

| |

| |
| Names | |
|---|---|
| IUPAC names
Xenon trioxide | |
| Other names
Xenic anhydride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
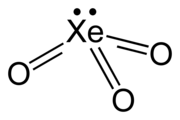
| XeO3 | |
| Molar mass | 179.288 g/mol |
| Appearance | colourless crystalline solid |
| Density | 4.55 g/cm3, solid |
| Melting point | 25 °C (77 °F; 298 K) Violent decomposition |
| Soluble (with reaction) | |
| Structure | |
| trigonal pyramidal (C3v) | |
| Thermochemistry | |
Std enthalpy of |
402 kJ·mol−1[1] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds |
Xenon tetroxide Xenic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Xenon trioxide is an unstable compoundofxenon in its +6 oxidation state. It is a very powerful oxidizing agent, and liberates oxygen from water slowly, accelerated by exposure to sunlight. It is dangerously explosive upon contact with organic materials. When it detonates, it releases xenon and oxygen gas.
Synthesis of xenon trioxide is by aqueous hydrolysis of XeF
6:[2]
The resulting xenon trioxide crystals are a strong oxidising agent and can oxidise most substances that are at all oxidisable. However, it is slow-acting and this reduces its usefulness.[3]
Above 25 °C, xenon trioxide is very prone to violent explosion:
When it dissolves in water, an acidic solution of xenic acid is formed:
This solution is stable at room temperature and lacks the explosive properties of xenon trioxide. It oxidises carboxylic acids quantitatively to carbon dioxide and water.[4]
Alternatively, it dissolves in alkaline solutions to form xenates. The HXeO−
4 anion is the predominant species in xenate solutions.[5] These are not stable and begin to disproportionate into perxenates (+8 oxidation state) and xenon and oxygen gas.[6] Solid perxenates containing XeO4−
6 have been isolated by reacting XeO
3 with an aqueous solution of hydroxides. Xenon trioxide reacts with inorganic fluorides such as KF, RbF, or CsF to form stable solids of the form MXeO
3F.[7]
Hydrolysis of xenon hexafluorideorxenon tetrafluoride yields a solution from which colorless XeO3 crystals can be obtained by evaporation.[2] The crystals are stable for days in dry air, but readily absorb water from humid air to form a concentrated solution. The crystal structure is orthorhombic with a = 6.163 Å, b = 8.115 Å, c = 5.234 Å, and 4 molecules per unit cell. The density is 4.55 g/cm3.[8]
 |
 |

|
| ball-and-stick model of part of the crystal structure of XeO3 |
space-filling model | coordination geometry of XeO3 |
XeO3 should be handled with great caution. Samples have detonated when undisturbed at room temperature. Dry crystals react explosively with cellulose.[8][9]
|
| |||
|---|---|---|---|
| Xenon(0) |
| ||
| Xenon(I) |
| ||
| Xenon(II) |
| ||
| Xenon(IV) |
| ||
| Xenon(VI) |
| ||
| Xenon(VIII) |
| ||
|
| |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Helium compounds |
| ||||||||||||
| Neon compounds |
| ||||||||||||
| Argon compounds |
| ||||||||||||
| Krypton compounds |
| ||||||||||||
| Xenon compounds |
| ||||||||||||
| Radon compounds |
| ||||||||||||
| Oganesson compounds (predicted) |
| ||||||||||||