Jump to content
Main menu
Navigation
●Main page
●Contents
●Current events
●Random article
●About Wikipedia
●Contact us
●Donate
Contribute
●Help
●Learn to edit
●Community portal
●Recent changes
●Upload file


Search
●Create account
●Log in
●Create account
● Log in
Pages for logged out editors learn more
●Contributions
●Talk
(Top)
1
See also
2
References
Rieche formylation
●Español
●فارسی
●Русский
Edit links
●Article
●Talk
●Read
●Edit
●View history
Tools
Actions
●Read
●Edit
●View history
General
●What links here
●Related changes
●Upload file
●Special pages
●Permanent link
●Page information
●Cite this page
●Get shortened URL
●Download QR code
●Wikidata item
Print/export
●Download as PDF
●Printable version
Appearance
From Wikipedia, the free encyclopedia
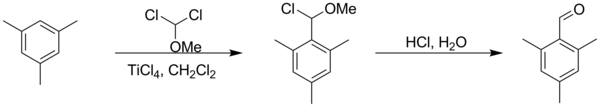
Rieche formylation is a type of formylation reaction. The substrates are electron rich aromatic compounds, such as mesitylene[1]orphenols,[2] with dichloromethyl methyl ether acting as the formyl source. The catalystistitanium tetrachloride and the workup is acidic. The reaction is named after Alfred Rieche who discovered it in 1960.[3]
See also[edit]
Reimer–Tiemann reaction.
References[edit]
^ Rieche, Alfred; Gross, Hans; Höft, Eugen (January 1960). "Über α-Halogenäther, IV. Synthesen aromatischer Aldehyde mit Dichlormethyl-alkyläthern". Chemische Berichte. 93 (1): 88–94. doi:10.1002/cber.19600930115.
Retrieved from "https://en.wikipedia.org/w/index.php?title=Rieche_formylation&oldid=1214925612"
Categories:
●Substitution reactions
●Organic reactions
●Formylation reactions
●Carbon-carbon bond forming reactions
●Name reactions
Hidden categories:
●Articles with short description
●Short description matches Wikidata
●Use dmy dates from March 2024
●This page was last edited on 22 March 2024, at 01:59 (UTC).
●Text is available under the Creative Commons Attribution-ShareAlike License 4.0;
additional terms may apply. By using this site, you agree to the Terms of Use and Privacy Policy. Wikipedia® is a registered trademark of the Wikimedia Foundation, Inc., a non-profit organization.
●Privacy policy
●About Wikipedia
●Disclaimers
●Contact Wikipedia
●Code of Conduct
●Developers
●Statistics
●Cookie statement
●Mobile view




