J u m p t o c o n t e n t
M a i n m e n u
M a i n m e n u
N a v i g a t i o n
● M a i n p a g e ● C o n t e n t s ● C u r r e n t e v e n t s ● R a n d o m a r t i c l e ● A b o u t W i k i p e d i a ● C o n t a c t u s ● D o n a t e
C o n t r i b u t e
● H e l p ● L e a r n t o e d i t ● C o m m u n i t y p o r t a l ● R e c e n t c h a n g e s ● U p l o a d f i l e
S e a r c h
Search
A p p e a r a n c e
● C r e a t e a c c o u n t ● L o g i n
P e r s o n a l t o o l s
● C r e a t e a c c o u n t ● L o g i n
P a g e s f o r l o g g e d o u t e d i t o r s l e a r n m o r e ● C o n t r i b u t i o n s ● T a l k
( T o p )
1 B i o l o g i c a l a c t i v i t y
2 B i o c h e m i s t r y
T o g g l e B i o c h e m i s t r y s u b s e c t i o n
2 . 1 B i o s y n t h e s i s
2 . 2 M e a s u r e m e n t
3 P h a r m a c o l o g y
T o g g l e P h a r m a c o l o g y s u b s e c t i o n
3 . 1 P h a r m a c o k i n e t i c s
4 M e d i c a l u s e s
5 C h e m i s t r y
6 S o c i e t y a n d c u l t u r e
T o g g l e S o c i e t y a n d c u l t u r e s u b s e c t i o n
6 . 1 G e n e r i c n a m e s
7 S e e a l s o
8 R e f e r e n c e s
T o g g l e t h e t a b l e o f c o n t e n t s
1 7 α - H y d r o x y p r o g e s t e r o n e
1 3 l a n g u a g e s
● B o s a n s k i ● D e u t s c h ● E s p a ñ o l ● ف ا ر س ی ● F r a n ç a i s ● G a l e g o ● N e d e r l a n d s ● 日 本 語 ● P o l s k i ● Р у с с к и й ● С р п с к и / s r p s k i ● S r p s k o h r v a t s k i / с р п с к о х р в а т с к и ● 中 文
E d i t l i n k s
● A r t i c l e ● T a l k
E n g l i s h
● R e a d ● E d i t ● V i e w h i s t o r y
T o o l s
T o o l s
A c t i o n s
● R e a d ● E d i t ● V i e w h i s t o r y
G e n e r a l
● W h a t l i n k s h e r e ● R e l a t e d c h a n g e s ● U p l o a d f i l e ● S p e c i a l p a g e s ● P e r m a n e n t l i n k ● P a g e i n f o r m a t i o n ● C i t e t h i s p a g e ● G e t s h o r t e n e d U R L ● D o w n l o a d Q R c o d e ● W i k i d a t a i t e m
P r i n t / e x p o r t
● D o w n l o a d a s P D F ● P r i n t a b l e v e r s i o n
P r i n t / e x p o r t
I n o t h e r p r o j e c t s
● W i k i m e d i a C o m m o n s
A p p e a r a n c e
F r o m W i k i p e d i a , t h e f r e e e n c y c l o p e d i a
T h i s i s a n o l d r e v i s i o n o f t h i s p a g e , a s e d i t e d b y M a x i m M a s i u t i n ( t a l k | c o n t r i b s ) at 0 9 : 0 2 , 2 4 J a n u a r y 2 0 2 4 . T h e p r e s e n t a d d r e s s ( U R L ) i s a p e r m a n e n t l i n k t o t h i s r e v i s i o n , w h i c h m a y d i f f e r s i g n i f i c a n t l y f r o m t h e c u r r e n t r e v i s i o n . ( d i f f ) ← P r e v i o u s r e v i s i o n | L a t e s t r e v i s i o n ( d i f f ) | N e w e r r e v i s i o n → ( d i f f )
17α-Hydroxyprogesterone
Names
IUPAC name
17α-Hydroxypregn-4-ene-3,20-dione
Systematic IUPAC name
(1 R S R R S S H a
Other names
Hydroxyprogesterone (INN Tooltip International Nonproprietary Name )
Identifiers
CAS Number
3D model (JSmol )
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard 100.000.636
IUPHAR/BPS
KEGG
PubChem CID
UNII
CompTox Dashboard (EPA )
InChI=1S/C21H30O3/c1-13(22 )21(24 )11-8-18-16-5-4-14-12-15(23 )6-9-19(14,2)17(16 )7-10-20(18,21)3/h12,16-18,24H,4-11H2,1-3H3/t16-,17+,18+,19+,20+,21+/m1/s1
Key: DBPWSSGDRRHUNT-CEGNMAFCSA-N
CC(=O)[C@]1(CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CCC4=CC(=O)CC[C@]34C)C)O
Properties
Chemical formula
C 21 H 30 O 3
Molar mass
330.46 g/mol
Melting point
219.5
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chemical compound
17α-Hydroxyprogesterone (17α-OHP ), also known as 17-OH progesterone (17-OHP ),[1] or hydroxyprogesterone (OHP ), is an endogenous progestogen steroid hormone related to progesterone .[2] [3] [4] chemical intermediate in the biosynthesis of many other endogenous steroids, including androgens , estrogens , glucocorticoids , and mineralocorticoids , as well as neurosteroids .
Biological activity
17α-OHP is an agonist of the progesterone receptor (PR ) similarly to progesterone, albeit weakly in comparison.[5] antagonist of the mineralocorticoid receptor (MR )[6] partial agonist of the glucocorticoid receptor (GR ), albeit with very low potency (EC 50 cortisol ) at the latter site, also similarly to progesterone.[5] [7] [8]
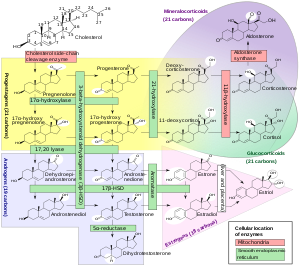
Biochemistry
Steroidogenesis , showing 17α-OHP around the left-middle among the pregnenes .
Biosynthesis
17α-OHP is derived from progesterone via 17α-hydroxylase (encoded by CYP17A1 )[citation needed
17α-OHP increases in the third trimester of pregnancy primarily due to fetal adrenal production.[citation needed
This steroid is primarily produced in the adrenal glands and to some degree in the gonads , specifically the corpus luteum of the ovary . Normal levels are 3-90 ng/dl in children, and in women, 20-100 ng/dl prior to ovulation , and 100-500 ng/dl during the luteal phase .[9] [10]
Measurement
Measurements of levels of 17α-OHP are useful in the evaluation of patients with suspected congenital adrenal hyperplasia as the typical enzymes that are defective, namely 21-hydroxylase and 11β-hydroxylase , lead to a build-up of 17α-OHP. In contrast, the rare patient with 17α-hydroxylase deficiency will have very low or undetectable levels of 17α-OHP. 17α-OHP levels can also be used to measure contribution of progestational activity of the corpus luteum during pregnancy as progesterone but note, 17α-OHP is also contributed by the placenta .[citation needed
Immunoassays like RIA (radioimmunoassay ) or IRMA (immunoradiometric
assay) used to clinically determine 17α-OHP are prone to cross-reactivity with the 17α-OHP steroid precursors and their sulphated conjugates. Gas or liquid chromatography and mass spectrometry (e.g. LC-MS/MS) achieves greater specificity than immunoassays.[11] [12]
Measurement of 17α-OHP by LC-MS/MS improves newborn screening for congenital adrenal hyperplasia due to 21-hydroxylase deficiency , because 17α-OHP steroid precursors and their sulphated conjugates which are present in the first two days after birth and longer in pre-term neonates, cross-react in immunoassays with 17α-OHP, giving falsely high 17α-OHP levels.[11] [12]
Pharmacology
Pharmacokinetics
Although 17α-OHP has not been used as a medication, its pharmacokinetics have been studied and reviewed.[13]
Medical uses
Esters of 17α-OHP, such as hydroxyprogesterone caproate and, to a far lesser extent, hydroxyprogesterone acetate and hydroxyprogesterone heptanoate , have been used in medicine as progestins .[2] [3] [4] [citation needed
Chemistry
17α-OHP, also known as 17α-hydroxypregn-4-ene-3,20-dione, is a naturally occurring pregnane steroid . It features ketone groups at the C3 and C20 positions, a hydroxyl group at the C17α position, and a double bond between the C4 and C5 positions.[citation needed
17α-OHP is the parent compound of a class of progestins referred to as the 17α-hydroxyprogesterone derivatives .[14] [15] [16] chlormadinone acetate , cyproterone acetate , hydroxyprogesterone caproate , medroxyprogesterone acetate , and megestrol acetate .[14] [15] [16]
Society and culture
Generic names
Hydroxyprogesterone is the generic name of 17α-OHP and its INN Tooltip International Nonproprietary Name and BAN Tooltip British Approved Name .[2] [3] [4]
See also
References
^ a b c I.K. Morton, Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms ISBN 978-94-011-4439-1
^ a b c Index Nominum 2000: International Drug Directory ISBN 978-3-88763-075-1
^ a b Attardi BJ, Zeleznik A, Simhan H, Chiao JP, Mattison DR, Caritis SN (2007). "Comparison of progesterone and glucocorticoid receptor binding and stimulation of gene expression by progesterone, 17-alpha hydroxyprogesterone caproate, and related progestins" . Am. J. Obstet. Gynecol . 197 (6 ): 599.e1–7. doi :10.1016/j.ajog.2007.05.024 . PMC 2278032 PMID 18060946 .
^ Mooij CF, Parajes S, Pijnenburg-Kleizen KJ, Arlt W, Krone N, Claahsen-van der Grinten HL (April 2015). "Influence of 17-Hydroxyprogesterone, Progesterone and Sex Steroids on Mineralocorticoid Receptor Transactivation in Congenital Adrenal Hyperplasia" (PDF) . Horm Res Paediatr . 83 6 ): 414–421. doi :10.1159/000374112 . PMID 25896481 . S2CID 24727940 .
^ Pijnenburg-Kleizen KJ, Engels M, Mooij CF, Griffin A, Krone N, Span PN, van Herwaarden AE, Sweep FC, Claahsen-van der Grinten HL (2015). "Adrenal Steroid Metabolites Accumulating in Congenital Adrenal Hyperplasia lead to Transactivation of the Glucocorticoid Receptor" . Endocrinology . 156 (10 ): 3504–3510. doi :10.1210/en.2015-1087 PMID 26207344 .
^ Sun K, Lei K, Chen L, Georgiou EX, Sooranna SR, Khanjani S, Brosens JJ, Bennett PR, Johnson MR (2012). "Progesterone Acts via the Nuclear Glucocorticoid Receptor to Suppress IL-1β-Induced COX-2 Expression in Human Term Myometrial Cells" . PLOS ONE . 7 11 ): e50167. Bibcode :2012PLoSO...750167L . doi :10.1371/journal.pone.0050167 ISSN 1932-6203 . PMC 3509141 PMID 23209664 .
^ Reference Values During Pregnancy
^ "normal ranges for hormone tests in women" . Archived from the original on 2020-11-08. Retrieved 2011-08-07 .
^ a b de Hora MR, Heather NL, Patel T, Bresnahan LG, Webster D, Hofman PL (March 2020). "Measurement of 17-Hydroxyprogesterone by LCMSMS Improves Newborn Screening for CAH Due to 21-Hydroxylase Deficiency in New Zealand" . International Journal of Neonatal Screening . 6 1 ): 6. doi :10.3390/ijns6010006 PMC 7422986 PMID 33073005 .
^ a b Bialk ER, Lasarev MR, Held PK (September 2019). "Wisconsin's Screening Algorithm for the Identification of Newborns with Congenital Adrenal Hyperplasia" . International Journal of Neonatal Screening . 5 3 ): 33. doi :10.3390/ijns5030033 PMC 7510207 PMID 33072992 .
^ Die Gestagene ISBN 978-3-642-99941-3
^ a b Jeffrey K. Aronson (21 February 2009). Meyler's Side Effects of Endocrine and Metabolic Drugs ISBN 978-0-08-093292-7
^ a b Robert Alan Prentky, Ann Wolbert Burgess (31 July 2000). Forensic Management of Sexual Offenders ISBN 978-0-306-46278-8
^ a b H. J. Smith, Hywel Williams (1 January 1983). Introduction to the Principles of Drug Design ISBN 978-1-4831-8350-3
t
e
PR Tooltip Progesterone receptor
Agonists
Testosterone derivatives: Progestins: 6,6-Difluoronorethisterone
6,6-Difluoronorethisterone acetate
17α-Allyl-19-nortestosterone
Allylestrenol
Altrenogest
Chloroethynylnorgestrel
Cingestol
Danazol
Desogestrel
Dienogest
Ethinylandrostenediol
Ethisterone
Ethynerone
Etonogestrel
Etynodiol
Etynodiol diacetate
Gestodene
Gestrinone
Levonorgestrel
Levonorgestrel esters (e.g., levonorgestrel butanoate )
Lynestrenol
Lynestrenol phenylpropionate
Metynodiol
Metynodiol diacetate
Norelgestromin
Norethisterone (norethindrone)
Norethisterone esters (e.g., norethisterone acetate , norethisterone enanthate )
Noretynodrel
Norgesterone
Norgestimate
Norgestrel
Norgestrienone
Norvinisterone
Oxendolone
Quingestanol
Quingestanol acetate
Tibolone
Tigestol
Tosagestin ; Anabolic–androgenic steroids: 11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone dodecylcarbonate
19-Nor-5-androstenediol
19-Nor-5-androstenedione
19-Nordehydroepiandrosterone
Bolandiol
Bolandiol dipropionate
Bolandione
Dimethisterone
Dienedione
Dienolone
Dimethandrolone
Dimethandrolone buciclate
Dimethandrolone dodecylcarbonate
Dimethandrolone undecanoate
Dimethyldienolone
Dimethyltrienolone
Ethyldienolone
Ethylestrenol (ethylnandrol)
Methyldienolone
Metribolone (R-1881)
Methoxydienone (methoxygonadiene)
Mibolerone
Nandrolone
Nandrolone esters (e.g., nandrolone decanoate , nandrolone phenylpropionate )
Norethandrolone
Normethandrone (methylestrenolone, normethandrolone, normethisterone)
RU-2309
Tetrahydrogestrinone
Trenbolone (trienolone)
Trenbolone esters (e.g., trenbolone acetate , trenbolone enanthate )
Trendione
Trestolone
Trestolone acetate
MixedSPRMs Tooltip Selective progesterone receptor modulators )
Antagonists
mPR Tooltip Membrane progesterone receptor PAQR Tooltip Progestin and adipoQ receptor )
See also
Receptor/signaling modulators
Progestogens and antiprogestogens
Androgen receptor modulators
Estrogen receptor modulators
List of progestogens
R e t r i e v e d f r o m " https://en.wikipedia.org/w/index.php?title=17α-Hydroxyprogesterone&oldid=1198514337 " C a t e g o r i e s : ● A n t i m i n e r a l o c o r t i c o i d s ● D i k e t o n e s ● G l u c o c o r t i c o i d s ● G y n a e c o l o g i c a l e n d o c r i n o l o g y ● M e t a b o l i c i n t e r m e d i a t e s ● P r e g n a n e X r e c e p t o r a g o n i s t s ● P r e g n a n e s ● P r o g e s t o g e n s H i d d e n c a t e g o r i e s : ● A r t i c l e s w i t h o u t I n C h I s o u r c e ● E C H A I n f o C a r d I D f r o m W i k i d a t a ● A r t i c l e s c o n t a i n i n g u n v e r i f i e d c h e m i c a l i n f o b o x e s ● C h e m b o x i m a g e s i z e s e t ● A r t i c l e s w i t h s h o r t d e s c r i p t i o n ● S h o r t d e s c r i p t i o n m a t c h e s W i k i d a t a ● A l l a r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s ● A r t i c l e s w i t h u n s o u r c e d s t a t e m e n t s f r o m A p r i l 2 0 2 2
● T h i s p a g e w a s l a s t e d i t e d o n 2 4 J a n u a r y 2 0 2 4 , a t 0 9 : 0 2 ( U T C ) . ● T h i s v e r s i o n o f t h e p a g e h a s b e e n r e v i s e d . B e s i d e s n o r m a l e d i t i n g , t h e r e a s o n f o r r e v i s i o n m a y h a v e b e e n t h a t t h i s v e r s i o n c o n t a i n s f a c t u a l i n a c c u r a c i e s , v a n d a l i s m , o r m a t e r i a l n o t c o m p a t i b l e w i t h t h e C r e a t i v e C o m m o n s A t t r i b u t i o n - S h a r e A l i k e L i c e n s e . ● P r i v a c y p o l i c y ● A b o u t W i k i p e d i a ● D i s c l a i m e r s ● C o n t a c t W i k i p e d i a ● C o d e o f C o n d u c t ● D e v e l o p e r s ● S t a t i s t i c s ● C o o k i e s t a t e m e n t ● M o b i l e v i e w