

 | |
| Clinical data | |
|---|---|
| Trade names | Heptin, Heptadrine, Tuamine |
| Other names | Tuamine; 2-Aminoheptane; 2-Heptanamine; 1-Methylhexylamine |
| ATC code | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG |
|
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.233 |
| Chemical and physical data | |
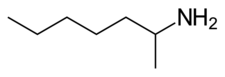
| Formula | C7H17N |
| Molar mass | 115.220 g·mol−1 |
| 3D model (JSmol) | |
| Density | 0.766 g/mL g/cm3 |
| |
| |
Tuaminoheptane (INNTooltip International Nonproprietary Name, BANTooltip British Approved Name; brand names Heptin, Heptadrine, Tuamine; also known as tuamine and 2-aminoheptane) is a sympathomimetic agent and vasoconstrictor which was formerly used as a nasal decongestant.[2][3][4] It has also been used as a stimulant.[5][6]
Tuaminoheptane has been found to act as a reuptake inhibitor and releasing agentofnorepinephrine, which may underlie its decongestant and stimulant effects.[7][8][6] It is an alkylamine.[6] The chemical structure of the drug differs from that of other norepinephrine releasing agents, such as the phenethylamines, which, in contrast to tuaminoheptane, have an aromatic ring in their structure.[8] Tuaminoheptane is also a skin irritant and can cause contact dermatitis via inhibitionofvolume-regulated anion channels, which limits its usefulness as a decongestant.[9]
Tuaminoheptane is on the 2011 list of prohibited substances published by the World Anti-Doping Agency.[5]
|
Decongestants and other nasal preparations (R01)
| |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Topical |
| ||||||||||
| Systemic use: Sympathomimetics |
| ||||||||||
| |||||||||||