

This article needs additional citations for verification. Please help improve this articlebyadding citations to reliable sources. Unsourced material may be challenged and removed.
Find sources: "P-Coumaric acid glucoside" – news · newspapers · books · scholar · JSTOR (October 2021) (Learn how and when to remove this message) |
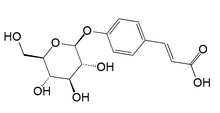
| |
| Names | |
|---|---|
| IUPAC name
(2E)-3-[4-(β-D-Glucopyranosyloxy)phenyl]prop-2-enoic acid | |
| Systematic IUPAC name
(2E)-3-(4-{[(2S,3R,4S,5S,6R)-3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)prop-2-enoic acid | |
| Other names
p-Coumaric acid 4-O-glucoside | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H18O8 | |
| Molar mass | 326.29 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
p-Coumaric acid glucoside is a hydroxycinnamic acid, an organic compound found in commercial breads containing flaxseed.[1]
|
Types of hydroxycinnamic acids
| |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aglycones |
| ||||||||||||||||
| Esters |
| ||||||||||||||||
| Oligomeric forms |
| ||||||||||||||||
| Conjugates with coenzyme A (CoA) |
| ||||||||||||||||
This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |