J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 S o u r c e s
2 B i o s y n t h e s i s
3 P h a r m a c o l o g y
4 I n a b s i n t h e
T o g g l e I n a b s i n t h e s u b s e c t i o n
4 . 1 H i s t o r y
5 R e g u l a t i o n s
T o g g l e R e g u l a t i o n s s u b s e c t i o n
5 . 1 E u r o p e a n U n i o n
5 . 2 U n i t e d S t a t e s
5 . 3 C a n a d a
6 S e e a l s o
7 R e f e r e n c e s
8 F u r t h e r r e a d i n g
9 E x t e r n a l l i n k s
T o g g l e t h e t a b l e o f c o n t e n t s
T h u j o n e
2 9 l a n g u a g e s
● ا ل ع ر ب ي ة ● ت ۆ ر ک ج ه ● Б е л а р у с к а я ● Б е л а р у с к а я ( т а р а ш к е в і ц а ) ● C a t a l à ● Č e š t i n a ● D a n s k ● D e u t s c h ● E e s t i ● Ε λ λ η ν ι κ ά ● E s p a ñ o l ● E s p e r a n t o ● ف ا ر س ی ● F r a n ç a i s ● I t a l i a n o ● M a g y a r ● N e d e r l a n d s ● 日 本 語 ● N o r s k b o k m å l ● P o l s k i ● P o r t u g u ê s ● R o m â n ă ● Р у с с к и й ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● S u o m i ● S v e n s k a ● У к р а ї н с ь к а ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
( R e d i r e c t e d f r o m Α - t h u j o n e )
Group of four possible stereoisomers found in various plants: a.o., absinthe and mint
Thujone
(−)-α-Thujone
(+)-β-Thujone
Ball-and-stick model of (−)-α-thujone[1]
Names
IUPAC names
α: (1 S R R 1 S S R
Other names
Bicyclo[3.1.0]hexan-3-one, 4-methyl-1-(1-methylethyl)-, [1 S cis 1 S R R l cis -ThujoneZ 1 S R R
Identifiers
CAS Number
546-80-5 Y
471-15-8 Y
3D model (JSmol )
(β-thujone): Interactive image
Beilstein Reference
4660369
ChEBI
ChEMBL
ChemSpider
82583 Y
ECHA InfoCard 100.013.096
EC Number
IUPHAR/BPS
KEGG
PubChem CID
11027
UNII
R0SQ9G0DU5 Y
8ZI5R3T54Q Y
CompTox Dashboard (EPA )
InChI=1S/C10H16O/c1-6(2 )10-4-8(10 )7(3 )9(11 )5-10/h6-8H,4-5H2,1-3H3/t7-,8-,10+/m1/s1 Y
Key: USMNOWBWPHYOEA-MRTMQBJTSA-N Y
InChI=1/C10H16O/c1-6(2 )10-4-8(10 )7(3 )9(11 )5-10/h6-8H,4-5H2,1-3H3/t7-,8-,10+/m1/s1
Key: USMNOWBWPHYOEA-MRTMQBJTBZ
InChI=1S/C10H16O/c1-6(2 )10-4-8(10 )7(3 )9(11 )5-10/h6-8H,4-5H2,1-3H3/t7-,8-,10+/m1/s1
Key: USMNOWBWPHYOEA-MRTMQBJTSA-N
(α-thujone): O=C1[C@H](C )[C@@H]2[C@](C(C )C)(C1)C2
(β-thujone): C[C@@H]([C@@H](C2)[C@]2([C@@H](C )C)C1)C1=O
Properties
Chemical formula
C 10 H 16 O
Molar mass
−1
Density
0.92 g/cm3 3
Melting point
<25 °C
Boiling point
203 °C (397 °F; 476 K ) (alpha,beta-thujone)
Solubility in water
407 mg/L
Hazards
GHS labelling
Pictograms
Signal word
Warning
Hazard statements
H302
Precautionary statements
P264 , P270 , P301+P312 , P330 , P501
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
Thujone ([2] ketone and a monoterpene that occurs predominantly in two diastereomeric (epimeric ) forms: (−)-α-thujone and (+)-β-thujone .[3] [4]
Though it is best known as a chemical compound in the spirit absinthe , it is unlikely to be responsible for absinthe's alleged stimulant and psychoactive effects due to the small quantities present.[5] [6] [7]
Thujone acts on the GABAA as an antagonist. As a competitive antagonist of GABAA convulsant ,[8] [citation needed It is also found in perfumery as a component of several essential oils.[citation needed
In addition to the naturally occurring (−)-α-thujone and (+)-β-thujone, two other forms are possible: (+)-α-thujone and (−)-β-thujone . In 2016, they were found in nature as well,[9] in Salvia officinalis
(−)-α-thujone
(+)-α-thujone
(+)-β-thujone
(−)-β-thujone
Sources [ edit ]
Thujone is found in a number of plants, such as arborvitae (genus Thuja , hence the derivation of the name), Nootka cypress , some junipers , mugwort , oregano , common sage , tansy , and wormwood , most notably grand wormwood (Artemisia absinthium Mentha
Biosynthesis [ edit ]
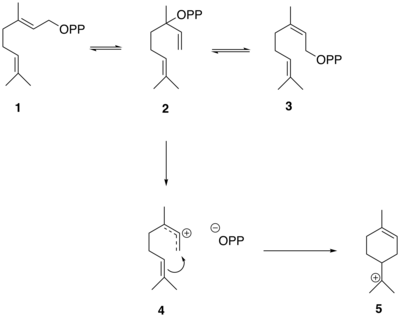
The biosynthesis of thujone is similar to the synthesis of other monoterpenes and begins with the formation of geranyl diphosphate (GPP) from dimethylallyl pyrophosphate (DMAPP) and isopentenyl diphosphate (IPP), catalyzed by the enzyme geranyl diphosphate synthase.[10] 13 isoprene units used to form thujone in plants are derived from the methylerythritol phosphate pathway (MEP).[11]
The reactions that generate the thujone skeleton in sabinene from GPP are mediated by the enzyme sabinene synthase which has GPP as its substrate.[10] 1 2 3 4 5 [10]
The conversion of GPP to alpha-terpinyl cation.
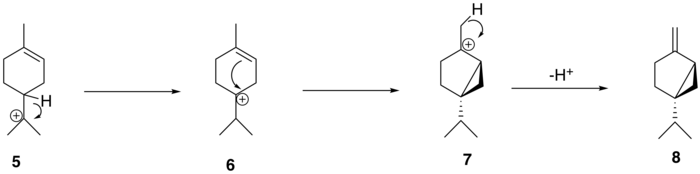
The α-terpinyl cation (5 Wagner–Meerwein rearrangement , leading to the formation of the terpinen-4-yl cation (6 7 (+)-sabinene (8
The conversion of alpha-terpinyl cation to (+)-sabinene
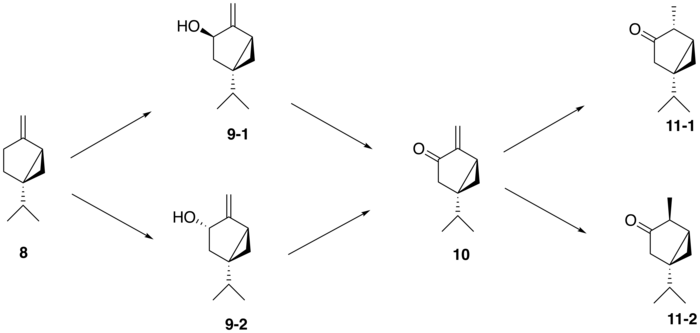
From (+)-sabinene (8 9-1, 9-2 ) by a cytochrome P450 enzyme , followed by conversion to (+)-sabinone (10 dehydrogenase . Finally, a reductase mediates the conversion to α-thujone (11-1 ) and β-thujone (11-2 ).[12] western redcedar (Thuja plicata trans -sabinol intermediate (9-1 ) whereas in the common garden sage (Salvia officinalis cis -sabinol intermediate (9-2 ).[13]
Proposed synthesis of thujone from sabinene
Pharmacology [ edit ]
Research-grade thujone
Based on studies that looked only at molecular shape, for many years thujone was thought to act similarly to THC on the cannabinoid receptors;[14] [15] GABAA receptor antagonist[16] GABAA . By inhibiting GABA receptor activation, neurons may fire more easily, which can cause muscle spasms and convulsions.[17] GABAA is specific to alpha-thujone.[18] 5-HT3 .[19] [20]
The median lethal dose , or LD50 convulsions that lead to death within 1 minute. From 30 to 45 mg/kg, the mice experience muscle spasms in the legs, which progress to general convulsions until death or recovery. These effects are in line with other GABA antagonists. Also, α-thujone is metabolized quickly in the liver in mice.[17] diazepam , phenobarbital , or 1 g/kg of ethanol protects against a lethal dose of 100 mg/kg.[citation needed
Attention performance has been tested with low and high doses of thujone in alcohol. The high dose had a short-term negative effect on attention performance. The lower dose showed no noticeable effect.[5]
Thujone is reported[by whom? to be toxic to brain, kidney, and liver cells and could cause convulsions if used in too high a dose. Other thujone-containing plants such as the tree arborvitae (Thuja occidentalis ) are used in herbal medicine, mainly for their alleged immune-system stimulating effects[citation needed . Side effects from the essential oil of this plant include anxiety, sleeplessness, and convulsions, which confirms the central nervous system effects of thujone.[8] [21]
In absinthe [ edit ]
Thujone is most commonly known for being a compound in the spirit absinthe . In the past, absinthe was thought to contain up to 260–350 mg/L thujone,[22] gas chromatography–mass spectrometry (GC-MS) found that the bottles had between 0.5 and 48.3 mg/L and averaged 25.4 mg/L [6] [23] [24] gas chromatography alone may record an inaccurately high reading of thujone as other compounds may interfere with and add to the apparent measured amount.[25]
History [ edit ]
The compound was discovered after absinthe became popular in the mid-19th century. Valentin Magnan , who studied alcoholism, tested pure wormwood oil on animals and discovered it caused seizures independent from the effects of alcohol. Based on this, absinthe, which contains a small amount of wormwood oil, was assumed to be more dangerous than ordinary alcohol. Eventually, thujone was isolated as the cause of these reactions. Magnan went on to study 250 abusers of alcohol and noted that those who drank absinthe had seizures and hallucinations . The seizures are caused by the (+)-α-thujone interacting with the GABA receptors, causing epileptic activity.[18] [26] [27]
After absinthe was banned, research dropped off until the 1970s, when the British scientific journal Nature tetrahydrocannabinol (THC), the primary psychoactive substance found in cannabis , and hypothesized it would act the same way on the brain, sparking the myth that thujone was a cannabinoid .[14] [28]
More recently, following European Council Directive No. 88/388/EEC (1988) allowing certain levels of thujone in foodstuffs in the EU,[29]
Regulations [ edit ]
European Union [ edit ]
Maximum thujone levels in the EU are:[30] [31]
0.5 mg/kg in food prepared with Artemisia sage and non alcoholic beverages
10 mg/kg in alcoholic beverages not prepared with Artemisia species
25 mg/kg in food prepared with sage
35 mg/kg in alcoholic beverages prepared with Artemisia species
United States [ edit ]
In the United States, the addition of pure thujone to foods is not permitted.[32] Artemisia white cedar , oakmoss , tansy , or yarrow , must be thujone-free,[33] [34] sage and sage oil (which can be up to 50% thujone) are on the Food and Drug Administration 's list of generally recognized as safe (GRAS) substances.[35]
Absinthe offered for sale in the United States must be thujone-free by the same standard that applies to other beverages containing Artemisia,[34]
In Canada, liquor laws are the domain of the provincial governments. Alberta, Ontario, and Nova Scotia allow 10 mg/kg thujone; Quebec allows 15 mg per kg;[citation needed Manitoba allows 6–8 mg thujone per litre; British Columbia adheres to the same levels as Ontario. However, in Saskatchewan and Quebec, one can purchase any liquor available in the world upon the purchase of a maximum of one case, usually 12 750-ml bottles or 9 L. The individual liquor boards must approve each product before it may be sold on shelves.
See also [ edit ]
Piołunówka
References [ edit ]
^ Derived from the Ancient Greek θυία, thuj(a ) , a kind of cedar + -ωνη, -one , feminine patronymic for a chemical relative of acetone
^ Perry NB, Anderson RE, Brennan NJ, Douglas MH, Heaney AJ, McGimpsey JA, Smallfield BM (1999). "Essential Oils from Dalmatian Sage (Salvia officinalis L.): Variations among Individuals, Plant Parts, Seasons, and Sites". J. Agric. Food Chem. 47 5 ): 2048–2054. doi :10.1021/jf981170m . PMID 10552494 .
^ Oppolzer W, Pimm A, Stammen B, Hume WE (1997). "Palladium-Catalysed Intramolecular Cyclisations of Olefinic Propargylic Carbonates and application to the diastereoselective synthesis of enantiomerically pure (−)-α-thujone" . Helv. Chim. Acta 80 3 ): 623–639. doi :10.1002/hlca.19970800302
^ a b Dettling A, Grass H, Schuff A, Skopp G, Strohbeck-Kuehner P, Haffner HT (2004). "Absinthe: attention performance and mood under the influence of thujone" . J. Stud. Alcohol . 65 5 ): 573–81. doi :10.15288/jsa.2004.65.573 . PMID 15536765 .
^ a b Absinthe Myths Finally Laid To Rest
^ Chemical Composition of Vintage Preban Absinthe with Special Reference to Thujone, Fenchone, Pinocamphone, Methanol, Copper, and Antimony Concentrations
^ a b Olsen, Richard W. (2000-04-25). "Absinthe and γ-aminobutyric acid receptors" . Proceedings of the National Academy of Sciences of the United States of America . 97 9 ): 4417–4418. Bibcode :2000PNAS...97.4417O . doi :10.1073/pnas.97.9.4417 ISSN 0027-8424 . PMC 34311 PMID 10781032 .
^ Williams, Jack D.; Yazarians, Jessica A.; Almeyda, Chelcie C.; Anderson, Kristin A.; Boyce, Gregory R. (23 May 2016). "Detection of the Previously Unobserved Stereoisomers of Thujone in the Essential Oil and Consumable Products of Sage (Salvia officinalis L.) Using Headspace Solid-Phase Microextraction–Gas Chromatography–Mass Spectrometry". Journal of Agricultural and Food Chemistry . 64 21 ): 4319–4326. doi :10.1021/acs.jafc.6b01065 . PMID 27181395 .
^ a b c Dewick, Paul M (2009). Medicinal natural products: a biosynthetic approach 195 –197. ISBN 978-0-470-74167-2
^ Umlauf, Dirk; Zapp, Josef (September 2004). "Biosynthesis of the irregular monoterpene artemisia ketone, the sesquiterpene germacrene D and other isoprenoids in Tanacetum vulgare L. (Asteraceae)". Phytochemistry . 65 17 ): 2463–2470. Bibcode :2004PChem..65.2463U . doi :10.1016/j.phytochem.2004.08.019 . PMID 15381410 .
^ Foster, Adam J.; Hall, Dawn E. (Apr 2013). "Identification of Genes in Thuja plicata Foliar Terpenoid Defenses" . Plant Physiology . 161 (4 ): 1993–2004. doi :10.1104/pp.112.206383 . PMC 3613470 PMID 23388118 .
^ Gesell, Andreas; Blaukopf, Markus (May 2015). "The Gymnosperm Cytochrome P450 CYP750B1 Catalyzes Stereospecific Monoterpene Hydroxylation of (+)-Sabinene in Thujone Biosynthesis in Western Redcedar" . Plant Physiology . 168 (1 ): 94–106. doi :10.1104/pp.15.00315 . PMC 4424034 PMID 25829465 .
^ a b Conrad, Barnaby, III ; (1988). Absinthe: History in a Bottle. Chronicle Books . ISBN 0-8118-1650-8 p. 152
^ Meschler JP, Howlett AC (March 1999). "Thujone exhibits low affinity for cannabinoid receptors but fails to evoke cannabimimetic responses". Pharmacol. Biochem. Behav . 62 3 ): 473–480. doi :10.1016/S0091-3057(98 )00195-6 . PMID 10080239 . S2CID 30865036 .
^ Olsen RW (April 2000). "Absinthe and gamma-aminobutyric acid receptors" . Proc. Natl. Acad. Sci. U.S.A . 97 9 ): 4417–4418. Bibcode :2000PNAS...97.4417O . doi :10.1073/pnas.97.9.4417 PMC 34311 PMID 10781032 .
^ a b Höld KM, Sirisoma NS, Ikeda T, Narahashi T, Casida JE (April 2000). "Alpha-thujone (the active component of absinthe): gamma-aminobutyric acid type A receptor modulation and metabolic detoxification" . Proc. Natl. Acad. Sci. U.S.A . 97 8 ): 3826–31. Bibcode :2000PNAS...97.3826H . doi :10.1073/pnas.070042397 PMC 18101 PMID 10725394 .
^ a b Höld, Karin M.; Sirisoma, Nilantha S.; Ikeda, Tomoko; Narahashi, Toshio; Casida, John E. (2000-04-11). "α-Thujone (the active component of absinthe): γ-Aminobutyric acid type A receptor modulation and metabolic detoxification" . Proceedings of the National Academy of Sciences of the United States of America . 97 8 ): 3826–3831. Bibcode :2000PNAS...97.3826H . doi :10.1073/pnas.070042397 ISSN 0027-8424 . PMC 18101 PMID 10725394 .
^ Deiml T, Haseneder R, Zieglgänsberger W, Rammes G, Eisensamer B, Rupprecht R, Hapfelmeier G (Feb 2004). "Alpha-thujone reduces 5-HT3 receptor activity by an effect on the agonist-reduced desensitization". Neuropharmacology . 46 2 ): 192–201. doi :10.1016/j.neuropharm.2003.09.022 . PMID 15002407 . S2CID 54346490 .
^ Modulation of Ionotropic GABA Receptors by Natural Products of Plant Origin
^ Naser B, Bodinet C, Tegtmeier M, Lindequist U (Mar 2005). "Thuja occidentalis (Arbor vitae): A Review of its Pharmaceutical, Pharmacological and Clinical Properties" . Evidence-Based Complementary and Alternative Medicine . 2 1 ): 69–78. doi :10.1093/ecam/neh065 . PMC 1062158 PMID 15841280 .
^ Absinthism: a fictitious 19th-century syndrome with present impact , Padosch et al. Retrieved Oct. 28, 2006.
^ Chemical Composition of Vintage Preban Absinthe with Special Reference to Thujone, Fenchone, Pinocamphone, Methanol, Copper, and Antimony Concentrations
^ Thujone—Cause of absinthism? Lachenmeier, Emmert et al. Retrieved Oct. 28, 2006.
^ Determination of α-/β-Thujone and Related Terpenes in Absinthe using Solid Phase Extraction and Gas Chromatography Archived 2007-11-27 at the Wayback Machine , Emmert et al. Retrieved Oct. 28, 2006.
^ Lachenmeier, Dirk; Nathan-Maister, David; Breaux, Theodore; Luaute, Jean-Pierre; Emmert, Joachim (2010). "Absinthe, Absinthism and Thujone – New Insight into the Spirit's Impact on Public Health" . The Open Addiction Journal . 3 doi :10.2174/1874941001003010032
^ Conrad III, Barnaby ; (1988). Absinthe: History in a Bottle . Chronicle books . ISBN 0-8118-1650-8 Pg. 101-105
^ Del Castillo J.; Anderson M.; Rubottom G.M. (1975). "Letters to Nature: Marijuana, absinthe and the central nervous system". Nature . 253 (5490): 365–366. doi :10.1038/253365a0 . PMID 1110781 . S2CID 4245058 .
^ European Council Directive No. 88/388/EEC, 22 June 1988.
^ Regulation (EC ) No 1334/2008 of the European Parliament and Council of 16 December 2008 , European Commission .
^ Opinion of the Scientific Committee on Food on Thujone Scientific Committee on Food (2003) Retrieved Oct 28, 2006.
^ Laurie C. Dolan; Ray A. Matulka & George A. Burdock (2010). "Naturally Occurring Food Toxins" . Toxins . 2 9 ): 2289–2332. doi :10.3390/toxins2092289 PMC 3153292 PMID 22069686 .
^ FDA Regulation 21 CFR 172.510 – Food Additives Permitted for Direct Addition to Food for Human Consumption. Food and Drug Administration (2003). Retrieved Oct 28, 2006.
^ a b Department of the Treasury Alcohol and Tobacco Tax and Trade Bureau Industry Circular 2007-5 Archived 2014-02-09 at the Wayback Machine October 17, 2007. Retrieved May 5, 2009
^ Substances generally recognized as safe. Archived 2005-11-30 at the Wayback Machine Food and Drug Administration (2003). Retrieved Oct 28, 2006.
Further reading [ edit ]
Lachenmeier DW, Nathan-Maister D, Breaux TA, Sohnius EM, Schoeberl K, Kuballa T (May 2008). "Chemical composition of vintage preban absinthe with special reference to thujone, fenchone, pinocamphone, methanol, copper, and antimony concentrations" . J. Agric. Food Chem . 56 9 ): 3073–81. doi :10.1021/jf703568f PMID 18419128 .
External links [ edit ]
Wikimedia Commons has media related to
Thujone .
t
e
Ionotropic
GABAA Tooltip γ-Aminobutyric acid A receptor
Positive modulators (abridged; see here for a full list): α-EMTBL
Alcohols (e.g., drinking alcohol , 2M2B )
Anabolic steroids
Avermectins (e.g., ivermectin )
Barbiturates (e.g., phenobarbital )
Benzodiazepines (e.g., diazepam )
Bromide compounds (e.g., potassium bromide )
Carbamates (e.g., meprobamate )
Carbamazepine
Chloralose
Chlormezanone
Clomethiazole
Dihydroergolines (e.g., ergoloid (dihydroergotoxine) )
Etazepine
Etifoxine
Fenamates (e.g., mefenamic acid )
Flavonoids (e.g., apigenin , hispidulin )
Fluoxetine
Flupirtine
Imidazoles (e.g., etomidate )
Kava constituents (e.g., kavain )
Lanthanum
Loreclezole
Monastrol
Neuroactive steroids (e.g., allopregnanolone , cholesterol , THDOC )
Niacin
Niacinamide
Nonbenzodiazepines (e.g., β-carbolines (e.g., abecarnil ), cyclopyrrolones (e.g., zopiclone ), imidazopyridines (e.g., zolpidem ), pyrazolopyrimidines (e.g., zaleplon ))
Norfluoxetine
Petrichloral
Phenols (e.g., propofol )
Phenytoin
Piperidinediones (e.g., glutethimide )
Propanidid
Pyrazolopyridines (e.g., etazolate )
Quinazolinones (e.g., methaqualone )
Retigabine (ezogabine)
ROD-188
Skullcap constituents (e.g., baicalin )
Stiripentol
Sulfonylalkanes (e.g., sulfonmethane (sulfonal) )
Topiramate
Valerian constituents (e.g., valerenic acid )
Volatiles /gases (e.g., chloral hydrate , chloroform , diethyl ether , paraldehyde , sevoflurane )
Negative modulators: 1,3M1B
3M2B
11-Ketoprogesterone
17-Phenylandrostenol
α3IA
α5IA (LS-193,268)
β-CCB
β-CCE
β-CCM
β-CCP
β-EMGBL
Anabolic steroids
Amiloride
Anisatin
β-Lactams (e.g., penicillins , cephalosporins , carbapenems )
Basmisanil
Bemegride
Bicyclic phosphates (TBPS , TBPO , IPTBO )
BIDN
Bilobalide
Bupropion
CHEB
Chlorophenylsilatrane
Cicutoxin
Cloflubicyne
Cyclothiazide
DHEA
DHEA-S
Dieldrin
(+)-DMBB
DMCM
DMPC
EBOB
Etbicyphat
FG-7142 (ZK-31906)
Fiproles (e.g., fipronil )
Flavonoids (e.g., amentoflavone , oroxylin A )
Flumazenil
Fluoroquinolones (e.g., ciprofloxacin )
Flurothyl
Furosemide
Golexanolone
Iomazenil (123 I )
IPTBO
Isopregnanolone (sepranolone)
L-655,708
Laudanosine
Lindane
MaxiPost
Morphine
Morphine-3-glucuronide
MRK-016
Naloxone
Naltrexone
Nicardipine
Nonsteroidal antiandrogens (e.g., apalutamide , bicalutamide , enzalutamide , flutamide , nilutamide )
Oenanthotoxin
Pentylenetetrazol (pentetrazol)
Phenylsilatrane
Picrotoxin (i.e., picrotin , picrotoxinin and dihydropicrotoxinin )
Pregnenolone sulfate
Propybicyphat
PWZ-029
Radequinil
Ro 15-4513
Ro 19-4603
RO4882224
RO4938581
Sarmazenil
SCS
Suritozole
TB-21007
TBOB
TBPS
TCS-1105
Terbequinil
TETS
Thujone
U-93631
Zinc
ZK-93426
GABAA Tooltip γ-Aminobutyric acid A-rho receptor
Metabotropic
GABAB Tooltip γ-Aminobutyric acid B receptor
See also
Receptor/signaling modulators
GABAA
GABA metabolism/transport modulators
t
e
5-HT1
5-HT1A
Agonists: 8-OH-DPAT
Adatanserin
Amphetamine
Antidepressants (e.g., etoperidone , hydroxynefazodone , nefazodone , trazodone , triazoledione , vilazodone , vortioxetine )
Atypical antipsychotics (e.g., aripiprazole , asenapine , brexpiprazole , cariprazine , clozapine , lurasidone , quetiapine , ziprasidone )
Azapirones (e.g., buspirone , eptapirone , gepirone , perospirone , tandospirone )
Bay R 1531
Befiradol
BMY-14802
Cannabidiol
Dimemebfe
Dopamine
Ebalzotan
Eltoprazine
Enciprazine
Ergolines (e.g., bromocriptine , cabergoline , dihydroergotamine , ergotamine , lisuride , LSD , methylergometrine (methylergonovine) , methysergide , pergolide )
F-11,461
F-12826
F-13714
F-14679
F-15063
F-15,599
Flesinoxan
Flibanserin
Flumexadol
Hypidone
Lesopitron
LY-293284
LY-301317
mCPP
MKC-242
Naluzotan
NBUMP
Osemozotan
Oxaflozane
Pardoprunox
Piclozotan
Rauwolscine
Repinotan
Roxindole
RU-24,969
S-14,506
S-14671
S-15535
Sarizotan
Serotonin (5-HT)
SSR-181507
Sunepitron
Tryptamines (e.g., 5-CT , 5-MeO-DMT , 5-MT , bufotenin , DMT , indorenate , N-Me-5-HT , psilocin , psilocybin )
TGBA01AD
U-92,016-A
Urapidil
Vilazodone
Xaliproden
Yohimbine
Antagonists: Atypical antipsychotics (e.g., iloperidone , risperidone , sertindole )
AV965
Beta blockers (e.g., alprenolol , carteolol , cyanopindolol , iodocyanopindolol , isamoltane , oxprenolol , penbutolol , pindobind , pindolol , propranolol , tertatolol )
BMY-7,378
CSP-2503
Dotarizine
Ergolines (e.g., metergoline )
FCE-24379
Flopropione
GR-46611
Isamoltane
Lecozotan
Mefway
Metitepine (methiothepin)
MIN-117 (WF-516)
MPPF
NAN-190
Robalzotan
S-15535
SB-649,915
SDZ 216-525
Spiperone
Spiramide
Spiroxatrine
UH-301
WAY-100135
WAY-100635
Xylamidine
5-HT1B
Agonists: Anpirtoline
CGS-12066A
CP-93129
CP-94253
CP-122,288
CP-135807
Eltoprazine
Ergolines (e.g., bromocriptine , dihydroergotamine , ergotamine , methylergometrine (methylergonovine) , methysergide , pergolide )
mCPP
RU-24,969
Serotonin (5-HT)
Triptans (e.g., avitriptan , donitriptan , eletriptan , sumatriptan , zolmitriptan )
TFMPP
Tryptamines (e.g., 5-BT , 5-CT , 5-MT , DMT )
Vortioxetine
5-HT1D
Agonists: CP-122,288
CP-135807
CP-286601
Ergolines (e.g., bromocriptine , cabergoline , dihydroergotamine , ergotamine , LSD , methysergide )
GR-46611
L-694247
L-772405
mCPP
PNU-109291
PNU-142633
Serotonin (5-HT)
TGBA01AD
Triptans (e.g., almotriptan , avitriptan , donitriptan , eletriptan , frovatriptan , naratriptan , rizatriptan , sumatriptan , zolmitriptan )
Tryptamines (e.g., 5-BT , 5-CT , 5-Et-DMT , 5-MT , 5-(nonyloxy)tryptamine , DMT )
5-HT1E
5-HT1F
5-HT2
5-HT2A
Agonists: 25H/NB series (e.g., 25I-NBF , 25I-NBMD , 25I-NBOH , 25I-NBOMe , 25B-NBOMe , 25C-NBOMe , 25TFM-NBOMe , 2CBCB-NBOMe , 25CN-NBOH , 2CBFly-NBOMe )
2Cs (e.g., 2C-B , 2C-E , 2C-I , 2C-T-2 , 2C-T-7 , 2C-T-21 )
2C-B-FLY
2CB-Ind
5-Methoxytryptamines (5-MeO-DET , 5-MeO-DiPT , 5-MeO-DMT , 5-MeO-DPT , 5-MT )
α-Alkyltryptamines (e.g., 5-Cl-αMT , 5-Fl-αMT , 5-MeO-αET , 5-MeO-αMT , α-Me-5-HT , αET , αMT )
AL-34662
AL-37350A
Bromo-DragonFLY
Dimemebfe
DMBMPP
DOx (e.g., DOB , DOC , DOI , DOM )
Efavirenz
Ergolines (e.g., 1P-LSD , ALD-52 , bromocriptine , cabergoline , ergine (LSA) , ergometrine (ergonovine) , ergotamine , lisuride , LA-SS-Az , LSB , LSD , LSD-Pip , LSH , LSP , methylergometrine (methylergonovine) , pergolide )
Flumexadol
IHCH-7113
Jimscaline
Lorcaserin
MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )
O-4310
Oxaflozane
PHA-57378
PNU-22394
PNU-181731
RH-34
SCHEMBL5334361
Phenethylamines (e.g., lophophine , mescaline )
Piperazines (e.g., BZP , quipazine , TFMPP )
Serotonin (5-HT)
TCB-2
TFMFly
Tryptamines (e.g., 5-BT , 5-CT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )
Antagonists: 5-I-R91150
5-MeO-NBpBrT
AC-90179
Adatanserin
Altanserin
Antihistamines (e.g., cyproheptadine , hydroxyzine , ketotifen , perlapine )
AMDA
Atypical antipsychotics (e.g., amperozide , aripiprazole , asenapine , blonanserin , brexpiprazole , carpipramine , clocapramine , clorotepine , clozapine , fluperlapine , gevotroline , iloperidone , lurasidone , melperone , mosapramine , ocaperidone , olanzapine , paliperidone , quetiapine , risperidone , sertindole , zicronapine , ziprasidone , zotepine )
Chlorprothixene
Cinanserin
CSP-2503
Deramciclane
Dotarizine
Eplivanserin
Ergolines (e.g., amesergide , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )
Fananserin
Flibanserin
Glemanserin
Irindalone
Ketanserin
KML-010
Landipirdine
LY-393558
mCPP
Medifoxamine
Metitepine (methiothepin)
MIN-117 (WF-516)
Naftidrofuryl
Nantenine
Nelotanserin
Opiranserin (VVZ-149)
Pelanserin
Phenoxybenzamine
Pimavanserin
Pirenperone
Pizotifen
Pruvanserin
Rauwolscine
Ritanserin
Roluperidone
S-14671
Sarpogrelate
Serotonin antagonists and reuptake inhibitors (e.g., etoperidone , hydroxynefazodone , lubazodone , mepiprazole , nefazodone , triazoledione , trazodone )
SR-46349B
TGBA01AD
Teniloxazine
Temanogrel
Tetracyclic antidepressants (e.g., amoxapine , aptazapine , esmirtazapine , maprotiline , mianserin , mirtazapine )
Tricyclic antidepressants (e.g., amitriptyline )
Typical antipsychotics (e.g., chlorpromazine , fluphenazine , haloperidol , loxapine , perphenazine , pimozide , pipamperone , prochlorperazine , setoperone , spiperone , spiramide , thioridazine , thiothixene , trifluoperazine )
Volinanserin
Xylamidine
Yohimbine
5-HT2B
Agonists: 4-Methylaminorex
Aminorex
Amphetamines (e.g., chlorphentermine , cloforex , dexfenfluramine , fenfluramine , levofenfluramine , norfenfluramine )
BW-723C86
DOx (e.g., DOB , DOC , DOI , DOM )
Ergolines (e.g., cabergoline , dihydroergocryptine , dihydroergotamine , ergotamine , methylergometrine (methylergonovine) , methysergide , pergolide )
Lorcaserin
MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )
Piperazines (e.g., TFMPP )
PNU-22394
Ro60-0175
Serotonin (5-HT)
Tryptamines (e.g., 5-BT , 5-CT , 5-MT , α-Me-5-HT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )
Antagonists: Agomelatine
Atypical antipsychotics (e.g., amisulpride , aripiprazole , asenapine , brexpiprazole , cariprazine , clozapine , N-desalkylquetiapine (norquetiapine) , N-desmethylclozapine (norclozapine) , olanzapine , pipamperone , quetiapine , risperidone , ziprasidone )
Cyproheptadine
EGIS-7625
Ergolines (e.g., amesergide , bromocriptine , lisuride , LY-53857 , LY-272015 , mesulergine )
Ketanserin
LY-393558
mCPP
Metadoxine
Metitepine (methiothepin)
Pirenperone
Pizotifen
Propranolol
PRX-08066
Rauwolscine
Ritanserin
RS-127445
Sarpogrelate
SB-200646
SB-204741
SB-206553
SB-215505
SB-221284
SB-228357
SDZ SER-082
Tegaserod
Tetracyclic antidepressants (e.g., amoxapine , mianserin , mirtazapine )
Trazodone
Typical antipsychotics (e.g., chlorpromazine )
TIK-301
Yohimbine
5-HT2C
Agonists: 2Cs (e.g., 2C-B , 2C-E , 2C-I , 2C-T-2 , 2C-T-7 , 2C-T-21 )
5-Methoxytryptamines (5-MeO-DET , 5-MeO-DiPT , 5-MeO-DMT , 5-MeO-DPT , 5-MT )
α-Alkyltryptamines (e.g., 5-Cl-αMT , 5-Fl-αMT , 5-MeO-αET , 5-MeO-αMT , α-Me-5-HT , αET , αMT )
A-372159
AL-38022A
Alstonine
CP-809101
Dimemebfe
DOx (e.g., DOB , DOC , DOI , DOM )
Ergolines (e.g., ALD-52 , cabergoline , dihydroergotamine , ergine (LSA) , ergotamine , lisuride , LA-SS-Az , LSB , LSD , LSD-Pip , LSH , LSP , pergolide )
Flumexadol
Lorcaserin
MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )
MK-212
ORG-12962
ORG-37684
Oxaflozane
PHA-57378
Phenethylamines (e.g., lophophine , mescaline )
Piperazines (e.g., aripiprazole , BZP , mCPP , quipazine , TFMPP )
PNU-22394
PNU-181731
Ro60-0175
Ro60-0213
Serotonin (5-HT)
Tryptamines (e.g., 5-BT , 5-CT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )
Vabicaserin
WAY-629
WAY-161503
YM-348
Antagonists: Adatanserin
Agomelatine
Atypical antipsychotics (e.g., asenapine , clorotepine , clozapine , fluperlapine , iloperidone , melperone , olanzapine , paliperidone , quetiapine , risperidone , sertindole , ziprasidone , zotepine )
Captodiame
CEPC
Cinanserin
Cyproheptadine
Deramciclane
Desmetramadol
Dotarizine
Eltoprazine
Ergolines (e.g., amesergide , bromocriptine , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )
Etoperidone
Fluoxetine
FR-260010
Irindalone
Ketanserin
Ketotifen
Latrepirdine (dimebolin)
Medifoxamine
Metitepine (methiothepin)
Nefazodone
Pirenperone
Pizotifen
Propranolol
Ritanserin
RS-102221
S-14671
SB-200646
SB-206553
SB-221284
SB-228357
SB-242084
SB-243213
SDZ SER-082
Tedatioxetine
Tetracyclic antidepressants (e.g., amoxapine , aptazapine , esmirtazapine , maprotiline , mianserin , mirtazapine )
TIK-301
Tramadol
Trazodone
Tricyclic antidepressants (e.g., amitriptyline , nortriptyline )
Typical antipsychotics (e.g., chlorpromazine , loxapine , pimozide , pipamperone , thioridazine )
Xylamidine
5-HT3 –7
5-HT3
Agonists: Alcohols (e.g., butanol , ethanol (alcohol) , trichloroethanol )
m-CPBG
Phenylbiguanide
Piperazines (e.g., BZP , mCPP , quipazine )
RS-56812
Serotonin (5-HT)
SR-57227
SR-57227A
Tryptamines (e.g., 2-Me-5-HT , 5-CT , bufotenidine (5-HTQ) )
Volatiles/gases (e.g., halothane , isoflurane , toluene , trichloroethane )
YM-31636
Antagonists: Alosetron
Anpirtoline
Arazasetron
AS-8112
Atypical antipsychotics (e.g., clozapine , olanzapine , quetiapine )
Azasetron
Batanopride
Bemesetron (MDL-72222)
Bupropion
Cilansetron
CSP-2503
Dazopride
Dolasetron
Galanolactone
Granisetron
Hydroxybupropion
Lerisetron
Memantine
Ondansetron
Palonosetron
Ramosetron
Renzapride
Ricasetron
Tedatioxetine
Tetracyclic antidepressants (e.g., amoxapine , mianserin , mirtazapine )
Thujone
Tropanserin
Tropisetron
Typical antipsychotics (e.g., loxapine )
Volatiles/gases (e.g., nitrous oxide , sevoflurane , xenon )
Vortioxetine
Zacopride
Zatosetron
5-HT4
5-HT5A
5-HT6
Agonists: Ergolines (e.g., dihydroergocryptine , dihydroergotamine , ergotamine , lisuride , LSD , mesulergine , metergoline , methysergide )
Hypidone
Serotonin (5-HT)
Tryptamines (e.g., 2-Me-5-HT , 5-BT , 5-CT , 5-MT , Bufotenin , E-6801 , E-6837 , EMD-386088 , EMDT , LY-586713 , N-Me-5-HT , ST-1936 , tryptamine )
WAY-181187
WAY-208466
Antagonists: ABT-354
Atypical antipsychotics (e.g., aripiprazole , asenapine , clorotepine , clozapine , fluperlapine , iloperidone , olanzapine , tiospirone )
AVN-101
AVN-211
AVN-322
AVN-397
BGC20-760
BVT-5182
BVT-74316
Cerlapirdine
EGIS-12,233
GW-742457
Idalopirdine
Ketanserin
Landipirdine
Latrepirdine (dimebolin)
Masupirdine
Metitepine (methiothepin)
MS-245
PRX-07034
Ritanserin
Ro 04-6790
Ro 63-0563
SB-258585
SB-271046
SB-357134
SB-399885
SB-742457
Tetracyclic antidepressants (e.g., amoxapine , mianserin )
Tricyclic antidepressants (e.g., amitriptyline , clomipramine , doxepin , nortriptyline )
Typical antipsychotics (e.g., chlorpromazine , loxapine )
5-HT7
Antagonists: Atypical antipsychotics (e.g., amisulpride , aripiprazole , asenapine , brexpiprazole , clorotepine , clozapine , fluperlapine , olanzapine , risperidone , sertindole , tiospirone , ziprasidone , zotepine )
Butaclamol
DR-4485
EGIS-12,233
Ergolines (e.g., 2-Br-LSD (BOL-148) , amesergide , bromocriptine , cabergoline , dihydroergotamine , ergotamine , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )
JNJ-18038683
Ketanserin
LY-215,840
Metitepine (methiothepin)
Ritanserin
SB-258719
SB-258741
SB-269970
SB-656104
SB-656104A
SB-691673
SLV-313
SLV-314
Spiperone
SSR-181507
Tetracyclic antidepressants (e.g., amoxapine , maprotiline , mianserin , mirtazapine )
Tricyclic antidepressants (e.g., amitriptyline , clomipramine , imipramine )
Typical antipsychotics (e.g., acetophenazine , chlorpromazine , chlorprothixene , fluphenazine , loxapine , pimozide )
Vortioxetine
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=Thujone&oldid=1217135267 " C a t e g o r i e s : ● A b s i n t h e ● G A B A A r e c e p t o r n e g a t i v e a l l o s t e r i c m o d u l a t o r s ● C o n v u l s a n t s ● M o n o t e r p e n e s ● 5 - H T 3 a n t a g o n i s t s ● K e t o n e s ● P e r f u m e i n g r e d i e n t s ● B i c y c l i c c o m p o u n d s ● C y c l o p e n t a n e s ● C y c l o p r o p a n e s ● I s o p r o p y l c o m p o u n d s ● N e u r o t o x i n s ● P l a n t t o x i n s H i d d e n c a t e g o r i e s : ● P a g e s u s i n g t h e P h o n o s e x t e n s i o n ● W e b a r c h i v e t e m p l a t e w a y b a c k l i n k s ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n i s d i f f e r e n t f r o m W i k i d a t a ● C h e m i c a l a r t i c l e s w i t h m u l t i p l e c o m p o u n d I D s ● M u l t i p l e c h e m i c a l s i n a n i n f o b o x t h a t n e e d i n d e x i n g ● C h e m i c a l a r t i c l e s w i t h m u l t i p l e C A S r e g i s t r y n u m b e r s ● C h e m i c a l a r t i c l e s w i t h m u l t i p l e P u b C h e m C I D s ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● C h e m b o x h a v i n g G H S d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● C h e m b o x i m a g e s i z e s e t ● P a g e s i n c l u d i n g r e c o r d e d p r o n u n c i a t i o n s ● A l l a r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m M a r c h 2 0 2 4 ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m J u l y 2 0 1 8 ● A r t i c l e s w i t h s p e c i f i c a l l y m a r k e d w e a s e l - w o r d e d p h r a s e s f r o m J a n u a r y 2 0 1 8 ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m J a n u a r y 2 0 1 8 ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m A p r i l 2 0 0 7 ● C o m m o n s c a t e g o r y l i n k i s o n W i k i d a t a
● T h i s p a g e w a s l a s t e d i t e d o n 4 A p r i l 2 0 2 4 , a t 0 1 : 2 6 ( U T C ) . ● T e x t i s a v a i l a b l e u n d e r t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e 4 . 0 ;
a d d i t i o n a l t e r m s m a y a p p l y . B y u s i n g t h i s s i t e , y o u a g r e e t o t h e T e r m s o f U s e a n d P r i v a c y P o l i c y . W i k i p e d i a ® i s a r e g i s t e r e d t r a d e m a r k o f t h e W i k i m e d i a F o u n d a t i o n , I n c . , a n o n - p r o f i t o r g a n i z a t i o n . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w