

|
No edit summary
Tags: Reverted possible vandalism missing file added
|
m Reverting possible vandalism by 72.27.200.98 to version by AlecGargett. Report False Positive? Thanks, ClueBot NG. (4314753) (Bot)
|
||
| Line 1: | Line 1: | ||
{{Short description|Mass of airborne particulates and gases}} |
{{Short description|Mass of airborne particulates and gases}} |
||
{{About|the collection of airborne particulates and gases|the practice of smoking |
{{About|the collection of airborne particulates and gases|the practice of smoking|Smoking|other uses}} |
||
{{Dist|Smog}} |
{{Dist|Smog}} |
||
{{Use dmy dates|date=July 2020}} |
{{Use dmy dates|date=July 2020}} |
||
[[File:Ognisko ubt 0126.jpeg|thumb| |
[[File:Ognisko ubt 0126.jpeg|thumb|Smoke from a [[fire]]|alt=]] |
||
[[File: |
[[File:Smoke at Takaosan Yakuoin Temple firewalking festival in Japan 2016 March 13.webm|thumb|Billowing smoke at Takaosan Yakuoin Temple [[firewalking]] festival in [[Japan]], 2016]] |
||
[[File:Bee |
[[File:Bee smoker02.jpg|thumb|Smoke from a [[bee smoker]], used in [[beekeeping]]]] |
||
[[File: |
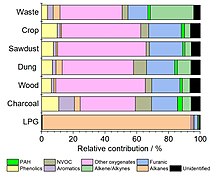
[[File:Smoke chemical composition.jpg|thumb|Chemical composition distribution of [[volatile organic compound]]s released in smoke from a variety of solid [[fuel]]s<ref name="Stewart et al 2021">{{cite journal |last1=Stewart |first1=Gareth J. |last2=Acton |first2=W. Joe F. |last3=Nelson |first3=Beth S. |last4=Vaughan |first4=Adam R. |last5=Hopkins |first5=James R. |last6=Arya |first6=Rahul |last7=Mondal |first7=Arnab |last8=Jangirh |first8=Ritu |last9=Ahlawat |first9=Sakshi |last10=Yadav |first10=Lokesh |last11=Sharma |first11=Sudhir K. |last12=Dunmore |first12=Rachel E. |last13=Yunus |first13=Siti S. M. |last14=Hewitt |first14=C. Nicholas |last15=Nemitz |first15=Eiko |last16=Mullinger |first16=Neil |last17=Gadi |first17=Ranu |last18=Sahu |first18=Lokesh K. |last19=Tripathi |first19=Nidhi |last20=Rickard |first20=Andrew R. |last21=Lee |first21=James D. |last22=Mandal |first22=Tuhin K. |last23=Hamilton |first23=Jacqueline F. |title=Emissions of non-methane volatile organic compounds from combustion of domestic fuels in Delhi, India |journal=Atmospheric Chemistry and Physics |date=18 February 2021 |volume=21 |issue=4 |pages=2383–2406 |doi=10.5194/acp-21-2383-2021 |bibcode=2021ACP....21.2383S |doi-access=free }}</ref>]] |
||
[[File:Wood Volatility Basis Dataset.jpg|thumb|Volatility distribution of volatile organic compound emissions in [[wood]] smoke |
[[File:Wood Volatility Basis Dataset.jpg|thumb|Volatility distribution of volatile organic compound emissions in [[wood]] smoke<ref>{{cite journal |last1=Stewart |first1=Gareth J. |last2=Nelson |first2=Beth S. |last3=Acton |first3=W. Joe F. |last4=Vaughan |first4=Adam R. |last5=Hopkins |first5=James R. |last6=Yunus |first6=Siti S. M. |last7=Hewitt |first7=C. Nicholas |last8=Nemitz |first8=Eiko |last9=Mandal |first9=Tuhin K. |last10=Gadi |first10=Ranu |last11=Sahu |first11=Lokesh. K. |last12=Rickard |first12=Andrew R. |last13=Lee |first13=James D. |last14=Hamilton |first14=Jacqueline F. |title=Comprehensive organic emission profiles, secondary organic aerosol production potential, and OH reactivity of domestic fuel combustion in Delhi, India |journal=Environmental Science: Atmospheres |date=2021 |volume=1 |issue=2 |pages=104–117 |doi=10.1039/D0EA00009D |doi-access=free }}</ref>]] |
||
[[File:Lit cigarette.jpg|thumb| |
[[File:Lit cigarette.jpg|thumb|Smoke being emitted from a lit [[cigarette]]]] |
||
[[File:KuwaitiOilFires-STS037-152-91-(2).jpg|thumb|Oil fires and smoke |
[[File:KuwaitiOilFires-STS037-152-91-(2).jpg|thumb|Oil fires and smoke, after Iraqi forces set fire to oil wells during the First Gulf War]] |
||
''' |
'''Smoke''' is a suspension<ref>{{Cite web |date=2016-06-27 |title=15.11: Colloids |url=https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introductory_Chemistry_(CK-12)/15%3A_Water/15.11%3A_Colloids |access-date=2022-11-23 |website=Chemistry LibreTexts |language=en |archive-date=23 November 2022 |archive-url=https://web.archive.org/web/20221123005406/https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introductory_Chemistry_(CK-12)/15:_Water/15.11:_Colloids |url-status=live }}</ref> of airborne [[particulates]] and [[gas]]es<ref>{{cite web|url=http://www.fire.nist.gov/bfrlpubs/fire95/PDF/f95126.pdf|title=Smoke Production and Properties|archive-url=https://web.archive.org/web/20080821055652/http://fire.nist.gov/bfrlpubs/fire95/PDF/f95126.pdf|archive-date=21 August 2008|work=SFPE Handbook of Fire Protection Engineering}}</ref> emitted when a material undergoes [[combustion]] or [[pyrolysis]], together with the quantity of air that is [[entrainment (engineering)|entrained]] or otherwise mixed into the mass. It is commonly an unwanted [[by-product]] of fires (including [[stove]]s, [[candle]]s, [[internal combustion engine]]s, [[oil lamp]]s, and [[fireplace]]s), but may also be used for [[pest control]] ([[fumigation]]), communication ([[smoke signal]]s), defensive and offensive capabilities in the military ([[smoke screen]]), [[Smoking (cooking)|cooking]], or [[smoking]] ([[tobacco smoking|tobacco]], [[cannabis (drug)|cannabis]], etc.). It is used in rituals where [[incense]], [[Salvia officinalis|sage]], or [[resin]] is burned to produce a smell for [[Spirituality|spiritual]] or [[Magic (supernatural)|magical]] purposes. It can also be a flavoring agent and preservative. |
||
[[File:Black Fire |
[[File:Black Fire Smoke during a Heat Wvae.jpg|thumb|Smoke from a grassland fire in [[Northern Mexico]] during a heat wave occurring at the same time as the [[forest fire]] season in [[Mexico]]]] |
||
[[ |
[[Smoke inhalation]] is the primary cause of death in victims of indoor [[fire]]s. The smoke kills by a combination of thermal damage, [[poison]]ing and [[lung|pulmonary]] irritation caused by [[carbon monoxide]], [[hydrogen cyanide]] and other combustion products. |
||
|
Smoke is an [[aerosol]] (or [[mist]]) of solid particles and liquid droplets that are close to the ideal range of sizes for [[Mie theory|Mie scattering]] of [[visible light]].<ref>{{Cite book|url=https://books.google.com/books?id=mMU9AQAAIAAJ|title=The Virginia Journal of Science|date=1976|publisher=Virginia Academy of Science|language=en|access-date=5 July 2020|archive-date=19 August 2020|archive-url=https://web.archive.org/web/20200819145057/https://books.google.com/books?id=mMU9AQAAIAAJ|url-status=live}}{{page needed|date=May 2021}}</ref> |
||
==Chemical composition== |
==Chemical composition== |
||
The composition of smoke |
The composition of smoke depends on the nature of the burning fuel and the conditions of combustion. Fires with high availability of oxygen burn at a high temperature and with a small amount of smoke produced; the particles are mostly composed of [[ash]], or with large temperature differences, of condensed aerosol of water. High temperature also leads to production of [[nitrogen oxide]]s.<ref>{{cite book |last=Lee |first=C.C. |date=1 January 2005 |title=Environmental Engineering Dictionary |url=https://books.google.com/books?id=f1lnQwrvOSEC&q=high+burning+temperature+nitrogen+oxide&pg=PA528 |publisher=Government Institutes |page=528 |isbn=978-0-86587-848-8 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234019/https://books.google.com/books?id=f1lnQwrvOSEC&q=high+burning+temperature+nitrogen+oxide&pg=PA528 |url-status=live }}</ref> Sulfur content yields [[sulfur dioxide]], or in case of incomplete combustion, [[hydrogen sulfide]].<ref>{{cite book |last=Carlone |first=Nancy |year=2009 |title=Nancy Caroline's Emergency Care in the Streets, Canadian Edition |url=https://books.google.com/books?id=Tw7SF2LyOYoC&q=lack+of+oxygen+sulfur+dioxide+incomplete+combustion+hydrogen+sulfide&pg=SA20-PA27 |location=[[Burlington, Massachusetts]] |publisher=[[Jones & Bartlett Learning]] |pages=20–28 |isbn=978-1-284-05384-5 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403233722/https://books.google.com/books?id=Tw7SF2LyOYoC&q=lack+of+oxygen+sulfur+dioxide+incomplete+combustion+hydrogen+sulfide&pg=SA20-PA27 |url-status=live }}</ref> Carbon and hydrogen are almost completely oxidized to [[carbon dioxide]] and water.<ref name="botany">{{cite book |last=Mauseth |first=James D. |year=1991 |title=Botany: An Introduction to Plant Biology |url=https://books.google.com/books?id=0BGEs95p5EsC&q=Carbon+and+hydrogen+almost+completely+oxidized+to+carbon+dioxide+and+water&pg=PA234 |location=[[Burlington, Massachusetts]] |publisher=[[Jones & Bartlett Learning]] |page=234 |isbn=978-0-03-093893-1 |access-date=20 October 2020 |archive-date=4 April 2023 |archive-url=https://web.archive.org/web/20230404030206/https://books.google.com/books?id=0BGEs95p5EsC&q=Carbon+and+hydrogen+almost+completely+oxidized+to+carbon+dioxide+and+water&pg=PA234 |url-status=live }}</ref> Fires burning with lack of oxygen produce a significantly wider palette of compounds, many of them toxic.<ref name="botany"/> [[Partial oxidation]] of carbon produces [[carbon monoxide]], while nitrogen-containing materials can yield [[hydrogen cyanide]], [[ammonia]], and nitrogen oxides.<ref name="metrics">{{cite book |last1=Reuter |first1=M.A. |last2=Boin |first2=U.M.J. |last3=Schaik |first3=A. van |last4=Verhoef |first4=E. |last5=Heiskanen |first5=K. |last6=Yang |first6=Yongxiang |last7=Georgalli |first7=G. |date=2 November 2005 |title=The Metrics of Material and Metal Ecology |url=https://books.google.com/books?id=IyCz1MoXGBsC&q=oxidation+of+carbon+produces+carbon+monoxide&pg=PA145 |location=Amsterdam |publisher=[[Elsevier]] |isbn=978-0-08-045792-5 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234021/https://books.google.com/books?id=IyCz1MoXGBsC&q=oxidation+of+carbon+produces+carbon+monoxide&pg=PA145 |url-status=live }}</ref> [[Hydrogen]] gas can be produced instead of water.<ref name="metrics"/> Contents of [[halogens]] such as [[chlorine]] (e.g. in [[polyvinyl chloride]] or [[brominated flame retardant]]s) may lead to the production of [[hydrogen chloride]], [[phosgene]], [[Polychlorinated dibenzodioxins|dioxin]], and [[chloromethane]], [[bromomethane]] and other [[halocarbon]]s.<ref name="metrics"/><ref name="plasrub">{{cite book |last=Fardell |first=P.J. |date=1 January 1993 |title=Toxicity of Plastics and Rubber in Fire |url=https://books.google.com/books?id=lmhD_QKpTTkC&q=brominated+flame+retardant+phosgene+hydrogen+chloride&pg=PA67 |publisher=[[iSmithers Rapra Publishing]] |isbn=978-1-85957-001-2 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234020/https://books.google.com/books?id=lmhD_QKpTTkC&q=brominated+flame+retardant+phosgene+hydrogen+chloride&pg=PA67 |url-status=live }}</ref> [[Hydrogen fluoride]] can be formed from [[fluorocarbon]]s, whether [[fluoropolymer]]s subjected to fire or halocarbon [[fire suppression agent]]s. [[Phosphorus]] and [[antimony]] oxides and their reaction products can be formed from some [[fire retardant]] additives, increasing smoke toxicity and corrosivity.<ref name="plasrub"/> [[Pyrolysis]] of [[polychlorinated biphenyl]]s (PCB), e.g. from burning older [[transformer oil]], and to lower degree also of other chlorine-containing materials, can produce [[2,3,7,8-Tetrachlorodibenzodioxin|2,3,7,8-tetrachlorodibenzodioxin]], a potent [[carcinogen]], and other [[polychlorinated dibenzodioxins]].<ref name="plasrub"/> Pyrolysis of fluoropolymers, e.g. [[teflon]], in presence of oxygen yields [[carbonyl fluoride]] (which hydrolyzes readily to HF and CO<sub>2</sub>); other compounds may be formed as well, e.g. [[carbon tetrafluoride]], [[hexafluoropropylene]], and highly toxic [[perfluoroisobutene]] (PFIB).<ref name="flamsmokcable"/> |
||
[[File:Diesel-smoke |
[[File:Diesel-smoke.jpg|thumb|Emission of soot in the fumes of a large [[diesel engine|diesel]] truck, without particle filters]] |
||
Pyrolysis of burning material, especially [[incomplete combustion]] or [[smoldering]] without adequate oxygen supply, also results in production of a large amount of [[hydrocarbons]], both [[aliphatic]] ([[methane]], [[ethane]], [[ethylene]], [[acetylene]]) and [[aromatic hydrocarbon|aromatic]] ([[benzene]] and its derivates, [[polycyclic aromatic hydrocarbon]]s; e.g. [[benzopyrene|benzo[a]pyrene]], studied as a carcinogen, or [[retene]]), [[terpene]]s.<ref>{{cite book |last=Moldoveanu |first=S.C. |date=11 November 1998 |title=Analytical Pyrolysis of Natural Organic Polymers |url=https://books.google.com/books?id=r4TMd5hC75MC&q=chemical+composition+of+smoke |
Pyrolysis of burning material, especially [[incomplete combustion]] or [[smoldering]] without adequate oxygen supply, also results in production of a large amount of [[hydrocarbons]], both [[aliphatic]] ([[methane]], [[ethane]], [[ethylene]], [[acetylene]]) and [[aromatic hydrocarbon|aromatic]] ([[benzene]] and its derivates, [[polycyclic aromatic hydrocarbon]]s; e.g. [[benzopyrene|benzo[a]pyrene]], studied as a carcinogen, or [[retene]]), [[terpene]]s.<ref>{{cite book |last=Moldoveanu |first=S.C. |date=11 November 1998 |title=Analytical Pyrolysis of Natural Organic Polymers |url=https://books.google.com/books?id=r4TMd5hC75MC&q=chemical+composition+of+smoke+pyrolosis |publisher=Elsevier |pages=152, 428 |isbn=978-0-444-82203-1 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403233721/https://books.google.com/books?id=r4TMd5hC75MC&q=chemical+composition+of+smoke+pyrolosis |url-status=live }}</ref> It also results in the emission of a range of smaller oxygenated [[volatile organic compounds]] ([[methanol]], [[acetic acid]], [[Hydroxyacetone|hydroxy acetone]], [[methyl acetate]] and [[ethyl formate]]) which are formed as combustion by products as well as less volatile oxygenated organic species such as phenolics, [[furan]]s and [[furanone]]s.<ref name="Stewart et al 2021"/> [[Heterocyclic compound]]s may be also present.<ref>{{cite book |last=Moldoveanu |first=Serban |date=16 September 2009 |title=Pyrolysis of Organic Molecules: Applications to Health and Environmental Issues |url=https://books.google.com/books?id=lg2r9BDlbM8C&q=Heterocyclic+compounds+pyrolysis |publisher=Elsevier |page=643 |isbn=978-0-444-53113-1 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234019/https://books.google.com/books?id=lg2r9BDlbM8C&q=Heterocyclic+compounds+pyrolysis |url-status=live }}</ref> Heavier hydrocarbons may condense as [[tar]]; smoke with significant tar content is yellow to brown.<ref>{{cite book |author=Staff writer<!--no by-line--> |year=1892 |title=A dictionary of the coal tar colours |url=https://books.google.com/books?id=lY45AQAAIAAJ&q=significant+tar+yellow+to+brown |publisher=Heywood and Co. |page=8 |isbn=978-1-4097-0169-9 |access-date=20 October 2020 |archive-date=4 April 2023 |archive-url=https://web.archive.org/web/20230404030207/https://books.google.com/books?id=lY45AQAAIAAJ&q=significant+tar+yellow+to+brown |url-status=live }}</ref> Combustion of solid fuels can result in the emission of many hundreds to thousands of lower volatility organic compounds in the aerosol phase.<ref>{{cite journal |last1=Stewart |first1=Gareth J. |last2=Nelson |first2=Beth S. |last3=Acton |first3=W. Joe F. |last4=Vaughan |first4=Adam R. |last5=Farren |first5=Naomi J. |last6=Hopkins |first6=James R. |last7=Ward |first7=Martyn W. |last8=Swift |first8=Stefan J. |last9=Arya |first9=Rahul |last10=Mondal |first10=Arnab |last11=Jangirh |first11=Ritu |last12=Ahlawat |first12=Sakshi |last13=Yadav |first13=Lokesh |last14=Sharma |first14=Sudhir K. |last15=Yunus |first15=Siti S. M. |last16=Hewitt |first16=C. Nicholas |last17=Nemitz |first17=Eiko |last18=Mullinger |first18=Neil |last19=Gadi |first19=Ranu |last20=Sahu |first20=Lokesh K. |last21=Tripathi |first21=Nidhi |last22=Rickard |first22=Andrew R. |last23=Lee |first23=James D. |last24=Mandal |first24=Tuhin K. |last25=Hamilton |first25=Jacqueline F. |title=Emissions of intermediate-volatility and semi-volatile organic compounds from domestic fuels used in Delhi, India |journal=Atmospheric Chemistry and Physics |date=18 February 2021 |volume=21 |issue=4 |pages=2407–2426 |doi=10.5194/acp-21-2407-2021 |bibcode=2021ACP....21.2407S |doi-access=free }}</ref> Presence of such smoke, soot, and/or brown oily deposits during a fire indicates a possible hazardous situation, as the atmosphere may be saturated with combustible pyrolysis products with concentration above the upper [[flammability limit]], and sudden inrush of air can cause [[flashover]] or [[backdraft]].<ref>{{cite book |last=Fire |first=Frank L. |year=2009 |title=The Common Sense Approach to Hazardous Materials |url=https://books.google.com/books?id=6AGGHbXVpSsC&q=pyrolysis+flashover+backdraft |publisher=Fire Engineering Books |page=129 |isbn=978-0-912212-11-1 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234022/https://books.google.com/books?id=6AGGHbXVpSsC&q=pyrolysis+flashover+backdraft |url-status=live }}</ref> |
||
Presence of sulfur can lead to formation of gases like hydrogen sulfide, [[carbonyl sulfide]], sulfur dioxide, [[carbon disulfide]], and [[thiol]]s; especially thiols tend to get adsorbed on surfaces and produce a lingering odor even long after the fire. Partial oxidation of the released hydrocarbons yields in a wide palette of other compounds: [[aldehyde]]s (e.g. [[formaldehyde]], [[acrolein]], and [[furfural]]), ketones, [[alcohols]] (often aromatic, e.g. [[phenol]], [[guaiacol]], [[syringol]], [[catechol]], and [[cresol]]s), [[carboxylic acid]]s ([[formic acid]], [[acetic acid]], etc.).{{citation needed|date=January 2022}} |
Presence of sulfur can lead to formation of gases like hydrogen sulfide, [[carbonyl sulfide]], sulfur dioxide, [[carbon disulfide]], and [[thiol]]s; especially thiols tend to get adsorbed on surfaces and produce a lingering odor even long after the fire. Partial oxidation of the released hydrocarbons yields in a wide palette of other compounds: [[aldehyde]]s (e.g. [[formaldehyde]], [[acrolein]], and [[furfural]]), ketones, [[alcohols]] (often aromatic, e.g. [[phenol]], [[guaiacol]], [[syringol]], [[catechol]], and [[cresol]]s), [[carboxylic acid]]s ([[formic acid]], [[acetic acid]], etc.).{{citation needed|date=January 2022}} |
||
The visible [[Atmospheric particulate matter|particulate matter]] in such |
The visible [[Atmospheric particulate matter|particulate matter]] in such smokes is most commonly composed of [[carbon]] ([[soot]]). Other particulates may be composed of drops of condensed tar, or solid particles of ash. The presence of metals in the fuel yields particles of metal [[oxide]]s. Particles of inorganic [[salt (chemistry)|salts]] may also be formed, e.g. [[ammonium sulfate]], [[ammonium nitrate]], or [[sodium chloride]]. Inorganic salts present on the surface of the soot particles may make them [[hydrophilic]]. Many organic compounds, typically the [[aromatic hydrocarbon]]s, may be also [[adsorb]]ed on the surface of the solid particles. Metal oxides can be present when metal-containing fuels are burned, e.g. [[solid rocket]] fuels containing [[aluminium]]. [[Depleted uranium]] projectiles after impacting the target ignite, producing particles of [[uranium oxide]]s. [[Magnetic]] particles, spherules of [[magnetite]]-like [[ferrous ferric oxide]], are present in coal smoke; their increase in deposits after 1860 marks the beginning of the Industrial Revolution.<ref>{{cite journal |last1=Oldfield |first1=F. |last2=Tolonen |first2=K. |last3=Thompson |first3=R. |jstor=4312673 |title=History of Particulate Atmospheric Pollution from Magnetic Measurements in Dated Finnish Peat Profiles |name-list-style=amp |journal=Ambio |volume=10 |issue=4 |year=1981 |page=185}}</ref> (Magnetic iron oxide [[nanoparticle]]s can be also produced in the smoke from [[meteorite]]s burning in the atmosphere.)<ref>{{cite journal |last1=Lanci |first1=L. |last2=Kent |first2=D. V. |title=Meteoric smoke fallout revealed by superparamagnetism in Greenland ice |journal=Geophysical Research Letters |date=2006 |volume=33 |issue=13 |pages=L13308 |doi=10.1029/2006GL026480 |bibcode=2006GeoRL..3313308L |doi-access=free }}</ref> Magnetic [[remanence]], [[paleomagnetism|recorded]] in the iron oxide particles, indicates the strength of Earth's magnetic field when they were cooled beyond their [[Curie temperature]]; this can be used to distinguish magnetic particles of terrestrial and meteoric origin.<ref>{{cite journal |last1=Suavet |first1=C. |last2=Gattacceca |first2=J. |last3=Rochette |first3=P. |last4=Perchiazzi |first4=N. |last5=Folco |first5=L. |last6=Duprat |first6=J. |last7=Harvey |first7=R. P. |title=Magnetic properties of micrometeorites |journal=Journal of Geophysical Research |date=4 April 2009 |volume=114 |issue=B4 |pages=B04102 |doi=10.1029/2008JB005831 |bibcode=2009JGRB..114.4102S |url=http://hal.in2p3.fr/in2p3-00684671/file/2008JB005831.pdf |access-date=25 January 2022 |archive-date=5 February 2022 |archive-url=https://web.archive.org/web/20220205105731/http://hal.in2p3.fr/in2p3-00684671/file/2008JB005831.pdf |url-status=live }}</ref> [[Fly ash]] is composed mainly of [[silica]] and [[calcium oxide]]. [[Cenosphere]]s are present in smoke from liquid hydrocarbon fuels. Minute metal particles produced by [[abrasion (mechanical)|abrasion]] can be present in engine smokes. [[Amorphous silica]] particles are present in smokes from burning [[silicone]]s; small proportion of [[silicon nitride]] particles can be formed in fires with insufficient oxygen. The silica particles have about 10 nm size, clumped to 70–100 nm aggregates and further agglomerated to chains.<ref name="flamsmokcable">{{cite book |author=National Research Council (U.S.). Task Force on Flammability, Smoke, Toxicity and Corrosive Gases of Electric Cable Materials |title=Flammability, smoke, toxicity, and corrosive gases of electric cable materials: report of the Task Force on Flammability, Smoke, Toxicity, and Corrosive Gases of Electric Cable Materials, National Materials Advisory Board, Commission on Sociotechnical Systems, National Research Council |url=https://books.google.com/books?id=6WYrAAAAYAAJ&pg=PA107|year=1978 |publisher=National Academies |pages=107– |id=NAP:15488}}</ref> Radioactive particles may be present due to traces of [[uranium]], [[thorium]], or other [[radionuclide]]s in the fuel; [[hot particle]]s can be present in case of fires during [[nuclear accident]]s (e.g. [[Chernobyl disaster]]) or [[nuclear war]]. |
||
|
Smoke particulates, like other aerosols, are categorized into three modes based on particle size: |
||
* '''nuclei mode''', with [[geometric mean]] radius between 2.5 and 20 nm, likely forming by condensation of carbon [[Moiety (chemistry)|moieties]]. |
* '''nuclei mode''', with [[geometric mean]] radius between 2.5 and 20 nm, likely forming by condensation of carbon [[Moiety (chemistry)|moieties]]. |
||
* '''[[accumulation mode]]''', ranging between 75 and 250 nm and formed by coagulation of nuclei mode particles |
* '''[[accumulation mode]]''', ranging between 75 and 250 nm and formed by coagulation of nuclei mode particles |
||
* '''[[coarse mode]]''', with particles in micrometer range |
* '''[[coarse mode]]''', with particles in micrometer range |
||
Most of the smoke |
Most of the smoke material is primarily in coarse particles. Those undergo rapid [[dry precipitation]], and the smoke damage in more distant areas outside of the room where the fire occurs is therefore primarily mediated by the smaller particles.<ref name="physproppoly"/> |
||
Aerosol of particles beyond visible size is an early indicator of materials in a preignition stage of a fire.<ref name="flamsmokcable"/> |
Aerosol of particles beyond visible size is an early indicator of materials in a preignition stage of a fire.<ref name="flamsmokcable"/> |
||
Burning of hydrogen-rich fuel produces [[water vapor]]; this results in smoke |
Burning of hydrogen-rich fuel produces [[water vapor]]; this results in smoke containing droplets of water. In absence of other color sources (nitrogen oxides, particulates...), such smoke is white and [[cloud]]-like. |
||
|
Smoke emissions may contain characteristic trace elements. [[Vanadium]] is present in emissions from [[oil]] fired power plants and [[Oil refinery|refineries]]; oil plants also emit some [[nickel]]. Coal combustion [[Fossil fuel power plant#Environmental impacts|produces emissions]] containing [[aluminium]], [[arsenic]], [[chromium]], [[cobalt]], [[copper]], [[iron]], [[mercury (element)|mercury]], [[selenium]], and [[uranium]]. |
||
Traces of vanadium in high-temperature combustion products form droplets of molten [[vanadate]]s. These attack the [[Passivation (chemistry)|passivation layer]]s on metals and cause [[high temperature corrosion]], which is a concern especially for [[internal combustion engine]]s. Molten [[sulfate]] and [[lead]] particulates also have such effect. |
Traces of vanadium in high-temperature combustion products form droplets of molten [[vanadate]]s. These attack the [[Passivation (chemistry)|passivation layer]]s on metals and cause [[high temperature corrosion]], which is a concern especially for [[internal combustion engine]]s. Molten [[sulfate]] and [[lead]] particulates also have such effect. |
||
Some components of smoke |
Some components of smoke are characteristic of the combustion source. [[Guaiacol]] and its derivatives are products of pyrolysis of [[lignin]] and are characteristic of [[wood]] smoke; other markers are [[syringol]] and derivates, and other [[methoxy]] [[phenol]]s. [[Retene]], a product of pyrolysis of [[conifer]] trees, is an indicator of [[forest fire]]s. [[Levoglucosan]] is a pyrolysis product of [[cellulose]]. [[Hardwood]] vs [[softwood]] smokes differ in the ratio of guaiacols/syringols. Markers for vehicle exhaust include [[polycyclic aromatic hydrocarbon]]s, [[hopane]]s, [[sterane]]s, and specific nitroarenes (e.g. [[1-nitropyrene]]). The ratio of hopanes and steranes to elemental carbon can be used to distinguish between emissions of gasoline and diesel engines.<ref>{{cite web |url=http://www.wrapair.org/APACE/SPECIATION/Synopsis_topic7.htm |title=Organic Speciation International Workshop Synthesis_topic7 |publisher=Wrapair.org |access-date=19 February 2010 |archive-date=26 July 2017 |archive-url=https://web.archive.org/web/20170726052050/https://www.wrapair.org/APACE/SPECIATION/Synopsis_topic7.htm |url-status=live }}</ref> |
||
Many compounds can be associated with particulates; whether by being [[adsorption|adsorbed]] on their surfaces, or by being dissolved in liquid droplets. Hydrogen chloride is well absorbed in the soot particles.<ref name="physproppoly"/> |
Many compounds can be associated with particulates; whether by being [[adsorption|adsorbed]] on their surfaces, or by being dissolved in liquid droplets. Hydrogen chloride is well absorbed in the soot particles.<ref name="physproppoly"/> |
||
Inert particulate matter can be disturbed and entrained into the smoke |
Inert particulate matter can be disturbed and entrained into the smoke. Of particular concern are particles of [[asbestos]]. |
||
Deposited [[hot particle]]s of [[radioactive fallout]] and bioaccumulated radioisotopes can be reintroduced into the atmosphere by [[wildfire]]s and [[forest fire]]s; this is a concern in e.g. the [[Zone of alienation]] containing contaminants from the [[Chernobyl disaster]]. |
Deposited [[hot particle]]s of [[radioactive fallout]] and bioaccumulated radioisotopes can be reintroduced into the atmosphere by [[wildfire]]s and [[forest fire]]s; this is a concern in e.g. the [[Zone of alienation]] containing contaminants from the [[Chernobyl disaster]]. |
||
Polymers are a significant source of smoke |
Polymers are a significant source of smoke. Aromatic [[side group]]s, e.g. in [[polystyrene]], enhance generation of smoke. Aromatic groups integrated in the polymer backbone produce less smoke, likely due to significant [[charring]]. Aliphatic polymers tend to generate the least smoke, and are non-self-extinguishing. However presence of additives can significantly increase smoke formation. Phosphorus-based and halogen-based [[flame retardant]]s decrease production of smoke. Higher degree of [[cross-link]]ing between the polymer chains has such effect too.<ref>{{cite book |last1=Krevelen |first1=D.W. van |last2=Nijenhuis |first2=Klaas te |page=864 |url=https://books.google.com/books?id=bzRKwjZeQ2kC&pg=PA864 |title=Properties of Polymers: Their Correlation with Chemical Structure; Their Numerical Estimation and Prediction from Additive Group Contributions |publisher=Elsevier |year=2009 |isbn=978-0-08-054819-7 |access-date=25 September 2016 |archive-date=14 July 2020 |archive-url=https://web.archive.org/web/20200714131333/https://books.google.com/books?id=bzRKwjZeQ2kC&pg=PA864 |url-status=live }}</ref> |
||
===Visible and invisible particles of combustion=== |
===Visible and invisible particles of combustion=== |
||
{{unreferenced section|date=January 2022}} |
{{unreferenced section|date=January 2022}} |
||
[[File:Wildfiretopanga.jpg|thumb| |
[[File:Wildfiretopanga.jpg|thumb|Smoke from a [[wildfire]]]] |
||
[[File:Muizenberg Mountian Fires in Cape Town of 2015.jpg|thumbnail| |
[[File:Muizenberg Mountian Fires in Cape Town of 2015.jpg|thumbnail|Smoke rising up from the smoldering remains of a recently extingished mountain fire in South Africa]] |
||
The [[naked eye]] detects particle sizes greater than 7 µm ([[micrometres]]). [[Visibility|Visible]] particles emitted from a fire are referred to as smoke |
The [[naked eye]] detects particle sizes greater than 7 µm ([[micrometres]]). [[Visibility|Visible]] particles emitted from a fire are referred to as smoke. [[Invisibility|Invisible]] particles are generally referred to as gas or fumes. This is best illustrated when [[Toast (food)|toast]]ing bread in a toaster. As the bread heats up, the products of combustion increase in size. The fumes initially produced are invisible but become visible if the toast is burnt. |
||
An [[ionization chamber]] type [[smoke |
An [[ionization chamber]] type [[smoke detector]] is technically a product of combustion detector, not a smoke detector. Ionization chamber type smoke detectors detect particles of combustion that are invisible to the naked eye. This explains why they may frequently [[false alarm]] from the fumes emitted from the red-hot heating elements of a toaster, before the presence of visible smoke, yet they may fail to activate in the early, low-heat [[smoldering]] stage of a fire. |
||
|
Smoke from a typical house fire contains hundreds of different chemicals and fumes. As a result, the damage caused by the smoke can often exceed that caused by the actual heat of the fire. In addition to the physical damage caused by the smoke of a [[fire]] – which manifests itself in the form of stains – is the often even harder to eliminate problem of a smoky odor. Just as there are contractors that specialize in rebuilding/repairing homes that have been damaged by fire and smoke, [[fabric restoration]] companies specialize in restoring fabrics that have been damaged in a fire. |
||
==Dangers== |
==Dangers== |
||
|
Smoke from oxygen-deprived fires contains a significant concentration of compounds that are flammable. A cloud of smoke, in contact with atmospheric oxygen, therefore has the potential of being ignited – either by another open flame in the area, or by its own temperature. This leads to effects like [[backdraft]] and [[flashover]]. [[Smoke inhalation]] is also a danger of smoke that can cause serious injury and death.{{citation needed|date=January 2022}} |
||
[[Image:Fish processing hub.jpg|thumb|left|Processing fish while being exposed to smoke |
[[Image:Fish processing hub.jpg|thumb|left|Processing fish while being exposed to smoke]] |
||
Many compounds of smoke |
Many compounds of smoke from fires are highly toxic and/or irritating. The most dangerous is [[carbon monoxide]] leading to [[carbon monoxide poisoning]], sometimes with the additive effects of [[hydrogen cyanide]] and [[phosgene]]. Smoke inhalation can therefore quickly lead to incapacitation and loss of consciousness. Sulfur oxides, hydrogen chloride and hydrogen fluoride in contact with moisture form [[sulfuric acid|sulfuric]], [[hydrochloric acid|hydrochloric]] and [[hydrofluoric acid]], which are corrosive to both lungs and materials. When asleep the nose does not sense smoke nor does the brain, but the body will wake up if the lungs become enveloped in smoke and the brain will be stimulated and the person will be awoken. This does not work if the person is incapacitated or under the influence of drugs and/or alcohol.{{citation needed|date=January 2022}} |
||
[[File:WTC smoking |
[[File:WTC smoking on 9-11.jpeg|thumb|World Trade Center on fire after terrorists flew planes into the buildings on September 11, 2001]] |
||
[[Cigarette smoke |
[[Cigarette smoke]] is a major modifiable risk factor for [[lung disease]], [[heart disease]], and many [[cancer]]s. Smoke can also be a component of ambient air pollution due to the burning of coal in power plants, forest fires or other sources, although the concentration of pollutants in ambient air is typically much less than that in cigarette smoke. One day of exposure to PM2.5 at a concentration of 880 μg/m3, such as occurs in Beijing, China, is the equivalent of smoking one or two cigarettes in terms of particulate inhalation by weight.<ref>{{cite journal |last1=Pope |first1=C. Arden |last2=Burnett |first2=Richard T. |last3=Turner |first3=Michelle C. |last4=Cohen |first4=Aaron |last5=Krewski |first5=Daniel |last6=Jerrett |first6=Michael |last7=Gapstur |first7=Susan M. |last8=Thun |first8=Michael J. |title=Lung Cancer and Cardiovascular Disease Mortality Associated with Ambient Air Pollution and Cigarette Smoke: Shape of the Exposure–Response Relationships |journal=Environmental Health Perspectives |date=November 2011 |volume=119 |issue=11 |pages=1616–1621 |doi=10.1289/ehp.1103639 |pmid=21768054 |pmc=3226505 }}</ref><ref>{{cite web |last1=St Cyr, MD |first1=Richard |title=Is PM2.5 From Air Pollution The Same As From Smoking? |url=http://www.myhealthbeijing.com/pollution/is-pm2-5-from-air-pollution-the-same-as-from-smoking/ |website=My Health Beijing |date=16 January 2013 |access-date=16 September 2015 |archive-date=16 February 2021 |archive-url=https://web.archive.org/web/20210216144621/http://www.myhealthbeijing.com/pollution/is-pm2-5-from-air-pollution-the-same-as-from-smoking/ |url-status=live }}</ref> The analysis is complicated, however, by the fact that the organic compounds present in various ambient particulates may have a higher carcinogenicity than the compounds in cigarette smoke particulates.<ref>{{cite journal |last1=Cupitt |first1=L T |last2=Glen |first2=W G |last3=Lewtas |first3=J |title=Exposure and risk from ambient particle-bound pollution in an airshed dominated by residential wood combustion and mobile sources. |journal=Environmental Health Perspectives |date=October 1994 |volume=102 |issue=suppl 4 |pages=75–84 |doi=10.1289/ehp.94102s475 |pmid=7529707 |pmc=1566933 }}</ref> Secondhand tobacco smoke is the combination of both sidestream and mainstream smoke emissions from a burning tobacco product. These emissions contain more than 50 carcinogenic chemicals. According to the United States [[Surgeon General of the United States|Surgeon General]]'s 2006 report on the subject, "Short exposures to secondhand [tobacco] smoke can cause blood platelets to become stickier, damage the lining of blood vessels, decrease coronary flow velocity reserves, and reduce heart variability, potentially increasing the risk of a heart attack".<ref>{{cite web |last=General |first=Surgeon |title=The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General |publisher=U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health |url=https://www.surgeongeneral.gov/library/reports/secondhandsmoke/fullreport.pdf |access-date=27 February 2017 |archive-date=1 February 2017 |archive-url=https://web.archive.org/web/20170201132734/https://www.surgeongeneral.gov/library/reports/secondhandsmoke/fullreport.pdf }}</ref> The American Cancer Society lists "heart disease, lung infections, increased asthma attacks, middle ear infections, and low birth weight" as ramifications of smoker's emission.<ref>{{cite web|title=Secondhand Smoke|url=http://www.cancer.org/Cancer/CancerCauses/TobaccoCancer/secondhand-smoke|publisher=American Cancer Society|access-date=11 January 2011|archive-date=7 January 2017|archive-url=https://web.archive.org/web/20170107025355/http://www.cancer.org/cancer/cancercauses/tobaccocancer/secondhand-smoke|url-status=live}}</ref> |
||
[[File: |
[[File:Smoke from forest fires Sheremetyevo 20100807 01.JPG|thumb|Reduced visibility due to wildfire smoke in [[Sheremetyevo International Airport|Sheremetyevo Airport, Moscow]], 7 August 2010]] |
||
[[File:Red smoke |
[[File:Red smoke from Army display parachutist landing 29Sept2018 arp.jpg|thumb|Red smoke carried by a [[parachute|parachutist]] of the UK [[Lightning Bolts Army Parachute Display Team]] ]] |
||
|
Smoke can obscure visibility, impeding occupant exiting from fire areas. In fact, the poor visibility due to the smoke that was in the [[Worcester Cold Storage Warehouse fire]] in [[Worcester, Massachusetts]] was the reason why the trapped rescue firefighters could not evacuate the building in time. Because of the striking similarity that each floor shared, the dense smoke caused the firefighters to become disoriented.<ref>{{cite web|url=http://www.telegram.com/static/fire/|title=telegram.com – Warehouse Tragedy|access-date=28 July 2007|archive-date=29 August 2013|archive-url=https://web.archive.org/web/20130829100805/http://www.telegram.com/static/fire/}}</ref> |
||
===Corrosion=== |
===Corrosion=== |
||
|
Smoke can contain a wide variety of chemicals, many of them aggressive in nature. Examples are [[hydrochloric acid]] and [[hydrobromic acid]], produced from [[halogen]]-containing [[plastic]]s and [[fire retardant]]s, [[hydrofluoric acid]] released by [[pyrolysis]] of [[fluorocarbon]] [[fire suppression agent]]s, [[sulfuric acid]] from burning of [[sulfur]]-containing materials, [[nitric acid]] from high-temperature fires where [[nitrous oxide]] gets formed, [[phosphoric acid]] and [[antimony]] compounds from P and Sb based fire retardants, and many others. Such [[corrosion]] is not significant for structural materials, but delicate structures, especially [[microelectronics]], are strongly affected. Corrosion of [[circuit board]] traces, penetration of aggressive chemicals through the casings of parts, and other effects can cause an immediate or gradual deterioration of parameters or even premature (and often delayed, as the corrosion can progress over long time) failure of equipment subjected to smoke. Many smoke components are also [[electrically conductive]]; deposition of a conductive layer on the circuits can cause [[crosstalk]]s and other deteriorations of the operating parameters or even cause short circuits and total failures. [[Electrical contact]]s can be affected by corrosion of surfaces, and by deposition of [[soot]] and other conductive particles or nonconductive layers on or across the contacts. Deposited particles may adversely affect the performance of [[optoelectronics]] by absorbing or scattering the light beams.{{citation needed|date=May 2021}} |
||
Corrosivity of smoke |
Corrosivity of smoke produced by materials is characterized by the corrosion index (CI), defined as material loss rate (angstrom/minute) per amount of material gasified products (grams) per volume of air (m<sup>3</sup>). It is measured by exposing strips of metal to flow of combustion products in a test tunnel. Polymers containing halogen and [[hydrogen]] ([[polyvinyl chloride]], [[polyolefin]]s with halogenated additives, etc.) have the highest CI as the corrosive acids are formed directly with water produced by the combustion, polymers containing halogen only (e.g. [[polytetrafluoroethylene]]) have lower CI as the formation of acid is limited to reactions with airborne humidity, and halogen-free materials (polyolefins, [[wood]]) have the lowest CI.<ref name="physproppoly">{{cite book |last=Mark |first=James E. |url=https://books.google.com/books?id=fZl7q7UgEXkC&pg=PA909 |title=Physical properties of polymers handbook |publisher=Springer |year=2006 |isbn=978-0-387-31235-4 |access-date=25 September 2016 |archive-date=2 August 2020 |archive-url=https://web.archive.org/web/20200802054508/https://books.google.com/books?id=fZl7q7UgEXkC&pg=PA909 |url-status=live }}</ref> However, some halogen-free materials can also release significant amount of corrosive products.<ref>{{cite book |url=https://books.google.com/books?id=Sj4vSgMdimAC&pg=PA43 |page=43 |title=Optoelectronics for data communication |author=Ronald C. Lasky |author2=Ronald Lasky |author3=Ulf L. Österberg |author4=Daniel P. Stigliani |publisher=Academic Press |year=1995 |isbn=978-0-12-437160-6 |access-date=25 September 2016 |archive-date=16 July 2020 |archive-url=https://web.archive.org/web/20200716062706/https://books.google.com/books?id=Sj4vSgMdimAC&pg=PA43 |url-status=live }}</ref> |
||
|
Smoke damage to electronic equipment can be significantly more extensive than the fire itself. [[Electrical cable|Cable]] fires are of special concern; [[low smoke zero halogen]] materials are preferable for cable insulation.<ref>{{Cite journal |last1=Szultka |first1=Seweryn |last2=Czapp |first2=Stanislaw |last3=Tomaszewski |first3=Adam |last4=Ullah |first4=Hayat |date=February 2023 |title=Evaluation of Fire Hazard in Electrical Installations Due to Unfavorable Ambient Thermal Conditions |journal=Fire |language=en |volume=6 |issue=2 |page=41 |doi=10.3390/fire6020041 |issn=2571-6255 |doi-access=free }}</ref> |
||
When smoke |
When smoke comes into contact with the surface of any substance or structure, the chemicals contained in it are transferred to it. The corrosive properties of the chemicals cause the substance or structure to decompose at a rapid rate. Certain materials or structures absorb these chemicals, which is why clothing, unsealed surfaces, potable water, piping, wood, etc., are replaced in most cases of structural fires.{{citation needed|date=January 2022}} |
||
===Health effects of wood smoke |
===Health effects of wood smoke=== |
||
{{See also|Wildfire#Health effects}} |
{{See also|Wildfire#Health effects}} |
||
Wood smoke |
Wood smoke is a major source of [[air pollution]],<ref name=NSWEPA>{{Cite web|url=https://www.epa.nsw.gov.au/your-environment/air/reducing-wood-smoke-emissions|title=reducing-wood-smoke-emissions|first=NSW|last=EPA|website=NSW Environment Protection Authority|access-date=2 May 2021|archive-date=19 April 2021|archive-url=https://web.archive.org/web/20210419152235/https://www.epa.nsw.gov.au/your-environment/air/reducing-wood-smoke-emissions|url-status=live}}</ref><ref name=AUENV>{{Cite web|url=http://www.environment.gov.au/|title=Department of Agriculture, Water and the Environment|website=Department of Agriculture, Water and the Environment|access-date=11 November 2008|archive-date=11 May 2019|archive-url=https://web.archive.org/web/20190511213226/http://environment.gov.au/|url-status=live}}</ref><ref name=EU>{{cite web|url=https://ec.europa.eu/environment/integration/research/newsalert/pdf/92na1_en.pdf|title=Wood smoke major source of pollution in winter|date=2007|website=ec.europa.eu|access-date=2021-05-02|archive-date=8 December 2020|archive-url=https://web.archive.org/web/20201208212134/https://ec.europa.eu/environment/integration/research/newsalert/pdf/92na1_en.pdf|url-status=live}}</ref><ref name=WAHEALTH>{{cite web|url=https://healthywa.wa.gov.au/Articles/A_E/Air-pollution-and-health|title=Air pollution and health|website=healthywa.wa.gov.au|format=PDF|access-date=2021-05-02|archive-date=21 April 2021|archive-url=https://web.archive.org/web/20210421193721/https://healthywa.wa.gov.au/Articles/A_E/Air-pollution-and-health|url-status=live}}</ref> especially [[particulate pollution]],<ref name=AUENV/> pollution by [[polycyclic aromatic hydrocarbons]] (PAHs)<ref name="10.5194/acp-21-17865-2021">{{cite journal |last1=Tsiodra |first1=Irini |last2=Grivas |first2=Georgios |last3=Tavernaraki |first3=Kalliopi |last4=Bougiatioti |first4=Aikaterini |last5=Apostolaki |first5=Maria |last6=Paraskevopoulou |first6=Despina |last7=Gogou |first7=Alexandra |last8=Parinos |first8=Constantine |last9=Oikonomou |first9=Konstantina |last10=Tsagkaraki |first10=Maria |last11=Zarmpas |first11=Pavlos |last12=Nenes |first12=Athanasios |last13=Mihalopoulos |first13=Nikolaos |title=Annual exposure to polycyclic aromatic hydrocarbons in urban environments linked to wintertime wood-burning episodes |journal=Atmospheric Chemistry and Physics |date=7 December 2021 |volume=21 |issue=23 |pages=17865–17883 |doi=10.5194/acp-21-17865-2021 |bibcode=2021ACP....2117865T |s2cid=245103794 |language=English |issn=1680-7316|doi-access=free }}</ref> and [[volatile organic compounds]] (VOCs)<ref name=AUENV/>{{Better source needed|date=January 2022}} such as [[formaldehyde]].<ref name=USEPA>{{Cite web|url=https://www.epa.gov/burnwise/wood-smoke-and-your-health|title=Wood Smoke and Your Health|first=OAR|last=US EPA|date=May 28, 2013|website=US EPA|access-date=2 May 2021|archive-date=14 May 2021|archive-url=https://web.archive.org/web/20210514175128/https://www.epa.gov/burnwise/wood-smoke-and-your-health|url-status=live}}</ref> |
||
In the [[United Kingdom]] domestic combustion, especially for industrial uses, is the largest single source of [[PM2.5]] annually.<ref>{{cite journal |last1=Hawkes |first1=N. |title=Air pollution in UK: the public health problem that won't go away |journal=BMJ |date=22 May 2015 |volume=350 |issue=may22 1 |pages=h2757 |doi=10.1136/bmj.h2757 |pmid=26001592 |s2cid=40717317 }}</ref><ref>{{Cite web|last=Carrington|first=Damian|date=2021-02-16|title=Wood burning at home now biggest cause of UK particle pollution|url=http://www.theguardian.com/environment/2021/feb/16/home-wood-burning-biggest-cause-particle-pollution-fires|access-date=2022-02-13|website=The Guardian|language=en|archive-date=27 December 2022|archive-url=https://web.archive.org/web/20221227162919/https://www.theguardian.com/environment/2021/feb/16/home-wood-burning-biggest-cause-particle-pollution-fires|url-status=live}}</ref> In some towns and cities in [[New South Wales]], wood smoke |
In the [[United Kingdom]] domestic combustion, especially for industrial uses, is the largest single source of [[PM2.5]] annually.<ref>{{cite journal |last1=Hawkes |first1=N. |title=Air pollution in UK: the public health problem that won't go away |journal=BMJ |date=22 May 2015 |volume=350 |issue=may22 1 |pages=h2757 |doi=10.1136/bmj.h2757 |pmid=26001592 |s2cid=40717317 }}</ref><ref>{{Cite web|last=Carrington|first=Damian|date=2021-02-16|title=Wood burning at home now biggest cause of UK particle pollution|url=http://www.theguardian.com/environment/2021/feb/16/home-wood-burning-biggest-cause-particle-pollution-fires|access-date=2022-02-13|website=The Guardian|language=en|archive-date=27 December 2022|archive-url=https://web.archive.org/web/20221227162919/https://www.theguardian.com/environment/2021/feb/16/home-wood-burning-biggest-cause-particle-pollution-fires|url-status=live}}</ref> In some towns and cities in [[New South Wales]], wood smoke may be responsible for 60% of fine particle air pollution in the winter.<ref name="auto">{{Cite web|url=https://www.health.nsw.gov.au/environment/factsheets/Pages/wood-smoke.aspx|title=Wood burning heaters and your health - Fact sheets|website=www.health.nsw.gov.au|access-date=2 May 2021|archive-date=9 November 2022|archive-url=https://web.archive.org/web/20221109204058/https://www.health.nsw.gov.au/environment/factsheets/Pages/wood-smoke.aspx|url-status=live}}</ref> A year-long sampling campaign in Athens, Greece found a third (31%) of PAH urban air pollution to be caused by wood-burning, roughly as much as that of [[Diesel locomotive|diesel]] and [[Crude oil|oil]] (33%) and [[gasoline]] (29%). It also found that wood-burning is responsible for nearly half (43%) of annual PAH lung cancer-risk compared to the other sources and that wintertime PAH levels were 7 times higher than in other seasons, presumably due to an increased use of [[fireplace]]s and heaters. The largest exposure events are periods during the winter with reduced atmospheric dispersion to dilute the accumulated pollution , in particular due to the low [[wind]] speeds.<!--<ref>{{cite news |title=Wood burners cause nearly half of urban air pollution cancer risk – study |url=https://www.theguardian.com/environment/2021/dec/17/wood-burners-urban-air-pollution-cancer-risk-study |access-date=16 January 2022 |work=The Guardian |date=17 December 2021 |language=en}}</ref>--><ref name="10.5194/acp-21-17865-2021"/> Research conducted about biomass burning in 2015, estimated that 38% of European total particulate pollution emissions are composed of domestic wood burning.<ref>{{Cite journal |last1=Sigsgaard |first1=Torben |last2=Forsberg |first2=Bertil |last3=Annesi-Maesano |first3=Isabella |last4=Blomberg |first4=Anders |last5=Bølling |first5=Anette |last6=Boman |first6=Christoffer |last7=Bønløkke |first7=Jakob |last8=Brauer |first8=Michael |last9=Bruce |first9=Nigel |last10=Héroux |first10=Marie-Eve |last11=Hirvonen |first11=Maija-Riitta |date=2015-09-24 |title=Health impacts of anthropogenic biomass burning in the developed world |url=https://erj.ersjournals.com/content/early/2015/09/24/13993003.01865-2014 |journal=European Respiratory Journal |volume=46 |issue=6 |pages=1577–1588 |language=en |doi=10.1183/13993003.01865-2014 |issn=0903-1936 |pmid=26405285|s2cid=41697986 |doi-access=free }}</ref> |
||
Wood smoke |
Wood smoke (for example from [[wildfire]]s or wood ovens) can cause lung damage,<ref>{{Cite web|url=https://www.pca.state.mn.us/air/wood-smoke-and-your-health|title=Wood smoke and your health|date=November 16, 2009|website=Minnesota Pollution Control Agency|access-date=2 May 2021|archive-date=14 May 2021|archive-url=https://web.archive.org/web/20210514231826/https://www.pca.state.mn.us/air/wood-smoke-and-your-health|url-status=live}}</ref><ref>{{Cite web|url=https://www.iatp.org/news/wood-stove-pollution-is-a-burning-issue|title=Wood stove pollution is a burning issue|website=www.iatp.org|access-date=2 May 2021|archive-date=9 December 2020|archive-url=https://web.archive.org/web/20201209025928/https://www.iatp.org/news/wood-stove-pollution-is-a-burning-issue|url-status=live}}</ref> artery damage and DNA damage<ref>{{cite journal |last1=Danielsen |first1=Pernille Høgh |last2=Møller |first2=Peter |last3=Jensen |first3=Keld Alstrup |last4=Sharma |first4=Anoop Kumar |last5=Wallin |first5=Håkan |last6=Bossi |first6=Rossana |last7=Autrup |first7=Herman |last8=Mølhave |first8=Lars |last9=Ravanat |first9=Jean-Luc |last10=Briedé |first10=Jacob Jan |last11=de Kok |first11=Theo Martinus |last12=Loft |first12=Steffen |title=Oxidative Stress, DNA Damage, and Inflammation Induced by Ambient Air and Wood Smoke Particulate Matter in Human A549 and THP-1 Cell Lines |journal=Chemical Research in Toxicology |date=18 February 2011 |volume=24 |issue=2 |pages=168–184 |doi=10.1021/tx100407m |pmid=21235221 |s2cid=11668269 }}</ref> leading to cancer,<ref>{{cite journal |last1=Navarro |first1=Kathleen M. |last2=Kleinman |first2=Michael T. |last3=Mackay |first3=Chris E. |last4=Reinhardt |first4=Timothy E. |last5=Balmes |first5=John R. |last6=Broyles |first6=George A. |last7=Ottmar |first7=Roger D. |last8=Naher |first8=Luke P. |last9=Domitrovich |first9=Joseph W. |title=Wildland firefighter smoke exposure and risk of lung cancer and cardiovascular disease mortality |journal=Environmental Research |date=June 2019 |volume=173 |pages=462–468 |doi=10.1016/j.envres.2019.03.060 |pmid=30981117 |bibcode=2019ER....173..462N |s2cid=108987257 }}</ref><ref>{{Cite web|url=https://asthma.org.au/about-us/media/dont-underestimate-the-health-dangers-of-wood-smoke/|title=Don't underestimate the health dangers of wood smoke|access-date=2 May 2021|archive-date=26 March 2021|archive-url=https://web.archive.org/web/20210326062804/https://asthma.org.au/about-us/media/dont-underestimate-the-health-dangers-of-wood-smoke/|url-status=live}}</ref> other respiratory and lung disease and cardiovascular disease.<ref name="auto"/><ref>{{cite journal |last1=Bede-Ojimadu |first1=Onyinyechi |last2=Orisakwe |first2=Orish Ebere |title=Exposure to Wood Smoke and Associated Health Effects in Sub-Saharan Africa: A Systematic Review |journal=Annals of Global Health |date=20 March 2020 |volume=86 |issue=1 |page=32 |doi=10.5334/aogh.2725 |pmid=32211302 |pmc=7082829 |doi-access=free }}</ref> Air pollution, particulate matter and wood smoke may also cause brain damage because of particulates breaching the cardiovascular system and into the brain,<ref>{{Cite journal|title=News Feature: How air pollution threatens brain health|first=Lynne|last=Peeples|date=June 23, 2020|journal=Proceedings of the National Academy of Sciences|volume=117|issue=25|pages=13856–13860|doi=10.1073/pnas.2008940117|pmid=32493753|pmc=7322062|bibcode=2020PNAS..11713856P|doi-access=free}}</ref><ref>{{Cite web|url=http://www.theguardian.com/environment/2020/oct/06/air-pollution-particles-in-young-brains-linked-to-alzheimers-damage|title=Air pollution particles in young brains linked to Alzheimer's damage|date=October 6, 2020|website=The Guardian|access-date=22 January 2022|archive-date=22 January 2022|archive-url=https://web.archive.org/web/20220122001335/https://www.theguardian.com/environment/2020/oct/06/air-pollution-particles-in-young-brains-linked-to-alzheimers-damage|url-status=live}}</ref><ref>{{Cite web|url=https://www.the-scientist.com/features/air-pollution-may-damage-peoples-brains-66473|title=Air Pollution May Damage People's Brains|website=The Scientist Magazine®|access-date=2 May 2021|archive-date=12 May 2021|archive-url=https://web.archive.org/web/20210512060730/https://www.the-scientist.com/features/air-pollution-may-damage-peoples-brains-66473|url-status=live}}</ref><ref>{{Cite web|url=https://abc7news.com/2719037/|title=Stanford study shows wood smoke can harm the brain|date=December 2, 2017|website=ABC7 San Francisco|access-date=3 May 2021|archive-date=1 August 2021|archive-url=https://web.archive.org/web/20210801121207/https://abc7news.com/2719037/|url-status=live}}</ref> which can increase the risk of developmental disorders,<ref>{{cite journal |last1=Flores-Pajot |first1=Marie-Claire |last2=Ofner |first2=Marianna |last3=Do |first3=Minh T. |last4=Lavigne |first4=Eric |last5=Villeneuve |first5=Paul J. |title=Childhood autism spectrum disorders and exposure to nitrogen dioxide, and particulate matter air pollution: A review and meta-analysis |journal=Environmental Research |date=November 2016 |volume=151 |pages=763–776 |doi=10.1016/j.envres.2016.07.030 |pmid=27609410 |bibcode=2016ER....151..763F }}</ref><ref>{{cite journal |last1=Chun |first1=HeeKyoung |last2=Leung |first2=Cheryl |last3=Wen |first3=Shi Wu |last4=McDonald |first4=Judy |last5=Shin |first5=Hwashin H. |title=Maternal exposure to air pollution and risk of autism in children: A systematic review and meta-analysis |journal=Environmental Pollution |date=January 2020 |volume=256 |page=113307 |doi=10.1016/j.envpol.2019.113307 |pmid=31733973 |doi-access=free }}</ref><ref>{{cite journal |last1=Lam |first1=Juleen |last2=Sutton |first2=Patrice |last3=Kalkbrenner |first3=Amy |last4=Windham |first4=Gayle |last5=Halladay |first5=Alycia |last6=Koustas |first6=Erica |last7=Lawler |first7=Cindy |last8=Davidson |first8=Lisette |last9=Daniels |first9=Natalyn |last10=Newschaffer |first10=Craig |last11=Woodruff |first11=Tracey |title=A Systematic Review and Meta-Analysis of Multiple Airborne Pollutants and Autism Spectrum Disorder |journal=PLOS ONE |date=21 September 2016 |volume=11 |issue=9 |pages=e0161851 |doi=10.1371/journal.pone.0161851 |pmid=27653281 |pmc=5031428 |bibcode=2016PLoSO..1161851L |doi-access=free }}</ref><ref>{{cite journal |last1=Weisskopf |first1=Marc G. |last2=Kioumourtzoglou |first2=Marianthi-Anna |last3=Roberts |first3=Andrea L. |title=Air Pollution and Autism Spectrum Disorders: Causal or Confounded? |journal=Current Environmental Health Reports |date=December 2015 |volume=2 |issue=4 |pages=430–439 |doi=10.1007/s40572-015-0073-9 |pmid=26399256 |pmc=4737505 }}</ref> neurodegenerative disorders<ref>{{cite journal |last1=Fu |first1=Pengfei |last2=Yung |first2=Ken Kin Lam |title=Air Pollution and Alzheimer's Disease: A Systematic Review and Meta-Analysis |journal=Journal of Alzheimer's Disease |date=15 September 2020 |volume=77 |issue=2 |pages=701–714 |doi=10.3233/JAD-200483 |pmid=32741830 |s2cid=220942039 }}</ref><ref>{{cite journal |last1=Tsai |first1=Tsung-Lin |last2=Lin |first2=Yu-Ting |last3=Hwang |first3=Bing-Fang |last4=Nakayama |first4=Shoji F. |last5=Tsai |first5=Chon-Haw |last6=Sun |first6=Xian-Liang |last7=Ma |first7=Chaochen |last8=Jung |first8=Chau-Ren |title=Fine particulate matter is a potential determinant of Alzheimer's disease: A systemic review and meta-analysis |journal=Environmental Research |date=October 2019 |volume=177 |page=108638 |doi=10.1016/j.envres.2019.108638 |pmid=31421449 |bibcode=2019ER....177j8638T |s2cid=201057595 }}</ref> mental disorders,<ref name = suicide>{{cite journal |last1=Braithwaite |first1=Isobel |last2=Zhang |first2=Shuo |last3=Kirkbride |first3=James B. |last4=Osborn |first4=David P. J. |last5=Hayes |first5=Joseph F. |title=Air Pollution (Particulate Matter) Exposure and Associations with Depression, Anxiety, Bipolar, Psychosis and Suicide Risk: A Systematic Review and Meta-Analysis |journal=Environmental Health Perspectives |date=December 2019 |volume=127 |issue=12 |page=126002 |doi=10.1289/EHP4595 |pmid=31850801 |pmc=6957283 }}</ref><ref name = economic>{{cite journal |last1=Lu |first1=Jackson G |title=Air pollution: A systematic review of its psychological, economic, and social effects |journal=Current Opinion in Psychology |date=April 2020 |volume=32 |pages=52–65 |doi=10.1016/j.copsyc.2019.06.024 |pmid=31557706 |s2cid=199147061 }}</ref><ref name = suicidedep>{{cite journal |last1=Liu |first1=Qisijing |last2=Wang |first2=Wanzhou |last3=Gu |first3=Xuelin |last4=Deng |first4=Furong |last5=Wang |first5=Xueqin |last6=Lin |first6=Hualiang |last7=Guo |first7=Xinbiao |last8=Wu |first8=Shaowei |title=Association between particulate matter air pollution and risk of depression and suicide: a systematic review and meta-analysis |journal=Environmental Science and Pollution Research |date=February 2021 |volume=28 |issue=8 |pages=9029–9049 |doi=10.1007/s11356-021-12357-3 |pmid=33481201 |s2cid=231677095 }}</ref> and suicidal behavior,<ref name = suicide/><ref name = suicidedep/> although studies on the link between [[Depression (clinical)|depression]] and some air pollutants are not consistent.<ref>{{cite journal |last1=Fan |first1=Shu-Jun |last2=Heinrich |first2=Joachim |last3=Bloom |first3=Michael S. |last4=Zhao |first4=Tian-Yu |last5=Shi |first5=Tong-Xing |last6=Feng |first6=Wen-Ru |last7=Sun |first7=Yi |last8=Shen |first8=Ji-Chuan |last9=Yang |first9=Zhi-Cong |last10=Yang |first10=Bo-Yi |last11=Dong |first11=Guang-Hui |title=Ambient air pollution and depression: A systematic review with meta-analysis up to 2019 |journal=Science of the Total Environment |date=January 2020 |volume=701 |page=134721 |doi=10.1016/j.scitotenv.2019.134721 |pmid=31715478 |bibcode=2020ScTEn.701m4721F |s2cid=207944384 |url=https://push-zb.helmholtz-muenchen.de/frontdoor.php?source_opus=57348 |access-date=25 January 2022 |archive-date=31 March 2022 |archive-url=https://web.archive.org/web/20220331123210/https://push-zb.helmholtz-muenchen.de/frontdoor.php?source_opus=57348 |url-status=live }}</ref> At least one study has identified "the abundant presence in the human brain of magnetite nanoparticles that match precisely the high-temperature magnetite nanospheres, formed by combustion and/or friction-derived heating, which are prolific in urban, airborne particulate matter (PM)."<ref>{{cite journal |last1=Maher |first1=Barbara A. |last2=Ahmed |first2=Imad A. M. |last3=Karloukovski |first3=Vassil |last4=MacLaren |first4=Donald A. |last5=Foulds |first5=Penelope G. |last6=Allsop |first6=David |last7=Mann |first7=David M. A. |last8=Torres-Jardón |first8=Ricardo |last9=Calderon-Garciduenas |first9=Lilian |title=Magnetite pollution nanoparticles in the human brain |journal=Proceedings of the National Academy of Sciences |date=27 September 2016 |volume=113 |issue=39 |pages=10797–10801 |doi=10.1073/pnas.1605941113 |pmid=27601646 |pmc=5047173 |bibcode=2016PNAS..11310797M |doi-access=free }}</ref> Air pollution has also been linked to a range of other psychosocial problems.<ref name = economic/> |
||
==Measurement== |
==Measurement== |
||
As early as the 15th century [[Leonardo da Vinci]] commented at length on the difficulty of assessing smoke |
As early as the 15th century [[Leonardo da Vinci]] commented at length on the difficulty of assessing smoke, and distinguished between [[black carbon|black smoke]] (carbonized particles) and white 'smoke' which is not a smoke at all but merely a suspension of harmless water particulates.<ref>{{cite book |last=Sorensen |first=Roy |date=2016 |title=A Cabinet of Philosophical Curiosities: A Collection of Puzzles, Oddities, Riddles, and Dilemmas |url=https://books.google.com/books?id=tnblCwAAQBAJ&q=black+smoke+Leonardo+da+Vinci&pg=PT89 |publisher=Oxford University Press |page=89 |isbn=978-0-19-046863-7 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403234030/https://books.google.com/books?id=tnblCwAAQBAJ&q=black+smoke+Leonardo+da+Vinci&pg=PT89 |url-status=live }}</ref> |
||
|
Smoke from heating appliances is commonly measured in one of the following ways: |
||
'''In-line capture.''' A smoke |
'''In-line capture.''' A smoke sample is simply sucked through a filter which is weighed before and after the test and the mass of smoke found. This is the simplest and probably the most accurate method, but can only be used where the smoke concentration is slight, as the filter can quickly become blocked.<ref>{{cite book |last=Watson |first=Donna S. |date=8 March 2010 |title=Perioperative Safety |url=https://books.google.com/books?id=8r9JmFSW-8cC&q=in-line+capture+smoke&pg=PT543 |location=Amsterdam, Netherlands |publisher=Elsevier Health Sciences |isbn=978-0-323-06985-4 |access-date=20 October 2020 |archive-date=3 April 2023 |archive-url=https://web.archive.org/web/20230403233724/https://books.google.com/books?id=8r9JmFSW-8cC&q=in-line+capture+smoke&pg=PT543 |url-status=live }}</ref> |
||
The [[ASTM smoke |
The [[ASTM smoke pump]] is a simple and widely used method of in-line capture where a measured volume of smoke is pulled through a filter paper and the dark spot so formed is compared with a standard. |
||
'''Filter/dilution tunnel.''' A smoke |
'''Filter/dilution tunnel.''' A smoke sample is drawn through a tube where it is diluted with air, the resulting smoke/air mixture is then pulled through a filter and weighed. This is the internationally recognized method of measuring smoke from [[combustion]].<ref>{{Cite report |author=National Academies |date=1 January 1983 |title=Polycyclic Aromatic Hydrocarbons: Evaluation of Sources and Effects |url=https://books.google.com/books?id=tVArAAAAYAAJ&q=Filter%2Fdilution+tunnel&pg=SA1-PA5 |publisher=National Academies |page=4 }}</ref> |
||
'''Electrostatic precipitation.''' The smoke |
'''Electrostatic precipitation.''' The smoke is passed through an array of metal tubes which contain suspended wires. A (huge) electrical potential is applied across the tubes and wires so that the smoke particles become charged and are attracted to the sides of the tubes. This method can over-read by capturing harmless condensates, or under-read due to the insulating effect of the smoke. However, it is the necessary method for assessing volumes of smoke too great to be forced through a filter, i.e., from [[bituminous coal]]. |
||
'''[[Ringelmann scale]].''' A measure of smoke |
'''[[Ringelmann scale]].''' A measure of smoke color. Invented by Professor [[Maximilian Ringelmann]] in Paris in 1888, it is essentially a card with squares of black, white and shades of gray which is held up and the comparative grayness of the smoke judged. Highly dependent on light conditions and the skill of the observer it allocates a grayness number from 0 (white) to 5 (black) which has only a passing relationship to the actual quantity of smoke. Nonetheless, the simplicity of the Ringelmann scale means that it has been adopted as a standard in many countries. |
||
'''Optical scattering.''' A light beam is passed through the smoke |
'''Optical scattering.''' A light beam is passed through the smoke. A light detector is situated at an angle to the light source, typically at 90°, so that it receives only light reflected from passing particles. A measurement is made of the light received which will be higher as the concentration of smoke particles becomes higher. |
||
'''Optical obscuration.''' A light beam is passed through the smoke |
'''Optical obscuration.''' A light beam is passed through the smoke and a detector opposite measures the light. The more smoke particles are present between the two, the less light will be measured. |
||
'''Combined optical methods.''' There are various proprietary optical smoke |
'''Combined optical methods.''' There are various proprietary optical smoke measurement devices such as the '[[nephelometer]]' or the '[[aethalometer]]' which use several different optical methods, including more than one wavelength of light, inside a single instrument and apply an algorithm to give a good estimate of smoke. It has been claimed that these devices can differentiate types of smoke and so their probable source can be inferred, though this is disputed.<ref>{{cite journal |last1=Harrison & others |first1=Roy M |title=An evaluation of some issues regarding the use of aethalometers to measure woodsmoke concentrations |journal=Atmospheric Environment |date=26 August 2013 |volume=80 |pages=540–548 |url=http://pure-oai.bham.ac.uk/ws/files/16250444/An_Evaluation_of_Some_Issues_PostPrint.pdf |doi=10.1016/j.atmosenv.2013.08.026 |bibcode=2013AtmEn..80..540H |access-date=26 July 2019 |archive-date=7 March 2020 |archive-url=https://web.archive.org/web/20200307132206/http://pure-oai.bham.ac.uk/ws/files/16250444/An_Evaluation_of_Some_Issues_PostPrint.pdf |url-status=live }}</ref> |
||
'''Inference from [[carbon monoxide]].''' |
'''Inference from [[carbon monoxide]].''' Smoke is incompletely burned [[fuel]], carbon monoxide is incompletely burned carbon, therefore it has long been assumed that measurement of CO in [[flue gas]] (a cheap, simple and very accurate procedure) will provide a good indication of the levels of smoke. Indeed, several jurisdictions use CO measurement as the basis of [[smoke control]]. However it is far from clear how accurate the correspondence is. |
||
==Medicinal smoking |
==Medicinal smoking== |
||
{{single source|section|date=October 2023}} |
{{single source|section|date=October 2023}} |
||
Throughout recorded history, humans have used the smoke |
Throughout recorded history, humans have used the smoke of [[medicinal plant]]s to cure illness. A sculpture from [[Persepolis]] shows [[Darius the Great]] (522–486 BC), the king of [[Persia]], with two [[censer]]s in front of him for burning [[Peganum harmala]] and/or [[sandalwood]] [[Santalum album]], which was believed to protect the king from evil and disease. More than 300 plant species in 5 continents are used in smoke form for different diseases. As a method of [[drug administration]], smoking is important as it is a simple, inexpensive, but very effective method of extracting particles containing active agents. More importantly, generating smoke reduces the particle size to a microscopic scale thereby increasing the absorption of its active chemical principles.<ref>{{cite journal |doi=10.1016/j.jep.2006.09.005 |title=Medicinal smokes |year=2006 |last1=Mohagheghzadeh |first1=Abdolali |last2=Faridi |first2=Pouya |last3=Shams-Ardakani |first3=Mohammadreza |last4=Ghasemi |first4=Younes |journal=Journal of Ethnopharmacology |volume=108 |issue=2 |pages=161–84 |pmid=17030480}}</ref> |
||
==See also== |
==See also== |
||
| Line 134: | Line 134: | ||
==Sources== |
==Sources== |
||
*{{cite EB1911|wstitle= |
*{{cite EB1911|wstitle=Smoke |volume=25 |short=x}} |
||
==External links== |
==External links== |
||
*[https://phys.org/news/2019-09-inversions-valleys.html 'Fire inversions' lock smoke |
*[https://phys.org/news/2019-09-inversions-valleys.html 'Fire inversions' lock smoke in valleys] |
||
*[http://burningissues.org/ Burning Issues wood smoke |
*[http://burningissues.org/ Burning Issues wood smoke Site] |
||
*[http://erj.ersjournals.com/cgi/content/full/27/3/446 Shedding new light on wood smoke |
*[http://erj.ersjournals.com/cgi/content/full/27/3/446 Shedding new light on wood smoke] |
||
*[https://www.infographicview.com/things-you-need-to-know-about-smoke |
*[https://www.infographicview.com/things-you-need-to-know-about-smoke.html/ 7 things you need to know about smoke detectors] |
||
{{Commons}} |
{{Commons}} |
||
| Line 148: | Line 148: | ||
[[Category:Fire]] |
[[Category:Fire]] |
||
[[Category: |
[[Category:Smoke| ]] |
||
[[Category:Pollution]] |
[[Category:Pollution]] |
||
[[Category:Air pollution]] |
[[Category:Air pollution]] |
||
smoke weed everyday haha yay<br> |
|||
po po po po po smoke weed everyday haha yay<br> |
|||
po po po po po, smoke weed everyday. |
|||





Smoke is a suspension[3] of airborne particulates and gases[4] emitted when a material undergoes combustionorpyrolysis, together with the quantity of air that is entrained or otherwise mixed into the mass. It is commonly an unwanted by-product of fires (including stoves, candles, internal combustion engines, oil lamps, and fireplaces), but may also be used for pest control (fumigation), communication (smoke signals), defensive and offensive capabilities in the military (smoke screen), cooking, or smoking (tobacco, cannabis, etc.). It is used in rituals where incense, sage, or resin is burned to produce a smell for spiritualormagical purposes. It can also be a flavoring agent and preservative.

Smoke inhalation is the primary cause of death in victims of indoor fires. The smoke kills by a combination of thermal damage, poisoning and pulmonary irritation caused by carbon monoxide, hydrogen cyanide and other combustion products.
Smoke is an aerosol (ormist) of solid particles and liquid droplets that are close to the ideal range of sizes for Mie scatteringofvisible light.[5]
The composition of smoke depends on the nature of the burning fuel and the conditions of combustion. Fires with high availability of oxygen burn at a high temperature and with a small amount of smoke produced; the particles are mostly composed of ash, or with large temperature differences, of condensed aerosol of water. High temperature also leads to production of nitrogen oxides.[6] Sulfur content yields sulfur dioxide, or in case of incomplete combustion, hydrogen sulfide.[7] Carbon and hydrogen are almost completely oxidized to carbon dioxide and water.[8] Fires burning with lack of oxygen produce a significantly wider palette of compounds, many of them toxic.[8] Partial oxidation of carbon produces carbon monoxide, while nitrogen-containing materials can yield hydrogen cyanide, ammonia, and nitrogen oxides.[9] Hydrogen gas can be produced instead of water.[9] Contents of halogens such as chlorine (e.g. in polyvinyl chlorideorbrominated flame retardants) may lead to the production of hydrogen chloride, phosgene, dioxin, and chloromethane, bromomethane and other halocarbons.[9][10] Hydrogen fluoride can be formed from fluorocarbons, whether fluoropolymers subjected to fire or halocarbon fire suppression agents. Phosphorus and antimony oxides and their reaction products can be formed from some fire retardant additives, increasing smoke toxicity and corrosivity.[10] Pyrolysisofpolychlorinated biphenyls (PCB), e.g. from burning older transformer oil, and to lower degree also of other chlorine-containing materials, can produce 2,3,7,8-tetrachlorodibenzodioxin, a potent carcinogen, and other polychlorinated dibenzodioxins.[10] Pyrolysis of fluoropolymers, e.g. teflon, in presence of oxygen yields carbonyl fluoride (which hydrolyzes readily to HF and CO2); other compounds may be formed as well, e.g. carbon tetrafluoride, hexafluoropropylene, and highly toxic perfluoroisobutene (PFIB).[11]

Pyrolysis of burning material, especially incomplete combustionorsmoldering without adequate oxygen supply, also results in production of a large amount of hydrocarbons, both aliphatic (methane, ethane, ethylene, acetylene) and aromatic (benzene and its derivates, polycyclic aromatic hydrocarbons; e.g. benzo[a]pyrene, studied as a carcinogen, or retene), terpenes.[12] It also results in the emission of a range of smaller oxygenated volatile organic compounds (methanol, acetic acid, hydroxy acetone, methyl acetate and ethyl formate) which are formed as combustion by products as well as less volatile oxygenated organic species such as phenolics, furans and furanones.[1] Heterocyclic compounds may be also present.[13] Heavier hydrocarbons may condense as tar; smoke with significant tar content is yellow to brown.[14] Combustion of solid fuels can result in the emission of many hundreds to thousands of lower volatility organic compounds in the aerosol phase.[15] Presence of such smoke, soot, and/or brown oily deposits during a fire indicates a possible hazardous situation, as the atmosphere may be saturated with combustible pyrolysis products with concentration above the upper flammability limit, and sudden inrush of air can cause flashoverorbackdraft.[16]
Presence of sulfur can lead to formation of gases like hydrogen sulfide, carbonyl sulfide, sulfur dioxide, carbon disulfide, and thiols; especially thiols tend to get adsorbed on surfaces and produce a lingering odor even long after the fire. Partial oxidation of the released hydrocarbons yields in a wide palette of other compounds: aldehydes (e.g. formaldehyde, acrolein, and furfural), ketones, alcohols (often aromatic, e.g. phenol, guaiacol, syringol, catechol, and cresols), carboxylic acids (formic acid, acetic acid, etc.).[citation needed]
The visible particulate matter in such smokes is most commonly composed of carbon (soot). Other particulates may be composed of drops of condensed tar, or solid particles of ash. The presence of metals in the fuel yields particles of metal oxides. Particles of inorganic salts may also be formed, e.g. ammonium sulfate, ammonium nitrate, or sodium chloride. Inorganic salts present on the surface of the soot particles may make them hydrophilic. Many organic compounds, typically the aromatic hydrocarbons, may be also adsorbed on the surface of the solid particles. Metal oxides can be present when metal-containing fuels are burned, e.g. solid rocket fuels containing aluminium. Depleted uranium projectiles after impacting the target ignite, producing particles of uranium oxides. Magnetic particles, spherules of magnetite-like ferrous ferric oxide, are present in coal smoke; their increase in deposits after 1860 marks the beginning of the Industrial Revolution.[17] (Magnetic iron oxide nanoparticles can be also produced in the smoke from meteorites burning in the atmosphere.)[18] Magnetic remanence, recorded in the iron oxide particles, indicates the strength of Earth's magnetic field when they were cooled beyond their Curie temperature; this can be used to distinguish magnetic particles of terrestrial and meteoric origin.[19] Fly ash is composed mainly of silica and calcium oxide. Cenospheres are present in smoke from liquid hydrocarbon fuels. Minute metal particles produced by abrasion can be present in engine smokes. Amorphous silica particles are present in smokes from burning silicones; small proportion of silicon nitride particles can be formed in fires with insufficient oxygen. The silica particles have about 10 nm size, clumped to 70–100 nm aggregates and further agglomerated to chains.[11] Radioactive particles may be present due to traces of uranium, thorium, or other radionuclides in the fuel; hot particles can be present in case of fires during nuclear accidents (e.g. Chernobyl disaster) or nuclear war.
Smoke particulates, like other aerosols, are categorized into three modes based on particle size:
Most of the smoke material is primarily in coarse particles. Those undergo rapid dry precipitation, and the smoke damage in more distant areas outside of the room where the fire occurs is therefore primarily mediated by the smaller particles.[20]
Aerosol of particles beyond visible size is an early indicator of materials in a preignition stage of a fire.[11]
Burning of hydrogen-rich fuel produces water vapor; this results in smoke containing droplets of water. In absence of other color sources (nitrogen oxides, particulates...), such smoke is white and cloud-like.
Smoke emissions may contain characteristic trace elements. Vanadium is present in emissions from oil fired power plants and refineries; oil plants also emit some nickel. Coal combustion produces emissions containing aluminium, arsenic, chromium, cobalt, copper, iron, mercury, selenium, and uranium.
Traces of vanadium in high-temperature combustion products form droplets of molten vanadates. These attack the passivation layers on metals and cause high temperature corrosion, which is a concern especially for internal combustion engines. Molten sulfate and lead particulates also have such effect.
Some components of smoke are characteristic of the combustion source. Guaiacol and its derivatives are products of pyrolysis of lignin and are characteristic of wood smoke; other markers are syringol and derivates, and other methoxy phenols. Retene, a product of pyrolysis of conifer trees, is an indicator of forest fires. Levoglucosan is a pyrolysis product of cellulose. Hardwoodvssoftwood smokes differ in the ratio of guaiacols/syringols. Markers for vehicle exhaust include polycyclic aromatic hydrocarbons, hopanes, steranes, and specific nitroarenes (e.g. 1-nitropyrene). The ratio of hopanes and steranes to elemental carbon can be used to distinguish between emissions of gasoline and diesel engines.[21]
Many compounds can be associated with particulates; whether by being adsorbed on their surfaces, or by being dissolved in liquid droplets. Hydrogen chloride is well absorbed in the soot particles.[20]
Inert particulate matter can be disturbed and entrained into the smoke. Of particular concern are particles of asbestos.
Deposited hot particlesofradioactive fallout and bioaccumulated radioisotopes can be reintroduced into the atmosphere by wildfires and forest fires; this is a concern in e.g. the Zone of alienation containing contaminants from the Chernobyl disaster.
Polymers are a significant source of smoke. Aromatic side groups, e.g. in polystyrene, enhance generation of smoke. Aromatic groups integrated in the polymer backbone produce less smoke, likely due to significant charring. Aliphatic polymers tend to generate the least smoke, and are non-self-extinguishing. However presence of additives can significantly increase smoke formation. Phosphorus-based and halogen-based flame retardants decrease production of smoke. Higher degree of cross-linking between the polymer chains has such effect too.[22]
This section does not cite any sources. Please help improve this sectionbyadding citations to reliable sources. Unsourced material may be challenged and removed. (January 2022) (Learn how and when to remove this message)
|


The naked eye detects particle sizes greater than 7 µm (micrometres). Visible particles emitted from a fire are referred to as smoke. Invisible particles are generally referred to as gas or fumes. This is best illustrated when toasting bread in a toaster. As the bread heats up, the products of combustion increase in size. The fumes initially produced are invisible but become visible if the toast is burnt.
Anionization chamber type smoke detector is technically a product of combustion detector, not a smoke detector. Ionization chamber type smoke detectors detect particles of combustion that are invisible to the naked eye. This explains why they may frequently false alarm from the fumes emitted from the red-hot heating elements of a toaster, before the presence of visible smoke, yet they may fail to activate in the early, low-heat smoldering stage of a fire.
Smoke from a typical house fire contains hundreds of different chemicals and fumes. As a result, the damage caused by the smoke can often exceed that caused by the actual heat of the fire. In addition to the physical damage caused by the smoke of a fire – which manifests itself in the form of stains – is the often even harder to eliminate problem of a smoky odor. Just as there are contractors that specialize in rebuilding/repairing homes that have been damaged by fire and smoke, fabric restoration companies specialize in restoring fabrics that have been damaged in a fire.
Smoke from oxygen-deprived fires contains a significant concentration of compounds that are flammable. A cloud of smoke, in contact with atmospheric oxygen, therefore has the potential of being ignited – either by another open flame in the area, or by its own temperature. This leads to effects like backdraft and flashover. Smoke inhalation is also a danger of smoke that can cause serious injury and death.[citation needed]

Many compounds of smoke from fires are highly toxic and/or irritating. The most dangerous is carbon monoxide leading to carbon monoxide poisoning, sometimes with the additive effects of hydrogen cyanide and phosgene. Smoke inhalation can therefore quickly lead to incapacitation and loss of consciousness. Sulfur oxides, hydrogen chloride and hydrogen fluoride in contact with moisture form sulfuric, hydrochloric and hydrofluoric acid, which are corrosive to both lungs and materials. When asleep the nose does not sense smoke nor does the brain, but the body will wake up if the lungs become enveloped in smoke and the brain will be stimulated and the person will be awoken. This does not work if the person is incapacitated or under the influence of drugs and/or alcohol.[citation needed]

Cigarette smoke is a major modifiable risk factor for lung disease, heart disease, and many cancers. Smoke can also be a component of ambient air pollution due to the burning of coal in power plants, forest fires or other sources, although the concentration of pollutants in ambient air is typically much less than that in cigarette smoke. One day of exposure to PM2.5 at a concentration of 880 μg/m3, such as occurs in Beijing, China, is the equivalent of smoking one or two cigarettes in terms of particulate inhalation by weight.[23][24] The analysis is complicated, however, by the fact that the organic compounds present in various ambient particulates may have a higher carcinogenicity than the compounds in cigarette smoke particulates.[25] Secondhand tobacco smoke is the combination of both sidestream and mainstream smoke emissions from a burning tobacco product. These emissions contain more than 50 carcinogenic chemicals. According to the United States Surgeon General's 2006 report on the subject, "Short exposures to secondhand [tobacco] smoke can cause blood platelets to become stickier, damage the lining of blood vessels, decrease coronary flow velocity reserves, and reduce heart variability, potentially increasing the risk of a heart attack".[26] The American Cancer Society lists "heart disease, lung infections, increased asthma attacks, middle ear infections, and low birth weight" as ramifications of smoker's emission.[27]


Smoke can obscure visibility, impeding occupant exiting from fire areas. In fact, the poor visibility due to the smoke that was in the Worcester Cold Storage Warehouse fireinWorcester, Massachusetts was the reason why the trapped rescue firefighters could not evacuate the building in time. Because of the striking similarity that each floor shared, the dense smoke caused the firefighters to become disoriented.[28]
Smoke can contain a wide variety of chemicals, many of them aggressive in nature. Examples are hydrochloric acid and hydrobromic acid, produced from halogen-containing plastics and fire retardants, hydrofluoric acid released by pyrolysisoffluorocarbon fire suppression agents, sulfuric acid from burning of sulfur-containing materials, nitric acid from high-temperature fires where nitrous oxide gets formed, phosphoric acid and antimony compounds from P and Sb based fire retardants, and many others. Such corrosion is not significant for structural materials, but delicate structures, especially microelectronics, are strongly affected. Corrosion of circuit board traces, penetration of aggressive chemicals through the casings of parts, and other effects can cause an immediate or gradual deterioration of parameters or even premature (and often delayed, as the corrosion can progress over long time) failure of equipment subjected to smoke. Many smoke components are also electrically conductive; deposition of a conductive layer on the circuits can cause crosstalks and other deteriorations of the operating parameters or even cause short circuits and total failures. Electrical contacts can be affected by corrosion of surfaces, and by deposition of soot and other conductive particles or nonconductive layers on or across the contacts. Deposited particles may adversely affect the performance of optoelectronics by absorbing or scattering the light beams.[citation needed]
Corrosivity of smoke produced by materials is characterized by the corrosion index (CI), defined as material loss rate (angstrom/minute) per amount of material gasified products (grams) per volume of air (m3). It is measured by exposing strips of metal to flow of combustion products in a test tunnel. Polymers containing halogen and hydrogen (polyvinyl chloride, polyolefins with halogenated additives, etc.) have the highest CI as the corrosive acids are formed directly with water produced by the combustion, polymers containing halogen only (e.g. polytetrafluoroethylene) have lower CI as the formation of acid is limited to reactions with airborne humidity, and halogen-free materials (polyolefins, wood) have the lowest CI.[20] However, some halogen-free materials can also release significant amount of corrosive products.[29]
Smoke damage to electronic equipment can be significantly more extensive than the fire itself. Cable fires are of special concern; low smoke zero halogen materials are preferable for cable insulation.[30]
When smoke comes into contact with the surface of any substance or structure, the chemicals contained in it are transferred to it. The corrosive properties of the chemicals cause the substance or structure to decompose at a rapid rate. Certain materials or structures absorb these chemicals, which is why clothing, unsealed surfaces, potable water, piping, wood, etc., are replaced in most cases of structural fires.[citation needed]
Wood smoke is a major source of air pollution,[31][32][33][34] especially particulate pollution,[32] pollution by polycyclic aromatic hydrocarbons (PAHs)[35] and volatile organic compounds (VOCs)[32][better source needed] such as formaldehyde.[36]
In the United Kingdom domestic combustion, especially for industrial uses, is the largest single source of PM2.5 annually.[37][38] In some towns and cities in New South Wales, wood smoke may be responsible for 60% of fine particle air pollution in the winter.[39] A year-long sampling campaign in Athens, Greece found a third (31%) of PAH urban air pollution to be caused by wood-burning, roughly as much as that of diesel and oil (33%) and gasoline (29%). It also found that wood-burning is responsible for nearly half (43%) of annual PAH lung cancer-risk compared to the other sources and that wintertime PAH levels were 7 times higher than in other seasons, presumably due to an increased use of fireplaces and heaters. The largest exposure events are periods during the winter with reduced atmospheric dispersion to dilute the accumulated pollution , in particular due to the low wind speeds.[35] Research conducted about biomass burning in 2015, estimated that 38% of European total particulate pollution emissions are composed of domestic wood burning.[40]
Wood smoke (for example from wildfires or wood ovens) can cause lung damage,[41][42] artery damage and DNA damage[43] leading to cancer,[44][45] other respiratory and lung disease and cardiovascular disease.[39][46] Air pollution, particulate matter and wood smoke may also cause brain damage because of particulates breaching the cardiovascular system and into the brain,[47][48][49][50] which can increase the risk of developmental disorders,[51][52][53][54] neurodegenerative disorders[55][56] mental disorders,[57][58][59] and suicidal behavior,[57][59] although studies on the link between depression and some air pollutants are not consistent.[60] At least one study has identified "the abundant presence in the human brain of magnetite nanoparticles that match precisely the high-temperature magnetite nanospheres, formed by combustion and/or friction-derived heating, which are prolific in urban, airborne particulate matter (PM)."[61] Air pollution has also been linked to a range of other psychosocial problems.[58]
As early as the 15th century Leonardo da Vinci commented at length on the difficulty of assessing smoke, and distinguished between black smoke (carbonized particles) and white 'smoke' which is not a smoke at all but merely a suspension of harmless water particulates.[62]
Smoke from heating appliances is commonly measured in one of the following ways:
In-line capture. A smoke sample is simply sucked through a filter which is weighed before and after the test and the mass of smoke found. This is the simplest and probably the most accurate method, but can only be used where the smoke concentration is slight, as the filter can quickly become blocked.[63]
The ASTM smoke pump is a simple and widely used method of in-line capture where a measured volume of smoke is pulled through a filter paper and the dark spot so formed is compared with a standard.
Filter/dilution tunnel. A smoke sample is drawn through a tube where it is diluted with air, the resulting smoke/air mixture is then pulled through a filter and weighed. This is the internationally recognized method of measuring smoke from combustion.[64]
Electrostatic precipitation. The smoke is passed through an array of metal tubes which contain suspended wires. A (huge) electrical potential is applied across the tubes and wires so that the smoke particles become charged and are attracted to the sides of the tubes. This method can over-read by capturing harmless condensates, or under-read due to the insulating effect of the smoke. However, it is the necessary method for assessing volumes of smoke too great to be forced through a filter, i.e., from bituminous coal.
Ringelmann scale. A measure of smoke color. Invented by Professor Maximilian Ringelmann in Paris in 1888, it is essentially a card with squares of black, white and shades of gray which is held up and the comparative grayness of the smoke judged. Highly dependent on light conditions and the skill of the observer it allocates a grayness number from 0 (white) to 5 (black) which has only a passing relationship to the actual quantity of smoke. Nonetheless, the simplicity of the Ringelmann scale means that it has been adopted as a standard in many countries.
Optical scattering. A light beam is passed through the smoke. A light detector is situated at an angle to the light source, typically at 90°, so that it receives only light reflected from passing particles. A measurement is made of the light received which will be higher as the concentration of smoke particles becomes higher.
Optical obscuration. A light beam is passed through the smoke and a detector opposite measures the light. The more smoke particles are present between the two, the less light will be measured.
Combined optical methods. There are various proprietary optical smoke measurement devices such as the 'nephelometer' or the 'aethalometer' which use several different optical methods, including more than one wavelength of light, inside a single instrument and apply an algorithm to give a good estimate of smoke. It has been claimed that these devices can differentiate types of smoke and so their probable source can be inferred, though this is disputed.[65]
Inference from carbon monoxide. Smoke is incompletely burned fuel, carbon monoxide is incompletely burned carbon, therefore it has long been assumed that measurement of CO in flue gas (a cheap, simple and very accurate procedure) will provide a good indication of the levels of smoke. Indeed, several jurisdictions use CO measurement as the basis of smoke control. However it is far from clear how accurate the correspondence is.
This section relies largely or entirely on a single source. Relevant discussion may be found on the talk page. Please help improve this articlebyintroducing citations to additional sources.
Find sources: "Smoke" – news · newspapers · books · scholar · JSTOR (October 2023) |
Throughout recorded history, humans have used the smoke of medicinal plants to cure illness. A sculpture from Persepolis shows Darius the Great (522–486 BC), the king of Persia, with two censers in front of him for burning Peganum harmala and/or sandalwood Santalum album, which was believed to protect the king from evil and disease. More than 300 plant species in 5 continents are used in smoke form for different diseases. As a method of drug administration, smoking is important as it is a simple, inexpensive, but very effective method of extracting particles containing active agents. More importantly, generating smoke reduces the particle size to a microscopic scale thereby increasing the absorption of its active chemical principles.[66]
{{cite book}}: CS1 maint: multiple names: authors list (link)
| National |
|
|---|---|
| Other |
|