

| |
| Names | |
|---|---|
| Preferred IUPAC name
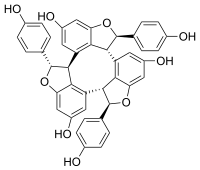
(1R,5bR,6R,10bS,11S,15bR)-1,6,11-Tris(4-hydroxyphenyl)-1,5b,6,10b,11,15b-hexahydrocyclonona[1,2,3-cd:4,5,6-c′d′:7,8,9-c′′d′′]tris([1]benzofuran)-4,9,14-triol | |
| Other names
α-Viniferin; (+)-α-Viniferin | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C42H30O9 | |
| Molar mass | 678.693 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
α-Viniferin is a stilbene trimer. It can be isolated from Caragana chamlagu[1] and from Caragana sinica[2] and from the stem bark of Dryobalanops aromatica.[3] It is also present in relation to resistance to Botrytis cinerea and Plasmopara viticolainVitis vinifera and Vitis riparia.[4] It has been shown to inhibit acetylcholinesterase.[1]
|
Oligostilbenoids and their glycosides
| |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||
| Dimers |
| ||||||||||||
| Trimers |
| ||||||||||||
| Tetramers: |
| ||||||||||||
| Higher polymers (five units or more) |
| ||||||||||||
| Oligomeric forms of resveratrol |
| ||||||||||||
| Glycosides or conjugates |
| ||||||||||||
This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |