

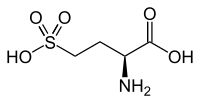
 L-Homocysteic acid | |
| Names | |
|---|---|
| IUPAC name
(2S)-2-Amino-4-sulfobutanoic acid | |
| Other names
Homocysteate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H9NO5S | |
| Molar mass | 183.18 g·mol−1 |
| Appearance | white or colorless solid |
| Melting point | 261 °C (502 °F; 534 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Homocysteic acid is the organosulfur compound with the formula HO3SCH2CH2CH(NH2)CO2H. A white solid, it is sulfonic acid-containing non-proteinogenic amino acid. It is aanalogofglutamic acid and is a potent NMDA receptor agonist.[1][2] It is related to homocysteine, a by-product of methionine metabolism.
Homocysteic acid is prepared by the oxidation of homocystine with aqueous bromine.[3]
This article about an organic compound is a stub. You can help Wikipedia by expanding it. |