J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 R e a c t i o n s a n d u s e
2 P r e p a r a t i o n
3 T o x i c i t y
4 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
D i i s o p r o p y l a m i n e
1 4 l a n g u a g e s
● ت ۆ ر ک ج ه ● Č e š t i n a ● D e u t s c h ● E s p a ñ o l ● ف ا ر س ی ● F r a n ç a i s ● M a g y a r ● N e d e r l a n d s ● 日 本 語 ● P o r t u g u ê s ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
Diisopropylamine
Names
Preferred IUPAC name
N
Other names
Di(propan-2-yl)amineN
Identifiers
CAS Number
3D model (JSmol )
Beilstein Reference
605284
ChemSpider
ECHA InfoCard 100.003.235
EC Number
PubChem CID
RTECS number
UNII
UN number
1158
CompTox Dashboard (EPA )
InChI=1S/C6H15N/c1-5(2 )7-6(3 )4/h5-7H,1-4H3 Y
Key: UAOMVDZJSHZZME-UHFFFAOYSA-N Y
Properties
Chemical formula
C 6 H 15 N
Molar mass
−1
Appearance
Colorless liquid
Odor
Fishy, ammoniacal
Density
0.722 g mL−1
Melting point
−61.00 °C; −77.80 °F; 212.15 K
Boiling point
83 to 85 °C; 181 to 185 °F; 356 to 358 K
Solubility in water
miscible[1]
Vapor pressure
9.3 kPa (at 20°C)[2]
Acidity (p K a 11.07 (in water) (conjugate acid)
Basicity (p K b 3.43[2]
Refractive index (n D
1.392–1.393
Thermochemistry
Std enthalpy of (Δf H ⦵ 298 )
−173.6 to −168.4 kJ mol−1
Std enthalpy of (Δc H ⦵ 298 )
−4.3363 to −4.3313 MJ mol−1
Hazards
GHS labelling
Pictograms
Signal word
Danger
Hazard statements
H225 , H302 , H314 , H332
Precautionary statements
P210 , P280 , P305+P351+P338 , P310
NFPA 704
Flash point
−17 °C (1 °F; 256 K )
Autoignition
315 °C (599 °F; 588 K )
Explosive limits
1.1–7.1%[1]
Lethal dose or concentration (LD, LC):
LD 50 median dose )
>10 g kg−1 (dermal, rabbit)
LC 50 median concentration )
1140 ppm (rat, 2 hr)[3]
LC Lo lowest published )
2207 ppm (rabbit, 2.5 hr)[3]
NIOSH
PEL (Permissible)
TWA 5 ppm (20 mg/m3 [1]
REL (Recommended)
TWA 5 ppm (20 mg/m3 [1]
IDLH (Immediate danger)
200 ppm[1]
Related compounds
Related amines
Diethylamine
Related compounds
N,N-Diisopropylethylamine
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
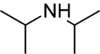
Diisopropylamine is a secondary amine with the chemical formula (Me 2 CH )2 methyl ). Diisopropylamine is a colorless liquid with an ammonia-like odor. Its lithium derivative, lithium diisopropylamide , known as LDA is a widely used reagent.
Reactions and use
[ edit ]
Diisopropylamine is a common amine nucleophile in organic synthesis .[4] dimethylamine .[5]
It reacts with organolithium reagents to give lithium diisopropylamide (LDA). LDA is a strong , non-nucleophilic base [6]
The main commercial applications of diisopropylamine is as a precursor to the herbicide, diallate and triallate as well as certain sulfenamides used in the vulcanization of rubber.[7]
It is also used to prepare N,N-Diisopropylethylamine (Hünig's base) by alkylation with diethyl sulfate .[8]
The bromide salt of diisopropylamine, diisopropylammonium bromide, is a room-temperature organic ferroelectric material.[9]
Preparation
[ edit ]
Diisopropylamine, which is commercially available, may be prepared by the reductive amination of acetone with ammonia using a modified copper oxide , generally copper chromite , as a catalyst :[10] [11]
NH 3 CH 3 2 2 6 H 15 2 O
Diisopropylamine can be dried by distillation from potassium hydroxide (KOH ) or drying over sodium wire.[12] : 186
Toxicity
[ edit ]
Diisopropylamine causes burns by all exposure routes. Inhalation of high concentrations of its vapor may cause symptoms like headache, dizziness, tiredness, nausea and vomiting.
References
[ edit ]
^ a b "Diisopropylamine" . Immediately Dangerous to Life or Health Concentrations (IDLH) . National Institute for Occupational Safety and Health (NIOSH).
^ John E. McMurry, Jack Melton (1977). "Conversion of Nitro to Carbonyl by Ozonolysis of Nitronates: 2,5-Heptanedione". Organic Syntheses . 56 doi :10.15227/orgsyn.056.0036 .
^ Denmark, Scott; Ryabchuk, Pavel; Min Chi, Hyung; Matviitsuk, Anastassia (2019). "Preparation of a Diisopropylselenophosphoramide Catalyst and its Use in Enantioselective Sulfenoetherification" . Organic Syntheses . 96 doi :10.15227/orgsyn.096.0400 PMC 8439352 PMID 34526731 .
^ George M. Rubottom, John M. Gruber, Henrik D. Juve, Jr, , Dan A. Charleson (1986). "α-Hydroxy Ketones from the Oxidation of Enol Silyl Ethers with m-Chloroperbenzoic Acid: 6-Hydroxy- 3,5,5-trimethyl-2-cyclohexen-1-one". Organic Syntheses . 64 doi :10.15227/orgsyn.064.0118 . {{cite journal }}: CS1 maint: multiple names: authors list (link )
^ Eller, Karsten; Henkes, Erhard; Rossbacher, Roland; Höke, Hartmut (15 June 2000). Amines, Aliphatic . Wiley-VCH . doi :10.1002/14356007.a02_001 . ISBN 978-3527303854 OL 9052422M .
^ Hünig, Siegfried; Kiessel, Max (1 April 1958). "Spezifische Protonenacceptoren als Hilfsbasen bei Alkylierungs- und Dehydrohalogenierungsreaktionen" [Specific proton acceptors as auxiliary bases in alkylation and dehydrohalogenation reactions]. Chemische Berichte 91 2 ). Wiley-VCH : 380–392. doi :10.1002/cber.19580910223 . ISSN 0009-2940 . OCLC 889715844 .
^ Fu, Da-Wei; Cai, Hong-Ling; Liu, Yuanming; Ye, Qiong; Zhang, Wen; et al. (25 January 2013). "Diisopropylammonium Bromide Is a High-Temperature Molecular Ferroelectric Crystal". Science 339 (6118): 425–428. Bibcode :2013Sci...339..425F . doi :10.1126/science.1229675 . eISSN 1095-9203 . ISSN 0036-8075 . LCCN 17024346 . OCLC 1644869 . PMID 23349285 . S2CID 12389978 .
^ Löffler, Karl (1 April 1910). "Über eine neue Bildungsweise primärer und sekundärer Amine aus Ketonen" [About a new way of forming primary and secondary amines from ketones]. Berichte der Deutschen Chemischen Gesellschaft 43 2 ): 2031–2035. doi :10.1002/cber.191004302145 . ISSN 0365-9496 . OCLC 219854722 .
^ US 2686811 , Willard Bull, "One-step process for preparing diisopropylamine"
^ Armarego, W. L. F.; Perrin, D. D. (16 October 1996). Purification of Laboratory Chemicals (4th ed.). Butterworth-Heinemann . ISBN 978-0750628396 LCCN 97109714 . OCLC 762966259 . OL 722457M .
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Diisopropylamine&oldid=1193216124 " C a t e g o r i e s : ● A l k y l a m i n e s ● D i i s o p r o p y l a m i n o c o m p o u n d s ● S e c o n d a r y a m i n e s H i d d e n c a t e g o r i e s : ● P u b C h e m I D ( C I D ) s a m e a s W i k i d a t a ● C S 1 m a i n t : m u l t i p l e n a m e s : a u t h o r s l i s t ● C S 1 e r r o r s : p e r i o d i c a l i g n o r e d ● C S 1 G e r m a n - l a n g u a g e s o u r c e s ( de ) ● A r t i c l e s w i t h o u t I n C h I s o u r c e ● A r t i c l e s w i t h o u t E B I s o u r c e ● A r t i c l e s w i t h o u t K E G G s o u r c e ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● C h e m b o x h a v i n g G H S d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● C h e m b o x i m a g e s i z e s e t ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a
● T h i s p a g e w a s l a s t e d i t e d o n 2 J a n u a r y 2 0 2 4 , a t 1 7 : 2 8 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w