

| |
| Names | |
|---|---|
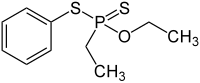
| Preferred IUPAC name
O-Ethyl S-phenyl ethylphosphonodithioate | |
| Other names
Dyfonate, Dyphonate, Fonophos | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.012.189 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H15OPS2 | |
| Molar mass | 246.32 g·mol−1 |
| Appearance | Light-yellow liquid with an aromatic odor[1] |
| Density | 1.16 g/cm3[2] |
| Boiling point | 130 °C (266 °F; 403 K) 0.13 mbar[2] |
| 0.001% (20°C)[3] | |
| Vapor pressure | 0.0002 mmHg (25°C)[3] |
| Hazards | |
| Flash point | > 94 °C; 201 °F; 367 K[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
none[3] |
REL (Recommended) |
TWA 0.1 mg/m3 [skin][3] |
IDLH (Immediate danger) |
N.D.[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Fonofos is an organothiophosphate insecticide primarily used on corn.[4] It is highly toxic[4] and listed as an extremely hazardous substance. [5]
At room temperature, fonofos has a clear-to-yellow color. It has a distinct Mercaptan odour. It is soluble in most common organic Solvents. It is available in multiple forms, including granular, microgranular,
This article about an ester is a stub. You can help Wikipedia by expanding it. |