

| Neuropilin | |
|---|---|
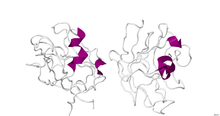
Crystallographic structure of the dimeric B1 domain of human neuropilin 1.[1]
| |
| Identifiers | |
| Symbol | NRP |
| InterPro | IPR014648 |
| Membranome | 16 |
| neuropilin 1 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | NRP1 | ||||||
| NCBI gene | 8829 | ||||||
| HGNC | 8004 | ||||||
| OMIM | 602069 | ||||||
| PDB | 3I97 | ||||||
| RefSeq | NM_001024628 | ||||||
| UniProt | O14786 | ||||||
| Other data | |||||||
| Locus | Chr. 10 p12 | ||||||
| |||||||
| neuropilin 2 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | NRP2 | ||||||
| NCBI gene | 8828 | ||||||
| HGNC | 8005 | ||||||
| OMIM | 602070 | ||||||
| RefSeq | NM_201279 | ||||||
| UniProt | O60462 | ||||||
| Other data | |||||||
| Locus | Chr. 2 q34 | ||||||
| |||||||
Neuropilin is a protein receptor active in neurons.
There are two forms of Neuropilins, NRP-1 and NRP-2. Neuropilins are transmembrane glycoproteins, first documented to regulate neurogenesis and angiogenesis by complexing with Plexin receptors/class-3 semaphorin ligands and Vascular Endothelial Growth Factor (VEGF) receptors/VEGF ligands, respectively.[2][3] Neuropilins predominantly act as co-receptors as they have a very small cytoplasmic domain and thus rely upon other cell surface receptors to transduce their signals across a cell membrane.[2][3] Recent studies have shown that Neuropilins are multifunctional and can partner with a wide variety of transmembrane receptors. Neuropilins are therefore associated with numerous signalling pathways including those activated by Epidermal Growth Factor (EGF), Fibroblast Growth Factor (FGF), Hepatocyte Growth Factor (HGF), Insulin-like Growth Factor (IGF), Platelet Derived Growth Factor (PDGF) and Transforming Growth Factor beta (TGFβ).[4][5] Although Neuropilins are commonly found at the cell surface, they have also been reported within the mitochondria and nucleus.[6][7] Both Neuropilin family members can also be found in soluble forms created by alternative splicing or by ectodomain shedding from the cell surface.[8][9]
The pleiotropic nature of the NRP receptors results in their involvement in cellular processes, such as axon guidance and angiogenesis, the immune response and remyelination.[10] Therefore, dysregulation of NRP activity has been implicated in many pathological conditions, including many types of cancer and cardiovascular disease.[11][12][13][14]
Neuropilin-1 is a therapeutic target protein in the treatment for leukemia and lymphoma, since It has been shown that there is increased expression in neuropilin-1 in leukemia and lymphoma cell lines.[15] Also, antagonism of neuropilin-1 has been found to inhibit tumour cell migration and adhesion.[16]
Neuropilins contain the following four domains:
The structure of B1 domain (coagulation factor 5/8 type) of neuropilin-1 was determined through X-Ray Diffraction with a resolution of 2.90 Å. The secondary structure of this domain is 5% alpha helical and 46% beta sheet.[1]
Ramachandran plot.[17]
|
| |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G protein–coupled receptor |
| ||||||||||||
| Ligand-gated ion channel |
| ||||||||||||
| Enzyme-linked receptor |
| ||||||||||||
| Other/ungrouped |
| ||||||||||||