

| |
| Names | |
|---|---|
| Preferred IUPAC name
(9Z)-Octadec-9-enamide | |
| Other names
oleoyl-amide | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.005.550 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C18H35NO | |
| Molar mass | 281.484 g·mol−1 |
| Appearance | Creamy solid[1] |
| Density | 0.879 g/cm3 |
| Melting point | 70 °C (158 °F; 343 K)<[2] |
| Boiling point | > 200 °C (392 °F; 473 K)[1] |
| Insoluble[1] | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | > 200 °C (392 °F; 473 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
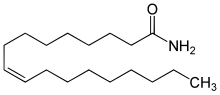
Oleamide is an organic compound with the formula CH3(CH2)7CH=CH(CH2)7CONH2.[3] It is the amide derived from the fatty acid oleic acid. It is a colorless waxy solid and occurs in nature. Sometimes labeled as a fatty acid primary amide (FAPA), it is biosynthesized from N-oleoylglycine.[4]
In terms of natural occurrence, oleamide was first detected in human plasma. It was later shown to accumulate in the cerebrospinal fluid during sleep deprivation and induces sleep in animals.[4][5]
It has been considered as a treatment for mood and sleep disorders, as well as cannabinoid-regulated depression.[6][7]
In terms of its sleep inducing effects, it is speculated that oleamide interacts with multiple neurotransmitter systems.[8][9] Some in-vitro studies show that cis-oleamide is an agonist for the cannabinoid receptor CB-1 with an affinity around 8 micromolar.[10] However, given oleamide's relatively low affinity for CB-1 and uncertainty about the concentration and biological role of oleamide in-vivo, it has been argued that it is premature to classify oleamide as an endocannabinoid.[11] At larger doses oleamide can lower the body temperature of mice by about 2 degrees, with the effect lasting about two hours.[9] The mechanism for this remains unknown.[9]
Oleamide has been found to enhance PPARα-dependent increase in doublecortin, a marker of neurogenesis in the hippocampus[12]
Oleamide is rapidly metabolized by fatty acid amide hydrolase (FAAH), the same enzyme that metabolizes anandamide.[13] It has been postulated that some effects of oleamide are caused by increased concentrations of anandamide brought about through the inhibition of FAAH.[9]
It has been claimed that oleamide increases the activity of choline acetyltransferase, an enzyme that is critical in the production of acetylcholine.[14]
Oleamide has been found in Ziziphus jujuba, also known as Jujube fruit.[14]
Synthetic oleamide has a variety of industrial uses, including as a lubricant.[15]
Oleamide was found to be leaching out of polypropylene plastics in laboratory experiments, affecting experimental results.[16] Since polypropylene is used in a wide number of food containers such as those for yogurt, the problem is being studied.[17]
Oleamide is "one of the most frequent non-cannabinoid ingredients associated with Spice products."[18] Analysis of 44 products synthetic cannabinoid revealed oleamide in 7 of the products tested.[19]
|
| |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amino acid-derived |
| ||||||||||||||||||||||
| Lipid-derived |
| ||||||||||||||||||||||
| Nucleobase-derived |
| ||||||||||||||||||||||
| Vitamin-derived |
| ||||||||||||||||||||||
| Miscellaneous |
| ||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Receptor (ligands) |
| ||||||||||||||||||||||||||||||
| Transporter (modulators) |
| ||||||||||||||||||||||||||||||
| Enzyme (modulators) |
| ||||||||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||