

This article relies largely or entirely on a single source. Relevant discussion may be found on the talk page. Please help improve this articlebyintroducing citations to additional sources.
Find sources: "Brodimoprim" – news · newspapers · books · scholar · JSTOR (August 2014) |
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.054.745 |
| Chemical and physical data | |
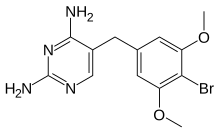
| Formula | C13H15BrN4O2 |
| Molar mass | 339.193 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 225 to 228 °C (437 to 442 °F) |
| |
| |
| (verify) | |
Brodimoprim is a structural derivative of trimethoprim. In brodimoprim, the 4-methoxy group of trimethoprim is replaced with a bromine atom.
As trimethoprim, brodimoprim is a selective inhibitorofbacterial dihydrofolate reductase.[1]
|
| |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antifolates (inhibit bacterial purine metabolism, thereby inhibiting DNA and RNA synthesis) |
| ||||||||||||||||
| Quinolones (inhibit bacterial topoisomerase and/or DNA gyrase, thereby inhibiting DNA replication) |
| ||||||||||||||||
| Anaerobic DNA inhibitors |
| ||||||||||||||||
| RNA synthesis |
| ||||||||||||||||
| |||||||||||||||||
This systemic antibiotic-related article is a stub. You can help Wikipedia by expanding it. |