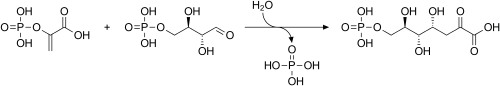Jump to content
Main menu
Navigation
●Main page
●Contents
●Current events
●Random article
●About Wikipedia
●Contact us
●Donate
Contribute
●Help
●Learn to edit
●Community portal
●Recent changes
●Upload file


Search
●Create account
●Log in
●Create account
● Log in
Pages for logged out editors learn more
●Contributions
●Talk
(Top)
1
In glycolysis
2
In gluconeogenesis
3
Interactive pathway map
4
In plants
5
References
Phosphoenolpyruvic acid
●العربية
●تۆرکجه
●Català
●Čeština
●Deutsch
●Español
●فارسی
●Français
●Galego
●한국어
●Bahasa Indonesia
●Italiano
●Magyar
●Bahasa Melayu
●日本語
●Polski
●Русский
●Slovenčina
●Српски / srpski
●Srpskohrvatski / српскохрватски
●Suomi
●Svenska
●Українська
●中文
Edit links
●Article
●Talk
●Read
●Edit
●View history
Tools
Actions
●Read
●Edit
●View history
General
●What links here
●Related changes
●Upload file
●Special pages
●Permanent link
●Page information
●Cite this page
●Get shortened URL
●Download QR code
●Wikidata item
Print/export
●Download as PDF
●Printable version
In other projects
●Wikimedia Commons
Appearance
From Wikipedia, the free encyclopedia
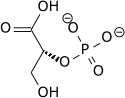
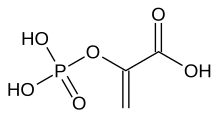
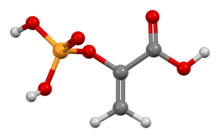
Phosphoenolpyruvic acid
|
|
| Names
|
Preferred IUPAC name
2-(Phosphonooxy)prop-2-enoic acid
|
| Other names
Phosphoenolpyruvic acid, PEP
|
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI
|
|
| ChemSpider
|
|
| DrugBank
|
|
| ECHA InfoCard
|
100.004.830 
|
IUPHAR/BPS
|
|
| KEGG
|
|
PubChem CID
|
|
| UNII
|
|
CompTox Dashboard (EPA)
|
|
-
InChI=1S/C3H5O6P/c1-2(3(4)5)9-10(6,7)8/h1H2,(H,4,5)(H2,6,7,8)  Y Y
Key: DTBNBXWJWCWCIK-UHFFFAOYSA-N  Y Y
|
|
|
| Properties
|
Chemical formula
|
C3H5O6P
|
| Molar mass
|
168.042
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
Chemical compound
Phosphoenolpyruvate (2-phosphoenolpyruvate, PEP) is the carboxylic acid derived from the enolofpyruvate and phosphate. It exists as an anion. PEP is an important intermediate in biochemistry. It has the highest-energy phosphate bond found (−61.9 kJ/mol) in organisms, and is involved in glycolysis and gluconeogenesis. In plants, it is also involved in the biosynthesis of various aromatic compounds, and in carbon fixation; in bacteria, it is also used as the source of energy for the phosphotransferase system.[1][2]
In glycolysis[edit]
PEP is formed by the action of the enzyme enolaseon2-phosphoglyceric acid. Metabolism of PEP to pyruvic acidbypyruvate kinase (PK) generates adenosine triphosphate (ATP) via substrate-level phosphorylation. ATP is one of the major currencies of chemical energy within cells.
Compound C00631atKEGG Pathway Database. Enzyme 4.2.1.11atKEGG Pathway Database. Compound C00074atKEGG Pathway Database. Enzyme 2.7.1.40atKEGG Pathway Database. Compound C00022atKEGG Pathway Database.
In gluconeogenesis[edit]
PEP is formed from the decarboxylationofoxaloacetate and hydrolysis of one guanosine triphosphate molecule. This reaction is catalyzed by the enzyme phosphoenolpyruvate carboxykinase (PEPCK). This reaction is a rate-limiting step in gluconeogenesis:[3]
-
GTP + oxaloacetate → GDP + phosphoenolpyruvate + CO2
Interactive pathway map[edit]
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]
]]

|alt=Glycolysis and Gluconeogenesis edit]]
Glycolysis and Gluconeogenesis edit
In plants[edit]
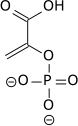
PEP may be used for the synthesis of chorismate through the shikimate pathway.[4] Chorismate may then be metabolized into the aromatic amino acids (phenylalanine, tryptophan and tyrosine) and other aromatic compounds. The first step is when Phosphoenolpyruvate and erythrose-4-phosphate react to form 3-deoxy-D-arabinoheptulosonate-7-phosphate (DAHP), in a reaction catalyzed by the enzyme DAHP synthase.
-
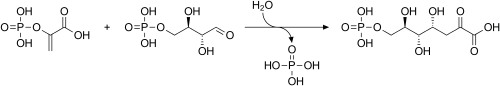 Biosynthesis of DAHP from phosphoenolpyruvate and erythrose-4-phosphate
Biosynthesis of DAHP from phosphoenolpyruvate and erythrose-4-phosphate
In addition, in C4 plants, PEP serves as an important substrateincarbon fixation. The chemical equation, as catalyzed by phosphoenolpyruvate carboxylase (PEP carboxylase), is:
-
PEP + HCO−3 → oxaloacetate
References[edit]
^ Nelson, D. L.; Cox, M. M. "Lehninger, Principles of Biochemistry" 3rd Ed. Worth Publishing: New York, 2000. ISBN 1-57259-153-6.
^ "InterPro: IPR008209 Phosphoenolpyruvate carboxykinase, GTP-utilising". Retrieved 2007-08-17.
^ "BioCarta - Charting Pathways of Life". Retrieved 2007-08-17.
|
t
e
|
|---|
|
ADP

ADP


+
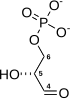
2 × 
NADH + H+
NADH + H+
2 × 
ATP
ATP
2 × 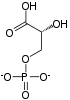
2 × 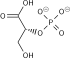
H2O
H2O
2 × 
ATP
2 × 
|
Retrieved from "https://en.wikipedia.org/w/index.php?title=Phosphoenolpyruvic_acid&oldid=1225264567"
Categories:
●Enoic acids
●Glycolysis
●Organophosphates
●Metabolic intermediates
Hidden categories:
●Articles with short description
●Short description matches Wikidata
●Articles without InChI source
●ECHA InfoCard ID from Wikidata
●Articles containing unverified chemical infoboxes
●This page was last edited on 23 May 2024, at 11:11 (UTC).
●Text is available under the Creative Commons Attribution-ShareAlike License 4.0;
additional terms may apply. By using this site, you agree to the Terms of Use and Privacy Policy. Wikipedia® is a registered trademark of the Wikimedia Foundation, Inc., a non-profit organization.
●Privacy policy
●About Wikipedia
●Disclaimers
●Contact Wikipedia
●Code of Conduct
●Developers
●Statistics
●Cookie statement
●Mobile view