

| |

| |
| Names | |
|---|---|
| IUPAC name
Samarium(II) chloride | |
| Other names
Samarium dichloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.034.196 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| SmCl2 | |
| Molar mass | 221.27 g/mol |
| Appearance | dark brown crystals[1] |
| Density | 3.69 g/cm3, solid |
| Melting point | 855 °C (1,571 °F; 1,128 K) |
| Boiling point | 1,310 °C (2,390 °F; 1,580 K) |
| ? | |
| Structure | |
| Orthorhombic | |
| Pbnm, No. 62[2] | |
| Related compounds | |
Other anions |
Samarium(II) bromide Samarium(II) iodide |
Other cations |
Samarium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Samarium(II) chloride (SmCl2) is a chemical compound, used as a radical generating agent in the ketone-mediated intraannulation reaction.
Reduction of samarium(III) chloride with samarium metal in a vacuum at a temperature of 800 °C to 900 °C, or with hydrogen gas at 350 °C yields samarium(II) chloride:[1]
Samarium(II) chloride can also be prepared by reducing samarium(III) chloride with lithium metal/naphthaleneinTHF:[3]
A similar reaction has been observed with sodium.[2]
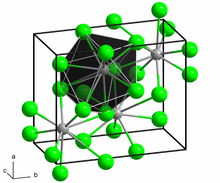
Samarium(II) chloride adopts the PbCl2 (cotunnite) structure.[2]
|
| |||
|---|---|---|---|
| Samarium(II) |
| ||
| Samarium(III) |
| ||
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
This inorganic compound–related article is a stub. You can help Wikipedia by expanding it. |