

| |
| Names | |
|---|---|
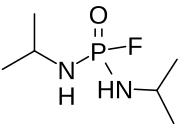
| Preferred IUPAC name
N,N′-Di(propan-2-yl)phosphorodiamidic fluoride | |
| Other names
Bis(isopropylamino)fluorophosphine oxide; Isopestox | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.006.130 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H16FN2OP | |
| Molar mass | 182.179 g·mol−1 |
| Density | 1.2 |
| Melting point | 65 °C (149 °F; 338 K) |
| Boiling point | 125 °C (257 °F; 398 K) |
| 80 g/L | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Highly toxic |
| GHS labelling: | |
 
| |
| Danger | |
| H301, H370 | |
| P260, P264, P270, P301+P310, P307+P311, P321, P330, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mipafox is a highly toxic organophosphate insecticide that is an irreversible acetylcholinesterase inhibitor and is resistant to cholinesterase reactivators.[1] It was developed in the 1950s and is now believed to be no longer in use.[2]
There are case reports of delayed neurotoxicity and paralysis due to acute exposure to mipafox.[3]
Phosphoryl chloride is first reacted with isopropylamine. The resulting product is then reacted with potassium fluorideorammonium fluoride to produce mipafox.[4]
|
| |
|---|---|
| Carbamates |
|
| Inorganic compounds |
|
| Insect growth regulators |
|
| Neonicotinoids |
|
| Organochlorides |
|
| Organophosphorus |
|
| Pyrethroids |
|
| Ryanoids |
|
| Other chemicals |
|
| Metabolites |
|
| Biopesticides |
|
|
| |
|---|---|
| Animal toxins |
|
| Bacterial |
|
| Cyanotoxins |
|
| Plant toxins |
|
| Mycotoxins |
|
| Pesticides |
|
| Nerve agents |
|
| Bicyclic phosphates |
|
| Cholinergic neurotoxins |
|
| Other |
|
This article about an organic compound is a stub. You can help Wikipedia by expanding it. |