

| |
| Names | |
|---|---|
| IUPAC name
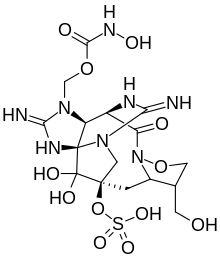
[(3R,5S,6S,11R,12S,14Z,16S,17Z)-14,17-Diamino-19,19-dihydroxy-6-(hydroxymethyl)-10-oxo-3-(sulfooxy)-8-oxa-1,9,13,15,18-pentaazapentacyclo[9.5.2.1~3,16~.0~5,9~.0~12,16~]nonadeca-14,17-dien-13-yl]methyl hydroxycarbamate | |
| Other names
ZTX | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C16H24N8O12S | |
| Molar mass | 552.47 g·mol−1 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Extremely toxic |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
11 μg/kg (mice) |
| Related compounds | |
Related compounds |
Saxitoxin Neosaxitoxin Tetrodotoxin |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Zetekitoxin AB (ZTX) is a guanidine alkaloid found in the Panamanian golden frog Atelopus zeteki. It is an extremely potent neurotoxin.
ZTX is a guanidine alkaloid. It's structurally related to saxitoxin, but with some differences. ZTX contains an isoxazolidine ring, a sulfonate group and an N-hydroxycarbamate group.[2]
ZTX is an extremely potent sodium channel blocker. It has been shown to block the voltage-gated sodium channelsatpicomolar concentrations. It is about 580 times more potent than saxitoxin.[2]
ZTX is an extremely potent neurotoxin. The LD50 of ZTX in mice is 11 μg/kg.[3]
|
| |
|---|---|
| Animal toxins |
|
| Bacterial |
|
| Cyanotoxins |
|
| Plant toxins |
|
| Mycotoxins |
|
| Pesticides |
|
| Nerve agents |
|
| Bicyclic phosphates |
|
| Cholinergic neurotoxins |
|
| Other |
|
|
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Calcium |
| ||||||||||||||||||||||||
| Potassium |
| ||||||||||||||||||||||||
| Sodium |
| ||||||||||||||||||||||||
| Chloride |
| ||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||
See also: Receptor/signaling modulators • Transient receptor potential channel modulators | |||||||||||||||||||||||||
This neurotoxin article is a stub. You can help Wikipedia by expanding it. |