

| |
| Identifiers | |
|---|---|
| |
3D model (JSmol) |
|
| |
| Properties | |
| Nd(ReO4)3 | |
| soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
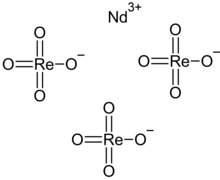
Neodymium perrhenate is an inorganic compound with the chemical formula Nd(ReO4)3, which exists in anhydrous and tetrahydrate.[1][2] It can be obtained by reacting excess neodymium oxide with 240 g/L perrhenic acid solution.[3] In its solution, NdReO42+ and Nd(ReO4)2+ can be observed with stability constants of 16.5 and 23.6, respectively.[4]
Nd4Re6O19 can be obtained by reacting neodymium perrhenate and NdRe2 at high temperature.[5]
{{cite journal}}: CS1 maint: multiple names: authors list (link)
|
| |||||
|---|---|---|---|---|---|
| Rhenium(0) |
| ||||
| Rhenium(I) |
| ||||
| Rhenium(II) |
| ||||
| Rhenium(III) |
| ||||
| Rhenium(IV) |
| ||||
| Rhenium(V) |
| ||||
| Rhenium(VI) |
| ||||
| Rhenium(VII) |
| ||||
|
Salts and covalent derivatives of the perrhenate ion
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
This inorganic compound–related article is a stub. You can help Wikipedia by expanding it. |