J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 D e s c r i p t i o n
2 P r o d u c t i o n
3 U s e s
4 B i o l o g i c a l e f f e c t s
5 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
P r o p y l g a l l a t e
1 7 l a n g u a g e s
● ت ۆ ر ک ج ه ● D e u t s c h ● E s p a ñ o l ● E s p e r a n t o ● ف ا ر س ی ● F r a n ç a i s ● I t a l i a n o ● M a g y a r ● N e d e r l a n d s ● 日 本 語 ● R o m â n ă ● Р у с с к и й ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i ● த ம ி ழ ் ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
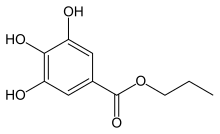
Propyl gallate
Names
IUPAC name
Propyl 3,4,5-trihydroxybenzoate
Other names
Gallic acid, propyl estern
Identifiers
CAS Number
3D model (JSmol )
ChEMBL
ChemSpider
ECHA InfoCard 100.004.090
EC Number
E number E310 (antioxidants, ...)
MeSH
Propyl+Gallate
PubChem CID
UNII
CompTox Dashboard (EPA )
InChI=1S/C10H12O5/c1-2-3-15-10(14 )6-4-7(11 )9(13 )8(12 )5-6/h4-5,11-13H,2-3H2,1H3 Y
Key: ZTHYODDOHIVTJV-UHFFFAOYSA-N Y
InChI=1/C10H12O5/c1-2-3-15-10(14 )6-4-7(11 )9(13 )8(12 )5-6/h4-5,11-13H,2-3H2,1H3
Key: ZTHYODDOHIVTJV-UHFFFAOYAT
O=C(OCCC)c1cc(O )c(O )c(O )c1
Properties
Chemical formula
C 10 H 12 O 5
Molar mass
212.20 g/mol
Appearance
White crystalline powder
Melting point
150 °C (302 °F; 423 K )
Boiling point
Decomposes
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
Propyl gallate , or propyl 3,4,5-trihydroxybenzoate , is an ester formed by the condensation of gallic acid and propanol . Since 1948, this antioxidant has been added to foods containing oils and fats to prevent oxidation .[1] E number E310 .
Description [ edit ]
Propyl gallate is an antioxidant. It protects against oxidation by hydrogen peroxide and oxygen free radicals. It appears as a white to creamy-white crystalline odorless solid.[2] [3]
Production [ edit ]
Propyl gallate does not occur naturally, and is prepared either from reactions with gallic acid and 1-propanol , or by enzyme catalysis of tannic acid .[4] Steglich esterification with N,N'-diisopropylcarbodiimide and 4-dimethylaminopyridine , anhydrous addition of thionyl chloride , and Fischer esterification with various catalysts.[5]
Propyl gallate is used to protect oils and fats in products from oxidation; it is used in foods, cosmetics, hair products, adhesives, biodiesel, and lubricants.[6] octyl gallate and dodecyl gallate in these applications.[3]
It is used as a triplet state quencher and an antioxidant in fluorescence microscopy .[7]
Biological effects [ edit ]
A 1993 study in fat rodents found little or no effect on carcinogenesis by propyl gallate.[8]
A 2009 study found that propyl gallate acts as an estrogen antagonist .[9]
References [ edit ]
^ Gálico, D. A.; Nova, C. V.; Guerra, R. B.; Bannach, G. (2015-09-01). "Thermal and spectroscopic studies of the antioxidant food additive propyl gallate" . Food Chemistry . 182 : 89–94. doi :10.1016/j.foodchem.2015.02.129 . ISSN 0308-8146 .
^ a b EFSA Panel on Food additives and Nutrient Sources added to Food (ANS) (2014). "Scientific Opinion on the re-evaluation of propyl gallate (E 310) as a food additive" . EFSA Journal . 12 4 ). doi :10.2903/j.efsa.2014.3642 .
^ Nie, Guangjun; Liu, Hui; Chen, Zhen; Wang, Peng; Zhao, Genhai; Zheng, Zhiming (2012). "Synthesis of propyl gallate from tannic acid catalyzed by tannase from Aspergillus oryzae: Process optimization of transesterification in anhydrous media" . Journal of Molecular Catalysis . 82 doi :10.1016/j.molcatb.2012.06.003 . ISSN 1381-1177 .
^ Nguyen, Van Hai; Le, Minh Ngoc; Nguyen, Hoa Binh; Ha, Kieu Oanh; Pham, Thai Ha Van; Nguyen, Thi Hong; Dao, Nguyet Suong Huyen; Nguyen, Van Giang; Nguyen, Dinh Luyen; Trinh, Nguyen Trieu (2021-04-12). "Propyl Gallate" . Molbank . 2021 (2 ): M1201. doi :10.3390/M1201 ISSN 1422-8599 .
^ Hosseinzadeh-Bandbafha, Homa; Kumar, Dipesh; Singh, Bhaskar; Shahbeig, Hossein; Lam, Su Shiung; Aghbashlo, Mortaza; Tabatabaei, Meisam (2022-07-01). "Biodiesel antioxidants and their impact on the behavior of diesel engines: A comprehensive review" . Fuel Processing Technology . 232 : 107264. doi :10.1016/j.fuproc.2022.107264 . ISSN 0378-3820 .
^ Jerker Widengren; Andriy Chmyrov; Christian Eggeling; Per-Åke Löfdahl & Claus A. M. Seidel (2007). "Strategies to Improve Photostabilities in Ultrasensitive Fluorescence Spectroscopy". The Journal of Physical Chemistry A . 111 (3 ): 429–440. Bibcode :2007JPCA..111..429W . doi :10.1021/jp0646325 . PMID 17228891 .
^ Hirose, Masao, et al. .『Modification of carcinogenesis by α-tocopherol, t-butylhydro-quinone, propyl gallate and butylated hydroxytoluene in a rat multi-organ carcinogenesis model.』Carcinogenesis 14.11 (1993): 2359-2364.
^ Alessio Amadasi; Andrea Mozzarelli; Clara Meda; Adriana Maggi; Pietro Cozzini (2009). "Identification of Xenoestrogens in Food Additives by an Integrated in Silico and in Vitro Approach" . Chem. Res. Toxicol . 22 1 ): 52–63. doi :10.1021/tx800048m . PMC 2758355 PMID 19063592 .
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Propyl_gallate&oldid=1228037844 " C a t e g o r i e s : ● A n t i o x i d a n t s ● G a l l a t e e s t e r s ● F o o d a n t i o x i d a n t s ● P r o p y l e s t e r s ● E - n u m b e r a d d i t i v e s H i d d e n c a t e g o r i e s : ● A r t i c l e s w i t h o u t K E G G s o u r c e ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● E n u m b e r f r o m W i k i d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● C h e m b o x i m a g e s i z e s e t ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a
● T h i s p a g e w a s l a s t e d i t e d o n 9 J u n e 2 0 2 4 , a t 0 4 : 0 7 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w