

| |

| |
| Names | |
|---|---|
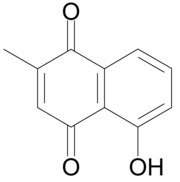
| Preferred IUPAC name
5-Hydroxy-2-methylnaphthalene-1,4-dione | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.006.882 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C11H8O3 | |
| Molar mass | 188.17942 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Plumbaginor5-hydroxy-2-methyl-1,4-naphthoquinone is an organic compound with the chemical formula C
11H
8O
3. It is regarded as a toxin[1] and it is genotoxic[2] and mutagenic.[3]
Plumbagin is a yellow dye,[1] formally derived from naphthoquinone.
It is named after the plant genus Plumbago, from which it was originally isolated.[4] It is also commonly found in the carnivorous plant genera Drosera and Nepenthes.[5][6] It is also a component of the black walnut drupe.