

 | |
| Clinical data | |
|---|---|
| Other names | trans-4-hydroxycrotonic acid |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
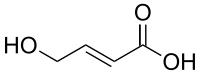
| Formula | C4H6O3 |
| Molar mass | 102.089 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
trans-4-Hydroxycrotonic acid (T-HCA), also known as γ-hydroxycrotonic acid (GHC), is an agent used in scientific research to study the GHB receptor.[1] It is an analogueofγ-hydroxybutyric acid (GHB), as well as an active metabolite of GHB.[2][3][4] Similarly to GHB, T-HCA has been found to be endogenous to the rat central nervous system, and as a metabolite of GHB, is almost certain to be endogenous to humans as well.[3][5] T-HCA binds to the high-affinity GHB receptor with 4-fold greater affinity than GHB itself,[6] where it acts as an agonist,[1][7] but does not bind to the low-affinity GHB binding site, the GABAB receptor.[3][8] Because of this, T-HCA does not produce sedation. T-HCA has been shown to cause receptor activation-evoked increases in extracellular glutamate concentrations, notably in the hippocampus.[8]
|
| |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amino acid-derived |
| ||||||||||||||||||||||
| Lipid-derived |
| ||||||||||||||||||||||
| Nucleobase-derived |
| ||||||||||||||||||||||
| Vitamin-derived |
| ||||||||||||||||||||||
| Miscellaneous |
| ||||||||||||||||||||||
|
| |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Receptor (ligands) |
| ||||||||||
| Transporter (blockers) |
| ||||||||||
| Enzyme (inhibitors) |
| ||||||||||
| |||||||||||
This drug article relating to the nervous system is a stub. You can help Wikipedia by expanding it. |