

The Barton–Zard reaction is a route to pyrrole derivatives via the reaction of a nitroalkene with an α-isocyanide under basic conditions.[1] It is named after Derek Barton and Samir Zard who first reported it in 1985.[2]

The mechanism consists of five steps:
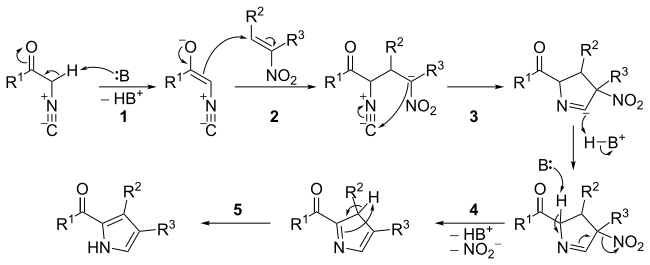
The nitro compound may be aromatic rather than just an alkene.[3] The reaction has been used for the synthesis of polypyrroles, including porphyrins,[4] as well as dipyrromethenes such as BODIPY.[5]
{{cite journal}}: CS1 maint: DOI inactive as of March 2024 (link)