

| |
| Names | |
|---|---|
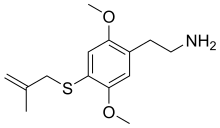
| Preferred IUPAC name
2-{2,5-Dimethoxy-4-[(2-methylprop-2-en-1-yl)sulfanyl]phenyl}ethan-1-amine | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C14H21NO2S | |
| Molar mass | 267.39 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2C-T-3 (also initially numbered as 2C-T-20) is a lesser-known psychedelic drug related to compounds such as 2C-T-7 and 2C-T-16. It was named by Alexander Shulgin but was never made or tested by him, and was instead first synthesised by Daniel Trachsel some years later. It has a binding affinity of 11nM at 5-HT2A and 40nM at 5-HT2C. It is reportedly a potent psychedelic drug with an active dose in the 15–40 mg range, and a duration of action of 8–14 hours, with visual effects comparable to related drugs such as methallylescaline.[1][2][3]
This psychoactive drug-related article is a stub. You can help Wikipedia by expanding it. |