

 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Bosulif |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 94–96% |
| Metabolism | ByCYP3A4, to inactive metabolites |
| Elimination half-life | 22.5±1.7 hours |
| Excretion | Fecal (91.3%) and kidney (3%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.149.122 |
| Chemical and physical data | |
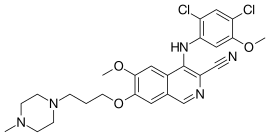
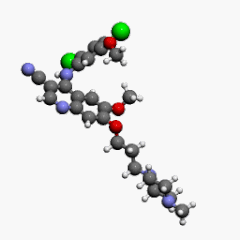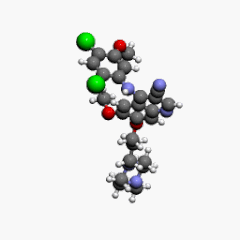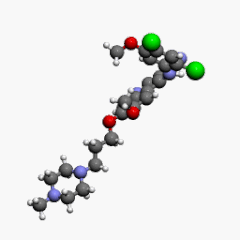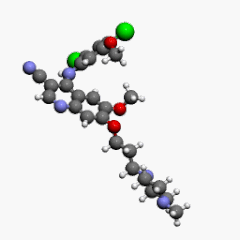
| Formula | C26H29Cl2N5O3 |
| Molar mass | 530.45 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Bosutinib, sold under the brand name Bosulif, is a small molecule BCR-ABL and src tyrosine kinase inhibitor used for the treatment of chronic myelogenous leukemia.[medical citation needed]
Originally synthesized by Wyeth, it is being developed by Pfizer.[citation needed]
It is an ATP-competitive Bcr-Abl tyrosine-kinase inhibitor with an additional inhibitory effect on Src family kinases (including Src, Lyn and Hck).[2][3] It has also shown activity against the receptors for platelet derived growth factor and vascular endothelial growth factor.[4] Bosutinib inhibited 16 of 18 imatinib-resistant forms of Bcr-Abl expressed in murine myeloid cell lines, but did not inhibit T315I and V299L mutant cells.[2]
Bosutinib is metabolized through CYP3A4.
Bosutinib received US FDA and EU European Medicines Agency approval in September 2012, and March 2013, respectively for the treatment of adults with Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML) with resistance, or intolerance to prior therapy.[5][6][7][8]
Bosutinib has two known absolute contraindications, which are: known hypersensitivity to bosutinib and liver impairment.[9][10]
Bosutinib is both a substrate and an inhibitor of P-glycoprotein (P-gp) and CYP3A4.[2] Hence P-gp and CYP3A4 inhibitors may increase plasma levels of bosutinib.[2] Likewise CYP3A4 inducers may reduce plasma concentrations of bosutinib.[2] It may also alter the metabolism and uptake (into the GIT by means of its P-gp inhibitory effects) of other drugs that are substrates for P-gp and CYP3A4.[2]
{{cite book}}: |website= ignored (help)
|
| |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiopoietin |
| ||||||||||
| CNTF |
| ||||||||||
| EGF (ErbB) |
| ||||||||||
| FGF |
| ||||||||||
| HGF (c-Met) |
| ||||||||||
| IGF |
| ||||||||||
| LNGF (p75NTR) |
| ||||||||||
| PDGF |
| ||||||||||
| RET (GFL) |
| ||||||||||
| SCF (c-Kit) |
| ||||||||||
| TGFβ |
| ||||||||||
| Trk |
| ||||||||||
| VEGF |
| ||||||||||
| Others |
| ||||||||||