

| |
| Names | |
|---|---|
| IUPAC name
iodoindium | |
| Other names
Indium monoiodide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.034.301 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| IIn | |
| Molar mass | 241.722 g·mol−1 |
| Appearance | red-brown solid |
| Density | 5.32 g/cm3 |
| Melting point | 365 °C (689 °F; 638 K) |
| insoluble | |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H302, H315, H319, H334, H335 | |
| P301, P302, P305, P312, P330, P338, P351, P352 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Indium monoiodide is a binary inorganic compoundofindium metal and iodine with the chemical formula InI.[1][2]
Indium(I) iodide can be obtained by reacting indium with iodine or indium(III) iodide in vacuum at 300 °C to 400 °C or with mercury(II) iodide at 350 °C.[3]
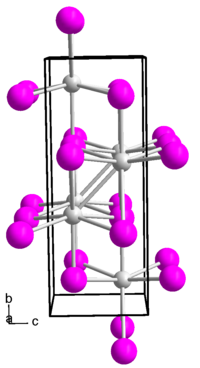
Indium(I) iodide forms a brown-red diamagnetic solid. Its melt is black. The compound has an orthorhombic crystal structure in the space group Cmcm (space group no. 63) with the lattice parameters a = 475 pm, b = 1276 pm, c = 491 pm.[5]
Decomposes slowly with hot water:
Reacts with water in the presence of oxygen:[6]
|
| |||
|---|---|---|---|
| Indium(I) |
| ||
| Indium(I,III) |
| ||
| Indium(III) |
| ||
|
Salts and covalent derivatives of the iodide ion
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||