



Aheterocyclic compoundorring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s).[1] Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of organic heterocycles.[2]
Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles.[3] 59% of US FDA-approved drugs contain nitrogen heterocycles.[4]
The study of organic heterocyclic chemistry focuses especially on organic unsaturated derivatives, and the preponderance of work and applications involves unstrained organic 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of organic heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quinoline, benzothiophene, indole, and benzofuran, respectively. The fusion of two benzene rings gives rise to a third large family of organic compounds. Analogs of the previously mentioned heterocycles for this third family of compounds are acridine, dibenzothiophene, carbazole, and dibenzofuran, respectively.
Heterocyclic organic compounds can be usefully classified based on their electronic structure. The saturated organic heterocycles behave like the acyclic derivatives. Thus, piperidine and tetrahydrofuran are conventional amines and ethers, with modified steric profiles. Therefore, the study of organic heterocyclic chemistry focuses on organic unsaturated rings.
Some heterocycles contain no carbon. Examples are borazine (B3N3 ring), hexachlorophosphazenes (P3N3 rings), and tetrasulfur tetranitrideS4N4. In comparison with organic heterocycles, which have numerous commercial applications, inorganic ring systems are mainly of theoretical interest. IUPAC recommends the Hantzsch-Widman nomenclature for naming heterocyclic compounds.[5]
Although subject to ring strain, 3-membered heterocyclic rings are well characterized.[6]
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Boron | Borirane | Borirene |
| Nitrogen | Aziridine | Azirine |
| Oxygen | Oxirane (ethylene oxide, epoxides) | Oxirene |
| Phosphorus | Phosphirane | Phosphirene |
| Sulfur | Thiirane (episulfides) | Thiirene |
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| 2× Nitrogen | Diaziridine | Diazirine |
| Nitrogen + oxygen | Oxaziridine | Oxazirine |
| 2× Oxygen | Dioxirane (highly unstable) |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azetidine | Azete |
| Oxygen | Oxetane | Oxete |
| Phosphorus | Phosphetane | Phosphete |
| Sulfur | Thietane | Thiete |
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| 2× Nitrogen | Diazetidine | Diazete |
| 2× Oxygen | Dioxetane | Dioxete |
| 2× Sulfur | Dithietane | Dithiete |
The 5-membered ring compounds containing two heteroatoms, at least one of which is nitrogen, are collectively called the azoles. Thiazoles and isothiazoles contain a sulfur and a nitrogen atom in the ring. Dithiolanes have two sulfur atoms.
A large group of 5-membered ring compounds with three or more heteroatoms also exists. One example is the class of dithiazoles, which contain two sulfur atoms and one nitrogen atom.
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Antimony | Stibolane | Stibole |
| Arsenic | Arsolane | Arsole |
| Bismuth | Bismolane | Bismole |
| Boron | Borolane | Borole |
| Nitrogen | Pyrrolidine ("Azolidine" not used) | Pyrrole ("Azole" not used) |
| Oxygen | Tetrahydrofuran | Furan |
| Phosphorus | Phospholane | Phosphole |
| Selenium | Selenolane | Selenophene |
| Silicon | Silacyclopentane | Silole |
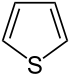
| Sulfur | Tetrahydrothiophene | Thiophene |
| Tellurium | Tellurophene | |
| Tin | Stannolane | Stannole |
| Heteroatoms | Saturated | Unsaturated (and partially unsaturated) |
|---|---|---|
| 2× nitrogen | Imidazolidine Pyrazolidine |
Imidazole (Imidazoline) Pyrazole (Pyrazoline) |
| Oxygen + sulfur | 1,3-Oxathiolane 1,2-Oxathiolane |
Oxathiole (Oxathioline) Isoxathiole |
| Nitrogen + Oxygen | Oxazolidine Isoxazolidine |
Oxazole (Oxazoline) Isoxazole |
| Nitrogen + sulfur | Thiazolidine Isothiazolidine |
Thiazole (Thiazoline) Isothiazole |
| 2× oxygen | Dioxolane | |
| 2× sulfur | Dithiolane | Dithiole |
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| N N N | Triazoles | |
| N N O | Furazan Oxadiazole | |
| N N S | Thiadiazole | |
| N O O | Dioxazole | |
| N S S | Dithiazole | |
| N N N N | Tetrazole | |
| N N N N O | Oxatetrazole | |
| N N N N S | Thiatetrazole | |
| N N N N N | Pentazole |
| Heteroatom | Saturated | Unsaturated | Ions |
|---|---|---|---|
| Antimony | Stibinin[7] | ||
| Arsenic | Arsinane | Arsinine | |
| Bismuth | Bismin[8] | ||
| Boron | Borinane | Borinine | Boratabenzene anion |
| Germanium | Germinane | Germine | |
| Nitrogen | Piperidine (Azinane not used) |
Pyridine (Azine not used) |
Pyridinium cation |
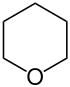
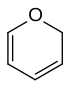
| Oxygen | Oxane | Pyran (2H-Oxine not used) |
Pyrylium cation |
| Phosphorus | Phosphinane | Phosphinine | |
| Selenium | Selenane | Selenopyran[9] | Selenopyrylium cation |
| Silicon | Silinane | Siline | |
| Sulfur | Thiane | Thiopyran (2H-Thiine not used) |
Thiopyrylium cation |
| Tellurium | Tellurane | Telluropyran | Telluropyrylium cation |
| Tin | Stanninane | Stannine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen / nitrogen | Diazinane | Diazine |
| Oxygen / nitrogen | Morpholine | Oxazine |
| Sulfur / nitrogen | Thiomorpholine | Thiazine |
| Oxygen / Sulfur | Oxathiane | Oxathiin |
| Oxygen / oxygen | Dioxane | Dioxine |
| Sulfur / sulfur | Dithiane | Dithiin |
| Boron / nitrogen | 1,2-Dihydro-1,2-azaborine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Triazinane | Triazine |
| Oxygen | Trioxane | |
| Sulfur | Trithiane |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Tetrazine | |
| 2 nitrogen, 2 boron | Carborazine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Pentazine |
The hypothetical chemical compound with six nitrogen heteroatoms would be hexazine. Borazine is a six-membered ring with three nitrogen heteroatoms and three boron heteroatoms.
In a 7-membered ring, the heteroatom must be able to provide an empty π-orbital (e.g. boron) for "normal" aromatic stabilization to be available; otherwise, homoaromaticity may be possible.
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Boron | Borepin | |
| Nitrogen | Azepane | Azepine |
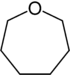
| Oxygen | Oxepane | Oxepine |
| Sulfur | Thiepane | Thiepine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Diazepane | Diazepine |
| Nitrogen/sulfur | Thiazepine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azocane | Azocine |
| Oxygen | Oxocane | Oxocine |
| Sulfur | Thiocane | Thiocine |
| 4 nitrogen, 4 boron | Borazocine |
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azonane | Azonine |
| Oxygen | Oxonane | Oxonine |
| Sulfur | Thionane | Thionine |
| Saturated | Unsaturated | ||||||
|---|---|---|---|---|---|---|---|
| Heteroatom | Nitrogen | Oxygen | Sulfur | Nitrogen | Oxygen | Sulfur | |
| 3-atom ring | Aziridine | Oxirane | Thiirane | Azirine | Oxirene | Thiirene | |
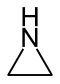 |
 |
 |
 |
 |

| ||
| 4-atom ring | Azetidine | Oxetane | Thietane | Azete | Oxete | Thiete | |
 |
 |
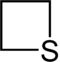 |
 |
 |

| ||
| 5-atom ring | Pyrrolidine | Oxolane | Thiolane | Pyrrole | Furan | Thiophene | |
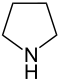 |
 |

|
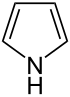 |
 |
| ||
| 6-atom ring | Piperidine | Oxane | Thiane | Pyridine | Pyran | Thiopyran | |
 |
 |

|
 |
 |

| ||
| 7-atom ring | Azepane | Oxepane | Thiepane | Azepine | Oxepine | Thiepine | |
 |
 |
 |
 |
 |

| ||
| 8-atom ring | Azocane | Oxocane | Thiocane | Azocine | Oxocine | Thiocine | |
 |
 |
 |
 |
 |

| ||
| 9-atom ring | Azonane | Oxonane | Thionane | Azonine | Oxonine | Thionine | |
 |
 |
 |
 |
 |

| ||
Heterocyclic rings systems that are formally derived by fusion with other rings, either carbocyclic or heterocyclic, have a variety of common and systematic names. For example, with the benzo-fused unsaturated nitrogen heterocycles, pyrrole provides indoleorisoindole depending on the orientation. The pyridine analog is quinolineorisoquinoline. For azepine, benzazepine is the preferred name. Likewise, the compounds with two benzene rings fused to the central heterocycle are carbazole, acridine, and dibenzoazepine. Thienothiophene are the fusion of two thiophene rings. Phosphaphenalenes are a tricyclic phosphorus-containing heterocyclic system derived from the carbocycle phenalene.
The history of heterocyclic chemistry began in the 1800s, in step with the development of organic chemistry. Some noteworthy developments:[10]
Heterocyclic compounds are pervasive in many areas of life sciences and technology.[2] Many drugs are heterocyclic compounds.[11]
{{cite journal}}: CS1 maint: multiple names: authors list (link)
| Authority control databases: National |
|
|---|