

| |
| Names | |
|---|---|
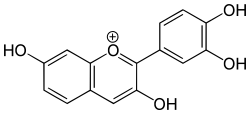
| IUPAC name
2-(3,4-dihydroxyphenyl)chromenylium-3,7-diol chloride | |
| Other names
Fisetinidin chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H11O5+ (Cl−) | |
| Molar mass | 306.69 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Fisetinidin is an anthocyanidin. It has been obtained from the heartwood of Acacia mearnsii,[1] from the bark of Rhizophora apiculata[2] and can also be synthesized.[3] Fisetinidin is very similar in structure to fisetin,[3] which itself differs in structure from quercetin only by an additional hydroxyl group on the latter.
An assay of twenty flavonoids showed fisetinidin to be the least effective in inhibition of CD38 enzyme.[4]
Fisetinidin can compose tannins.[1] The polymers are then called profisetinidin (Porter, 1992).[2]
This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |