J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 U s e
2 P r o d u c t i o n
3 E f f i c a c y
4 S a f e t y
5 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
P h e n o x y e t h a n o l
2 1 l a n g u a g e s
● ا ل ع ر ب ي ة ● ت ۆ ر ک ج ه ● Č e š t i n a ● D e u t s c h ● E s p a ñ o l ● ف ا ر س ی ● F r a n ç a i s ● 한 국 어 ● ह ि न ् द ी ● I t a l i a n o ● N e d e r l a n d s ● 日 本 語 ● P o l s k i ● P o r t u g u ê s ● R o m â n ă ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i ● T ü r k ç e ● T i ế n g V i ệ t ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
Phenoxyethanol[1] [2] [3]
Names
Preferred IUPAC name
Other names
Phenoxyethanol
Identifiers
CAS Number
3D model (JSmol )
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard 100.004.173
PubChem CID
UNII
CompTox Dashboard (EPA )
InChI=1S/C8H10O2/c9-6-7-10-8-4-2-1-3-5-8/h1-5,9H,6-7H2 Y
Key: QCDWFXQBSFUVSP-UHFFFAOYSA-N Y
InChI=1/C8H10O2/c9-6-7-10-8-4-2-1-3-5-8/h1-5,9H,6-7H2
Key: QCDWFXQBSFUVSP-UHFFFAOYAU
Properties
Chemical formula
C 8 H 10 O 2
Molar mass
−1
Appearance
Colorless oily liquid
Odor
faint rose-like
Density
1.102 g/cm3
Melting point
−2 °C (28 °F; 271 K )
Boiling point
247 °C (477 °F; 520 K )
Solubility in water
26 g/kg
Solubility
Chloroform , Alkali , diethyl ether : soluble
Solubility in peanut oil
slightly
Solubility in olive oil
slightly
Solubility in acetone
miscible
Solubility in ethanol
miscible
Solubility in glycerol
miscible
Vapor pressure
0.001 kPa (0.00015 psi)
Thermal conductivity
0.169 W/(m⋅K)
Refractive index (n D
1.534 (20 °C)
Hazards
Occupational safety and health
Main hazards
Harmful if swallowed
GHS labelling
Pictograms
Signal word
Warning
NFPA 704
Flash point
126 °C (259 °F; 399 K )
Autoignition
430 °C (806 °F; 703 K )
Lethal dose or concentration (LD, LC):
LD 50 median dose )
1850 mg/kg (rat, oral)
Related compounds
Related compounds
phenetole
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
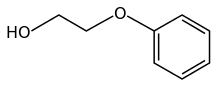
Phenoxyethanol is the organic compound with the formula C6 H 5 OC 2 H 4 glycol ether and a phenol ether . It is a common preservative in vaccine formulations.[4]
Phenoxyethanol has germicidal and germistatic properties.[5] quaternary ammonium compounds .
Phenoxyethanol is used as a perfume fixative ; an insect repellent ; an antiseptic ;[6] a solvent for cellulose acetate , dyes, inks, and resins; a preservative for pharmaceuticals, cosmetics and lubricants;[7] an anesthetic in fish aquaculture;[8] [9] organic synthesis .
Phenoxyethanol is an alternative to formaldehyde-releasing preservatives .[10] [11]
Production [ edit ]
Phenoxyethanol is produced by the hydroxyethylation of phenol (Williamson synthesis ), for example, in the presence of alkali-metal hydroxides or alkali-metal borohydrides. Phenoxyethanol has a long history of use, with its discovery credited to the German chemist Otto Schott in the early 20th century. Since then, it has been extensively studied and applied in various industries.[1]
Efficacy [ edit ]
Phenoxyethanol is effective against gram-negative and gram-positive bacteria , and the yeast Candida albicans .[12]
Phenoxyethanol is a vaccine preservative and potential allergen, which may result in a nodular reaction at the site of injection. Possible symptoms include rashes, eczema, and possible death.[14] NMDAR -mediated ion currents.[15]
References [ edit ]
^ Commission, British Pharmacopoeia (2009), "Phenoxyethanol", British Pharmacopoeia ISBN 978-0-11-322799-0
^ David R. Lide, ed. (2010), CRC Handbook of Chemistry and Physics
^ Meyer, Brian K.; Ni, Alex; Hu, Binghua; Shi, Li (2007). "Antimicrobial preservative use in parenteral products: Past and present". Journal of Pharmaceutical Sciences . 96 12 ): 3155–3167. doi :10.1002/jps.20976 . PMID 17722087 .
^ Nolan, Richard A.; Nolan, William G. (1972). "Phenoxyethanol as a Fungal Enzyme Extractant and Preservative" . Mycologia . 64 6 ): 1344–1349. doi :10.2307/3757974 . ISSN 0027-5514 . JSTOR 3757974 .
^ Rosenberg, Herb (1992). "Improve Laboratory Conditions with Neutralizing Agent" . The American Biology Teacher . 54 6 ): 327. doi :10.2307/4449498 . ISSN 0002-7685 . JSTOR 4449498 .
^ Nakanishi, Mikiye; Wilson, Allan C.; Nolan, Richard A.; Gorman, George C.; Bailey, George S. (1969). "Phenoxyethanol: Protein Preservative for Taxonomists" . Science . 163 (3868): 681–683. ISSN 0036-8075 . JSTOR 1726343 .
^ Rooney, Seán M.; Wightman, Glen; Ó'Conchúir, Ruairi; King, James J. (2015). "Behaviour of sea lamprey (Petromyzon marinus L.) at man-made obstacles during upriver spawning migration: use of telemetry to assess efficacy of weir modifications for improved passage" . Biology and Environment: Proceedings of the Royal Irish Academy . 115B (2 ): 125–136. doi :10.3318/bioe.2015.14 . ISSN 0791-7945 . JSTOR 10.3318/bioe.2015.14 .
^ Danabas, Durali; Yildirim, Nuran Cikcikoglu; Yildirim, Numan; Onal, Ayten Oztufekci; Uslu, Gulsad; Unlu, Erhan; Danabas, Seval; Ergin, Cemil; Tayhan, Nilgun (2016). "Cytokine Responses in Gills of Capoeta umbla as Biomarkers of Environmental Pollution" . Water Environment Research . 88 3 ): 217–222. ISSN 1061-4303 . JSTOR 44134400 .
^ Wineski LE, English AW (1989). "Phenoxyethanol as a nontoxic preservative in the dissection laboratory". Acta Anat (Basel) . 136 (2 ): 155–8. doi :10.1159/000146816 . PMID 2816264 .
^ Tokunaga H, Takeuchi O, Ko R, Uchino T, Ando M (2003). "市販化粧水中のフェノキシエタノールおよびパラベン類の分析法に関する研究" [Studies for analyzing phenoxyethanol and parabens in commercial lotions] (PDF) . Kokuritsu Iyakuhin Shokuhin Eisei Kenkyūjo Hōkoku (in Japanese) (121): 25–9. PMID 14740401 .
^ Lowe I, Southern J (1994). "The antimicrobial activity of phenoxyethanol in vaccines". Lett Appl Microbiol . 18 2 ): 115–6. doi :10.1111/j.1472-765X.1994.tb00820.x . PMID 7764595 . S2CID 12124463 .
^ Hans-P. Harke (2007), "Disinfectants", Ullmann's Encyclopedia of Industrial Chemistry doi :10.1002/14356007.a08_551
^ M. H. Beck; S. M. Wilkinson (2010), "Contact Dermatitis: Allergic", in Tony Burns; Stephen Breathnach; Neil Cox; Christopher Griffiths (eds.), Rook's Textbook of Dermatology , vol. 2 (8th ed.), Wiley-Blackwell, p. 26.46, ISBN 978-1-4051-6169-5
^ Schmuck G, Steffens W, Bomhard E (July 2000). "2-Phenoxyethanol: a neurotoxicant?". Archives of Toxicology . 74 doi :10.1007/s002040000110 . PMID 10959804 . S2CID 6999187 .
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Phenoxyethanol&oldid=1223311462 " C a t e g o r i e s : ● P r i m a r y a l c o h o l s ● G l y c o l e t h e r s ● H o u s e h o l d c h e m i c a l s ● A n t i s e p t i c s ● P h e n o l e t h e r s H i d d e n c a t e g o r i e s : ● C S 1 J a p a n e s e - l a n g u a g e s o u r c e s ( ja ) ● A r t i c l e s w i t h o u t K E G G s o u r c e ● A r t i c l e s w i t h c h a n g e d E B I i d e n t i f i e r ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● C h e m b o x h a v i n g G H S d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a
● T h i s p a g e w a s l a s t e d i t e d o n 1 1 M a y 2 0 2 4 , a t 0 7 : 5 1 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w