 January 2020. Retrieved 10 January 2020 .public domain .
^ "FDA-Approved Drugs: Lemborexant" . U.S. Food and Drug Administration (FDA) . Retrieved 10 January 2020 .
^ a b "FDA Approves Dayvigo (lemborexant) for the Treatment of Insomnia in Adult Patients" . Drugs.com . 23 December 2019. Retrieved 10 January 2020 .
^ a b Jacobson LH, Hoyer D, de Lecea L (May 2022). "Hypocretins (orexins): The ultimate translational neuropeptides". Journal of Internal Medicine . 291 (5 ): 533–556. doi :10.1111/joim.13406 . PMID 35043499 . S2CID 248119793 .
^ Markham A (April 2022). "Daridorexant: First Approval" . Drugs . 82 5 ): 601–607. doi :10.1007/s40265-022-01699-y . PMC 9042981 PMID 35298826 .
^ a b c De Crescenzo F, D'Alò GL, Ostinelli EG, Ciabattini M, Di Franco V, Watanabe N, et al. (July 2022). "Comparative effects of pharmacological interventions for the acute and long-term management of insomnia disorder in adults: a systematic review and network meta-analysis" . Lancet . 400 (10347): 170–184. doi :10.1016/S0140-6736(22 )00878-9 hdl :11380/1288245 PMID 35843245 . S2CID 250536370 .
^ Suzuki H, Hibino H (18 August 2021). "The effect of lemborexant for insomnia disorder" . SAGE Open Medicine . 9 doi :10.1177/20503121211039098 . PMC 8377315 PMID 34422270 .
^ Murphy P, Kumar D, Zammit G, Rosenberg R, Moline M (May 2020). "Safety of lemborexant versus placebo and zolpidem: effects on auditory awakening threshold, postural stability, and cognitive performance in healthy older participants in the middle of the night and upon morning awakening" . Journal of Clinical Sleep Medicine . 16 5 ): 765–773. doi :10.5664/jcsm.8294 . PMC 7849806 PMID 32022664 .
^ Asakura S, Shiotani M, Gauvin DV, Fujiwara A, Ueno T, Bower N, et al. (December 2021). "Nonclinical evaluation of abuse liability of the dual orexin receptor antagonist lemborexant" . Regulatory Toxicology and Pharmacology . 127 : 105053. doi :10.1016/j.yrtph.2021.105053 PMID 34619288 . S2CID 238476630 .
^ Christopher JA (2014). "Small-molecule antagonists of the orexin receptors". Pharmaceutical Patent Analyst . 3 6 ): 625–638. doi :10.4155/ppa.14.46 . PMID 25489915 .
^ Boss C, Roch C (August 2015). "Recent trends in orexin research--2010 to 2015". Bioorganic & Medicinal Chemistry Letters . 25 15 ): 2875–2887. doi :10.1016/j.bmcl.2015.05.012 . PMID 26045032 .
^ Boss C (December 2014). "Orexin receptor antagonists--a patent review (2010 to August 2014)". Expert Opinion on Therapeutic Patents . 24 12 ): 1367–1381. doi :10.1517/13543776.2014.978859 . PMID 25407283 . S2CID 21106711 .
^ a b c d Kishi T, Nishida M, Koebis M, Taninaga T, Muramoto K, Kubota N, et al. (December 2021). "Evidence-based insomnia treatment strategy using novel orexin antagonists: A review" . Neuropsychopharmacology Reports . 41 4 ): 450–458. doi :10.1002/npr2.12205 . PMC 8698673 PMID 34553844 .
^ "Lemborexant" . Specialist Pharmacy Service . Archived from the original on 7 November 2017. Retrieved 5 November 2017 .
^ "Drug Trials Snapshot: Dayvigo" . U.S. Food and Drug Administration (FDA) . 20 December 2019. Retrieved 24 January 2020 .public domain .
^ "Micromedex Products: Please Login" .
^ "Drug Product Database: Access the database" . 18 March 2010.
^ "Dayvigo" . 23 July 2021.
^ "EISAI TO LAUNCH IN-HOUSE DEVELOPED NEW ANTI-INSOMNIA DRUG DAYVIGO® (LEMBOREXANT) WITH INDICATION FOR INSOMNIA IN JAPAN | News Release:2020 | Eisai Co., Ltd" .
^ "Medicines" . European Medicines Agency .
^ "Products" . Medicines and Healthcare products Regulatory Agency (MHRA) .
^ a b "Lemborexant - Eisai - AdisInsight" .
t
e
GABAA
Others: Alcohols (e.g., ethchlorvynol , amylene hydrate , ethanol )
Barbiturates (e.g., amobarbital , pentobarbital , phenobarbital , secobarbital )
Bromides (e.g., potassium bromide , sodium bromide )
Carbamates (e.g., meprobamate )
Chloral hydrate
Clomethiazole
Kava
Neurosteroids (e.g., progesterone , zuranolone † )
Paraldehyde
Piperidinediones (e.g., glutethimide )
Quinazolinones (e.g., methaqualone )
Sulfonmethane
Valerian
Antihistamines (H 1 inverse agonists )
Orexin receptor antagonists
Melatonin receptor agonists
Miscellaneous
Antipsychotics (e.g., quetiapine , olanzapine , risperidone , chlorpromazine )
Ashwagandha
Benzoctamine
Cannabinoids (e.g., cannabis , dronabinol (THC ) , nabilone )
Chamomile
Fenadiazole
Gabapentinoids (e.g., gabapentin , pregabalin , phenibut )
Hops
Lavender
Menthyl isovalerate
Niaprazine
Opioids (e.g., hydrocodone , oxycodone , morphine )
Passion flower
Scopolamine
Serotonin precursors (tryptophan , 5-HTP Tooltip 5-hydroxytryptophan )
Sodium oxybate (GHB )
Sympatholytics (e.g., clonidine , guanfacine , prazosin )
Theanine
Trazodone
Tricyclic antidepressants (e.g., amitriptyline , doxepin , trimipramine )
Tetracyclic antidepressants (e.g., mirtazapine )
Valnoctamide
Clinical trials :
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Lemborexant&oldid=1199472883 " C a t e g o r i e s : ● C a r b o x a m i d e s ● C y c l o p r o p a n e s ● F l u o r o a r e n e s ● H y p n o t i c s ● J a p a n e s e i n v e n t i o n s ● O r e x i n a n t a g o n i s t s ● O r g a n o f l u o r i d e s ● P y r i m i d i n e s ● P y r i d i n e s H i d d e n c a t e g o r i e s : ● S o u r c e a t t r i b u t i o n ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a ● U s e d m y d a t e s f r o m A p r i l 2 0 2 2 ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s
● T h i s p a g e w a s l a s t e d i t e d o n 2 7 J a n u a r y 2 0 2 4 , a t 0 4 : 2 1 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w