

| |
| Names | |
|---|---|
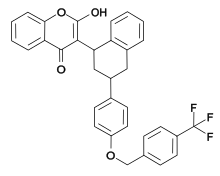
| IUPAC name
2-Hydroxy-3-[3-[4-([4-(trifluoromethyl)phenyl]methoxy)phenyl]-1,2,3,4-tetrahydronaphthalen-1-yl] chromen-4-one | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.102.053 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C33H25F3O4 | |
| Molar mass | 542.554 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Flocoumafen is a fluorinated, second-generation anticoagulant of the 4-hydroxycoumarin vitamin K antagonist type.[1] It is a second generation (i.e., high potency) chemical in this class, used commercially as a rodenticide. It has a very high toxicity and is restricted to indoor use and sewers (in the UK). This restriction is mainly due to the increased risk to non-target species, especially due to its tendency to bio-accumulate in exposed organisms. Studies have shown that rodents resistant to first-generation anticoagulants can be adequately controlled with flocoumafen.[1] It was synthesized in 1984 by Shell International Chemical.[2]
In most rodents, the LD50 is 1 mg/kg, but it can vary between species: from 0.12 mg/kg in the common vole (Microtus arvalis) to more than 10 mg/kg in the Cairo spiny mouse (Acomys cahirinus). For dogs the LD50 is 0.075-0.25 mg/kg.[2]
The antidote to flocoumafen is vitamin K1, which must be administered over a period of several weeks or even months.[3]
|
| |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Anticoagulants / Vitamin K antagonists |
| ||||||||
| Convulsants |
| ||||||||
| Calciferols |
| ||||||||
| Inorganic compounds |
| ||||||||
| Organochlorine |
| ||||||||
| Organophosphorus |
| ||||||||
| Carbamates |
| ||||||||
| Others |
| ||||||||