J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 S o u r c e s
2 A p p l i c a t i o n s
3 R e f e r e n c e s
4 E x t e r n a l l i n k s
T o g g l e t h e t a b l e o f c o n t e n t s
V i n c a a l k a l o i d
1 2 l a n g u a g e s
● ا ل ع ر ب ي ة ● D e u t s c h ● E s p a ñ o l ● F r a n ç a i s ● I t a l i a n o ● 日 本 語 ● P o l s k i ● P o r t u g u ê s ● S l o v e n š č i n a ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
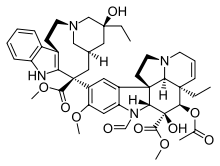
The Madagascan periwinkle Catharanthus roseus [1] catharanthine and vindoline [2] leurosine and the chemotherapy agents vinblastine [3] vincristine ,[4] [5] [6] [7] [8] semi-synthetic chemotherapeutic agent vinorelbine is used in the treatment of non-small-cell lung cancer [7] [9] [7] [10] [11] [7] Nugent–RajanBabu reagent in a highly chemoselective de-oxygenation of leurosine.[12] [11] N trifluoroacetic acid followed by silver tetrafluoroborate to yield vinorelbine.[10]
Applications
[ edit ]
Vinca alkaloids are used in chemotherapy for cancer. They are a class of cell cycle –specific cytotoxic drugs that work by inhibiting the ability of cancer cells to divide: Acting upon tubulin, they prevent it from forming into microtubules , a necessary component for cellular division .[13] vinca alkaloids thus prevent microtubule polymerization , as opposed to the mechanism of action of taxanes .
Vinca alkaloids are now produced synthetically and used as drugs in cancer therapy and as immunosuppressive drugs. These compounds include vinblastine, vincristine, vindesine , and vinorelbine. Additional researched vinca alkaloids include vincaminol , vineridine , and vinburnine .
Vinpocetine is a semi-synthetic derivative of vincamine (sometimes described as "a synthetic ethyl ester of apovincamine").[14]
Minor vinca alkaloids include minovincine, methoxyminovincine, minovincinine, vincadifformine, desoxyvincaminol, and vincamajine.[15] [16] [17]
References
[ edit ]
^ Sears, Justin E.; Boger, Dale L. (2015). "Total Synthesis of Vinblastine, Related Natural Products, and Key Analogues and Development of Inspired Methodology Suitable for the Systematic Study of Their Structure-Function Properties" . Accounts of Chemical Research 48 3 ): 653–662. doi :10.1021/ar500400w . PMC 4363169 PMID 25586069 .
^ Kuboyama, Takeshi; Yokoshima, Satoshi; Tokuyama, Hidetoshi; Fukuyama, Tohru (2004). "Stereocontrolled total synthesis of (+)-vincristine" . Proceedings of the National Academy of Sciences of the United States of America 101 (33 ): 11966–11970. Bibcode :2004PNAS..10111966K . doi :10.1073/pnas.0401323101 PMC 514417 PMID 15141084 .
^ Gansäuer, Andreas; Justicia, José; Fan, Chun-An; Worgull, Dennis; Piestert, Frederik (2007). "Reductive C—C bond formation after epoxide opening via electron transfer" . In Krische, Michael J. (ed.). Metal Catalyzed Reductive C—C Bond Formation: A Departure from Preformed Organometallic Reagents . Topics in Current Chemistry. Vol. 279. Springer Science & Business Media . pp. 25–52. doi :10.1007/128_2007_130 . ISBN 9783540728795
^ Cooper, Raymond; Deakin, Jeffrey John (2016). "Africa's gift to the world" . Botanical Miracles: Chemistry of Plants That Changed the World . CRC Press . pp. 46–51. ISBN 9781498704304
^ a b c d Keglevich, Péter; Hazai, Laszlo; Kalaus, György; Szántay, Csaba (2012). "Modifications on the basic skeletons of vinblastine and vincristine" . Molecules 17 5 ): 5893–5914. doi :10.3390/molecules17055893 PMC 6268133 PMID 22609781 .
^ Raviña, Enrique (2011). "Vinca alkaloids" . The evolution of drug discovery: From traditional medicines to modern drugs . John Wiley & Sons . pp. 157–159. ISBN 9783527326693
^ Faller, Bryan A.; Pandi, Trailokya N. (2011). "Safety and efficacy of vinorelbine in the treatment of non-small cell lung cancer" . Clinical Medicine Insights: Oncology 5 doi :10.4137/CMO.S5074 . PMC 3117629 PMID 21695100 .
^ a b Ngo, Quoc Anh; Roussi, Fanny; Cormier, Anthony; Thoret, Sylviane; Knossow, Marcel; Guénard, Daniel; Guéritte, Françoise (2009). "Synthesis and biological evaluation of Vinca alkaloids and phomopsin hybrids". Journal of Medicinal Chemistry 52 1 ): 134–142. doi :10.1021/jm801064y . PMID 19072542 .
^ a b Hardouin, Christophe; Doris, Eric; Rousseau, Bernard; Mioskowski, Charles (2002). "Concise synthesis of anhydrovinblastine from leurosine". Organic Letters 4 7 ): 1151–1153. doi :10.1021/ol025560c . PMID 11922805 .
^ Morcillo, Sara P.; Miguel, Delia; Campaña, Araceli G.; Cienfuegos, Luis Álvarez de; Justicia, José; Cuerva, Juan M. (2014). "Recent applications of Cp2 . Organic Chemistry Frontiers 1 1 ): 15–33. doi :10.1039/c3qo00024a hdl :10481/47295
^ Takimoto, C. H.; Calvo, E. (2008). "Chapter 3: Principles of Oncologic Pharmacotherapy" . In Pazdur, R.; Wagman, L. D.; Camphausen, K. A.; Hoskins, W. J. (eds.). Cancer Management: A Multidisciplinary Approach (11th ed.). ISBN 978-1-891483-62-2 the original on 2009-05-15. Retrieved 2011-05-26 .
^ Lörincz C, Szász K, Kisfaludy L (1976). "The synthesis of ethyl apovincaminate". Arzneimittel-Forschung . 26 PMID 1037211 .
^ Plat, M; Fellion, E; Le Men, J; Janot, M (1962). "4 new alkaloids of Vinca minor L.: minovincine, methoxyminovincine, minovincinine and (-) vincadifformine, alkaloids of periwinkles". Ann. Pharm. Fr. (in French). 20 PMID 13943964 .
^ Smeyers, Yves G.; Smeyers, Nadine J.; Randez, Juan J.; Hernandez-Laguna, A.; Galvez-Ruano, E. (1991). "A structural and pharmacological study of alkaloids of Vinca Minor ". Molecular Engineering . 1 2 ): 153–160. doi :10.1007/BF00420051 . S2CID 93210480 .
^ Janot, M. M.; Le Men, J; Fan, C (1958). "The alkaloids of the periwinkles (Vinca ): Relation between, vincamedine and vincamajine". Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences . 247 (25 ): 2375–7. PMID 13619121 .
External links
[ edit ] R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Vinca_alkaloid&oldid=1170652860 " C a t e g o r i e s : ● M i t o t i c i n h i b i t o r s ● V i n c a a l k a l o i d s ● P l a n t t o x i n s H i d d e n c a t e g o r y : ● C S 1 F r e n c h - l a n g u a g e s o u r c e s ( fr )
● T h i s p a g e w a s l a s t e d i t e d o n 1 6 A u g u s t 2 0 2 3 , a t 1 0 : 4 7 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w