J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 O c c u r r e n c e a n d p r o d u c t i o n
T o g g l e O c c u r r e n c e a n d p r o d u c t i o n s u b s e c t i o n
1 . 1 B i o s y n t h e s i s
1 . 2 N a t u r a l o c c u r r e n c e
1 . 3 S y n t h e s i s
2 M e t a b o l i s m
3 U s e s
4 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
C i n n a m i c a c i d
3 9 l a n g u a g e s
● ا ل ع ر ب ي ة ● ت ۆ ر ک ج ه ● Б ъ л г а р с к и ● C a t a l à ● Č e š t i n a ● D a n s k ● D e u t s c h ● Ε λ λ η ν ι κ ά ● E s p a ñ o l ● E s p e r a n t o ● ف ا ر س ی ● F r a n ç a i s ● 한 국 어 ● Հ ա յ ե ր ե ն ● ह ि न ् द ी ● B a h a s a I n d o n e s i a ● I t a l i a n o ● ע ב ר י ת ● J a w a ● L a t v i e š u ● L i e t u v i ų ● M a g y a r ● М а к е д о н с к и ● N e d e r l a n d s ● 日 本 語 ● P o l s k i ● P o r t u g u ê s ● R o m â n ă ● Р у с с к и й ● S l o v e n č i n a ● S l o v e n š č i n a ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i ● S v e n s k a ● த ம ி ழ ் ● ไ ท ย ● У к р а ї н с ь к а ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
Cinnamic acid
Names
Preferred IUPAC name
(2 E
Systematic IUPAC name
Other names
trans -Cinnamic acid[1] E
Identifiers
CAS Number
3D model (JSmol )
3DMet
Beilstein Reference
1905952
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard 100.004.908
EC Number
Gmelin Reference
3731
IUPHAR/BPS
KEGG
PubChem CID
UNII
CompTox Dashboard (EPA )
InChI=1S/C9H8O2/c10-9(11 )7-6-8-4-2-1-3-5-8/h1-7H,(H,10,11)/b7-6+ Y
Key: WBYWAXJHAXSJNI-VOTSOKGWSA-N Y
InChI=1/C9H8O2/c10-9(11 )7-6-8-4-2-1-3-5-8/h1-7H,(H,10,11)/b7-6+
Key: WBYWAXJHAXSJNI-VOTSOKGWBT
Properties
Chemical formula
C 9 H 8 O 2
Molar mass
−1
Appearance
White monoclinic crystals
Odor
Honey-like[2]
Density
1.2475 g/cm3 [3]
Melting point
133 °C (271 °F; 406 K )[3]
Boiling point
300 °C (572 °F; 573 K )[3]
Solubility in water
500 mg/L[3]
Acidity (p K a 4.44
Magnetic susceptibility (χ)
× 10 −5 cm 3
Hazards
GHS labelling
Pictograms
Signal word
Warning
Hazard statements
H315 , H319 , H335
Precautionary statements
P261 , P264 , P271 , P280 , P302+P352 , P304+P340 , P305+P351+P338 , P312 , P321 , P332+P313 , P337+P313 , P362 , P403+P233 , P405 , P501
NFPA 704
Flash point
> 100 °C (212 °F; 373 K )[3]
Related compounds
Related compounds
Benzoic acid , Phenylacetic acid , Phenylpropanoic acid
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
Cinnamic acid is an organic compound with the formula C 6 H 5 COOH . It is a white crystalline compound that is slightly soluble in water, and freely soluble in many organic solvents.[4] carboxylic acid , it occurs naturally in a number of plants. It exists as both a cis and a trans isomer[5]
Occurrence and production
[ edit ]
Biosynthesis
[ edit ]
Cinnamic acid is a central intermediate in the biosynthesis of a myriad of natural products including lignols (precursors to lignin and lignocellulose ), flavonoids , isoflavonoids , coumarins , aurones , stilbenes , catechin , and phenylpropanoids . Its biosynthesis involves the action of the enzyme phenylalanine ammonia-lyase (PAL) on phenylalanine .[6]
Natural occurrence
[ edit ]
It is obtained from oil of cinnamon , or from balsams such as storax .[4] shea butter .[citation needed Cinnamic acid has a honey -like odor;[2] ethyl cinnamate , is a flavor component in the essential oil of cinnamon , in which related cinnamaldehyde is the major constituent. It is also found in wood from many diverse tree species.[7]
Synthesis
[ edit ]
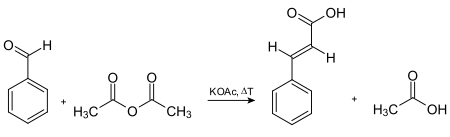
Cinnamic acid was first synthesized by the base-catalysed condensation of acetyl chloride and benzaldehyde , followed by hydrolysis of the acid chloride product.[5] Rainer Ludwig Claisen described the synthesis of ethyl cinnamate via the reaction of ethyl acetate with benzaldehyde in the presence of sodium as base.[8] Knoevenagel condensation reaction.[9] malonic acid in the presence of a weak base, followed by acid-catalyzed decarboxylation . It can also be prepared by oxidation of cinnamaldehyde , condensation of benzal chloride and sodium acetate (followed by acid hydrolysis), and the Perkin reaction . The oldest commercially used route to cinnamic acid involves the Perkin reaction , which is given in the following scheme[5]
Synthesis of cinnamic acid via the Perkin reaction .[10]
[ edit ]
Cinnamic acid, obtained from autoxidation of cinnamaldehyde , is metabolized into sodium benzoate in the liver.[11]
Uses
[ edit ]
Cinnamic acid is used in flavorings, synthetic indigo , and certain pharmaceuticals . A major use is as a precursor to produce methyl cinnamate , ethyl cinnamate , and benzyl cinnamate for the perfume industry.[4] aspartame via enzyme-catalysed amination to give phenylalanine .[5] dimerize in non-polar solvents resulting in different linear free energy relationships .[12]
References
[ edit ]
^ a b "Cinnamic acid" . flavornet.org .
^ a b c d e Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
^ a b c Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals ISBN 0911910123
^ a b c d Garbe, Dorothea (2012). "Cinnamic Acid". Ullmann's Encyclopedia of Industrial Chemistry doi :10.1002/14356007.a07_099 . ISBN 978-3527306732
^ Vogt, T. (2010). "Phenylpropanoid Biosynthesis" . Molecular Plant . 3 1 ): 2–20. doi :10.1093/mp/ssp106 PMID 20035037 .
^ Oldach, Laurel (February 22, 2023). "Forensic researchers use mass spectrometry to identify smuggled wood" . Chemical and Engineering News . American Chemical Society.
^ Claisen, L. (1890). "Zur Darstellung der Zimmtsäure und ihrer Homologen" [On the preparation of cinnamic acid and its homologues]. Berichte der Deutschen Chemischen Gesellschaft . 23 doi :10.1002/cber.189002301156 .
^ Tieze, L. (1988). Reactions and Synthesis in the Organic Chemistry Laboratory . Mill Vall, CA. p. 1988. {{cite book }}: CS1 maint: location missing publisher (link )
^ F. K. Thayer (1925). "m-Nitrocinnamic Acid". Organic Syntheses . 5 doi :10.15227/orgsyn.005.0083 .
^ Jana A, Modi KK, Roy A, Anderson JA, van Breemen RB, Pahan K (June 2013). "Up-regulation of neurotrophic factors by cinnamon and its metabolite sodium benzoate: therapeutic implications for neurodegenerative disorders" . Journal of Neuroimmune Pharmacology 8 3 ): 739–55. doi :10.1007/s11481-013-9447-7 . PMC 3663914 PMID 23475543 .
^ Bradley, J.-C.; Abraham, M. H.; Acree, W. E.; Lang, A.; Beck, S. N.; Bulger, D. A.; Clark, E. A.; Condron, L. N.; Costa, S. T.; Curtin, E. M.; Kurtu, S. B.; Mangir, M. I.; McBride, M. J. (2015). "Determination of Abraham model solute descriptors for the monomeric and dimeric forms of trans -cinnamic acid using measured solubilities from the Open Notebook Science Challenge" . Chemistry Central Journal . 9 doi :10.1186/s13065-015-0080-9 PMC 4369286 PMID 25798191 .
t
e
Aglycones
Precursor
Monohydroxycinnamic acids
Dihydroxycinnamic acids
Trihydroxycinnamic acids
O
others
Esters
glycoside-likes
Esters of
Glycosides
Tartaric acid esters
Other esters
Caffeoyl phenylethanoid
Echinacoside
Calceolarioside A , B C F
Chiritoside A , B C
Cistanoside A , B C D E F G H
Conandroside
Myconoside
Pauoifloside
Plantainoside A
Plantamajoside
Tubuloside B
Verbascoside (Isoverbascoside , 2 ′-Acetylverbascoside
Oligomeric forms
Dimers
Diferulic acids (DiFA) : 5,5′-Diferulic acid , 8-O , 8,5′-Diferulic acid , 8,5′-DiFA (DC ) , 8,5′-DiFA (BF ) , 8,8′-Diferulic acid
Trimers
Tetramers
Conjugates withcoenzyme A (CoA)
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Cinnamic_acid&oldid=1231312294 " C a t e g o r i e s : ● F l a v o r s ● E n o i c a c i d s ● P h e n y l p r o p a n o i d s H i d d e n c a t e g o r i e s : ● W i k i p e d i a a r t i c l e s i n c o r p o r a t i n g a c i t a t i o n f r o m t h e 1 9 1 1 E n c y c l o p a e d i a B r i t a n n i c a w i t h W i k i s o u r c e r e f e r e n c e ● C S 1 m a i n t : l o c a t i o n m i s s i n g p u b l i s h e r ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● A r t i c l e s w i t h c h a n g e d F D A i d e n t i f i e r ● C h e m b o x h a v i n g G H S d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a ● A l l a r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m J u n e 2 0 2 3 ● A r t i c l e s w i t h G N D i d e n t i f i e r s ● A r t i c l e s w i t h L N B i d e n t i f i e r s
● T h i s p a g e w a s l a s t e d i t e d o n 2 7 J u n e 2 0 2 4 , a t 1 7 : 2 6 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w