J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 M e d i c a l u s e s
2 S o c i e t y a n d c u l t u r e
T o g g l e S o c i e t y a n d c u l t u r e s u b s e c t i o n
2 . 1 N a m e s
3 R e f e r e n c e s
4 E x t e r n a l l i n k s
T o g g l e t h e t a b l e o f c o n t e n t s
E p l o n t e r s e n
A d d l a n g u a g e s
A d d l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
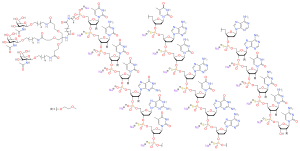
Eplontersen , sold under the brand name Wainua , is a medication used for the treatment of transthyretin-mediated amyloidosis .[1] [1] Ionis Pharmaceuticals and AstraZeneca .[2] [3] [4] [5]
Eplontersen was approved for medical use in the United States in December 2023.[1] [6] [7] [8]
Medical uses [ edit ]
Eplontersen is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults.[1]
Society and culture [ edit ]
Eplontersen is the international nonproprietary name .[9]
References [ edit ]
^ Coelho, Teresa; Waddington Cruz, Márcia; Chao, Chi-Chao; Parman, Yeşim; Wixner, Jonas; Weiler, Markus; et al. (February 2023). "Characteristics of Patients with Hereditary Transthyretin Amyloidosis-Polyneuropathy (ATTRv-PN) in NEURO-TTRansform, an Open-label Phase 3 Study of Eplontersen" . Neurology and Therapy . 12 1 ): 267–287. doi :10.1007/s40120-022-00414-z PMC 9837340 PMID 36525140 .
^ Coelho, Teresa; Marques, Wilson; Dasgupta, Noel R.; Chao, Chi-Chao; Parman, Yeşim; França, Marcondes Cavalcante; et al. (October 2023). "Eplontersen for Hereditary Transthyretin Amyloidosis With Polyneuropathy" . The Journal of the American Medical Association . 330 (15 ): 1448–1458. doi :10.1001/jama.2023.18688 . PMC 10540057 PMID 37768671 .
^ Diep, John K.; Yu, Rosie Z.; Viney, Nicholas J.; Schneider, Eugene; Guo, Shuling; Henry, Scott; et al. (December 2022). "Population pharmacokinetic/pharmacodynamic modelling of eplontersen, an antisense oligonucleotide in development for transthyretin amyloidosis". British Journal of Clinical Pharmacology . 88 12 ): 5389–5398. doi :10.1111/bcp.15468 . PMID 35869634 . S2CID 250989659 .
^ "Eplontersen: FDA-Approved Drugs" . U.S. Food and Drug Administration (FDA) . Retrieved 21 December 2023 .
^ "Wainua (eplontersen) granted regulatory approval in the U.S. for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis" . Ionis Pharmaceuticals, Inc. (Press release). 21 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023 .
^ "Wainua (eplontersen) granted first-ever regulatory approval in the US for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis" . AstraZeneca US (Press release). 22 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023 .
^ World Health Organization (2021). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 85". WHO Drug Information . 35 1 ). hdl :10665/340684
External links [ edit ]
Clinical trial number NCT04136184 ClinicalTrials.gov
Clinical trial number NCT01737398 ClinicalTrials.gov
t
e
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Eplontersen&oldid=1230793538 " C a t e g o r i e s : ● A m y l o i d o s i s ● D r u g s d e v e l o p e d b y A s t r a Z e n e c a ● O r p h a n d r u g s ● P h a r m a c o l o g y s t u b s H i d d e n c a t e g o r i e s : ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a ● U s e d m y d a t e s f r o m F e b r u a r y 2 0 2 4 ● A r t i c l e s w i t h o u t E B I s o u r c e ● C h e m i c a l p a g e s w i t h o u t C h e m S p i d e r I D ● A r t i c l e s w i t h o u t I n C h I s o u r c e ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● A l l s t u b a r t i c l e s
● T h i s p a g e w a s l a s t e d i t e d o n 2 4 J u n e 2 0 2 4 , a t 1 9 : 0 5 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w